J Korean Ophthalmol Soc.
2011 Feb;52(2):233-240.
Effect of Mitomycin C and 5-Fluorouracil on Cultured Human Nasal Mucosa Fibroblasts
- Affiliations
-
- 1Department of Ophthalmology, Catholic University of Daegu School of Medicine, Daegu, Korea. kimkh@cu.ac.kr
- 2Department of Ophthalmology, Kyungpook National University School of Medicine, Daegu, Korea.
Abstract
- PURPOSE
To investigate the effects of two antimetabolites, mitomycin C (MMC) and 5-fluorouracil (5-FU), on proliferation of cultured human nasal mucosa fibroblasts.
METHODS
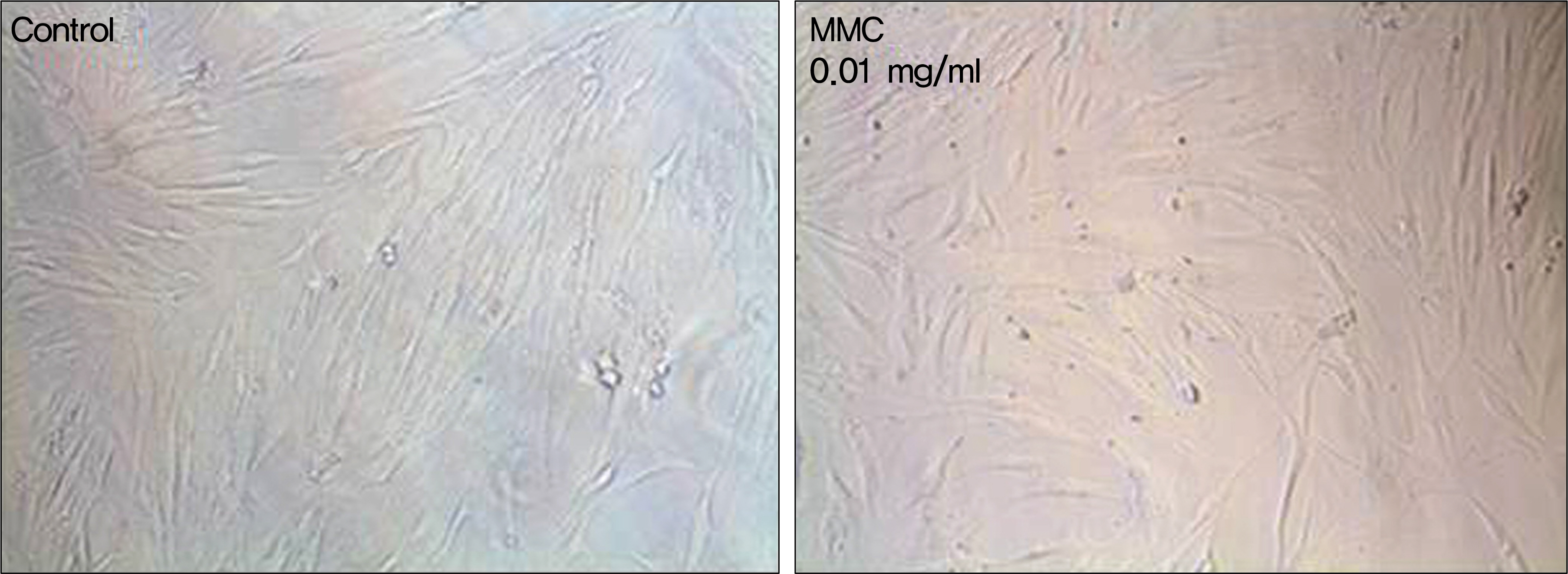
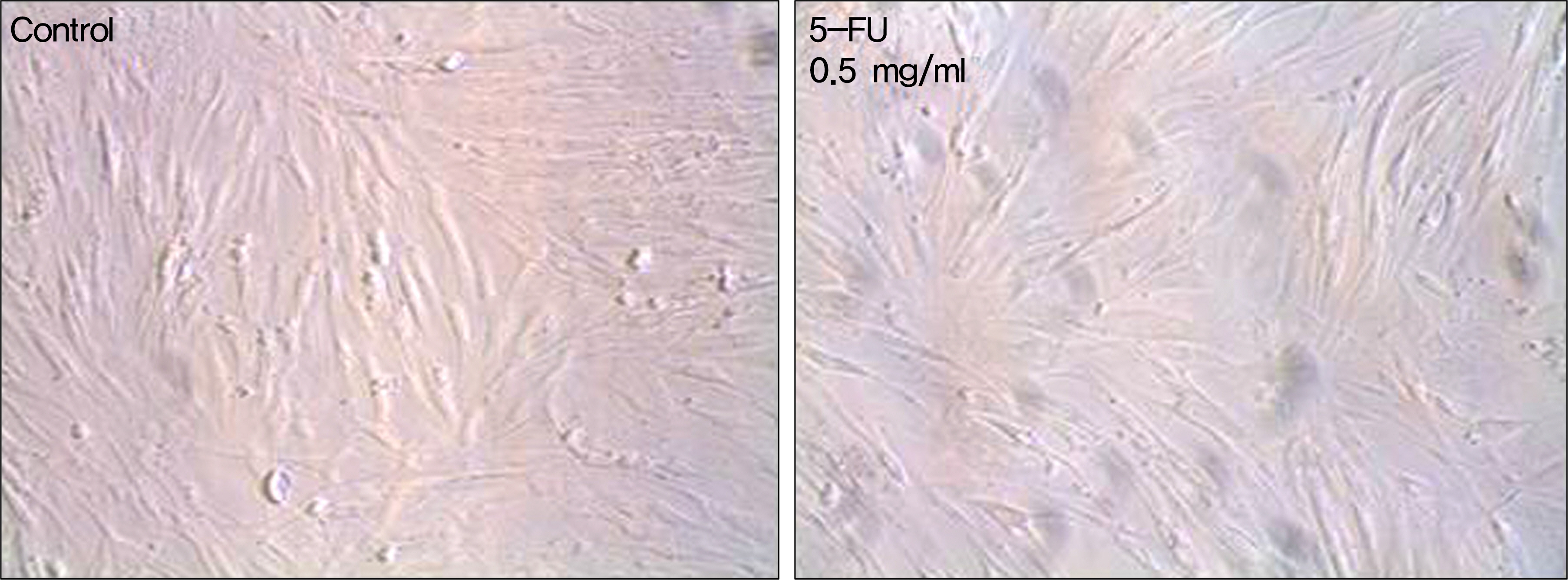
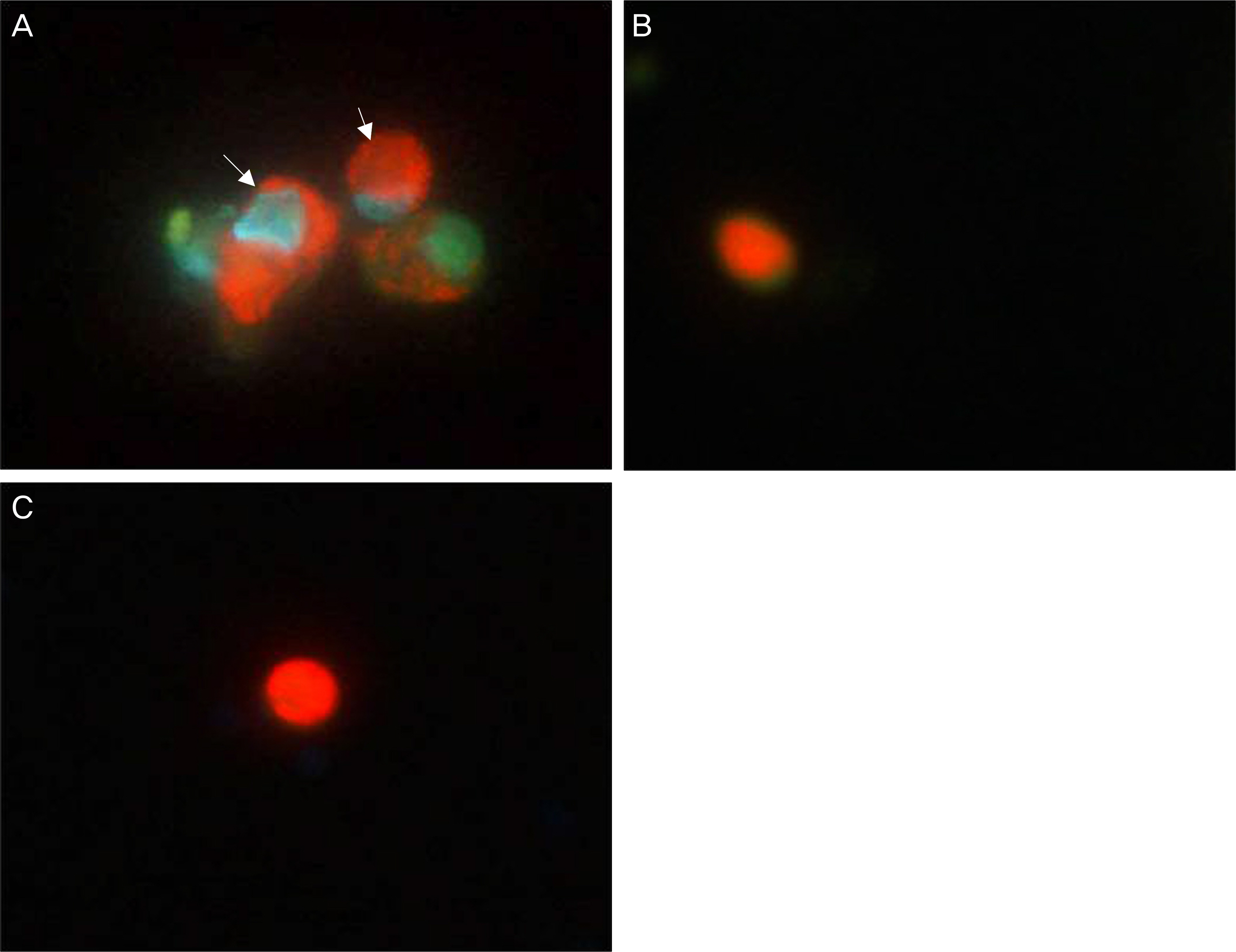
Human nasal mucosa fibroblasts were primarily cultured, and exposed to various concentrations of MMC and 5-FU for 5 minutes. Control fibroblasts were exposed to only DMEM media without the drugs. Effect of drugs on cell morphology was observed by phase-contrast microscopy. Cell viability and apoptosis were measured using MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl-tetrazolium bromide] assay and Acridine orange/Hoechst (AO/HO) staining, respectively.
RESULTS
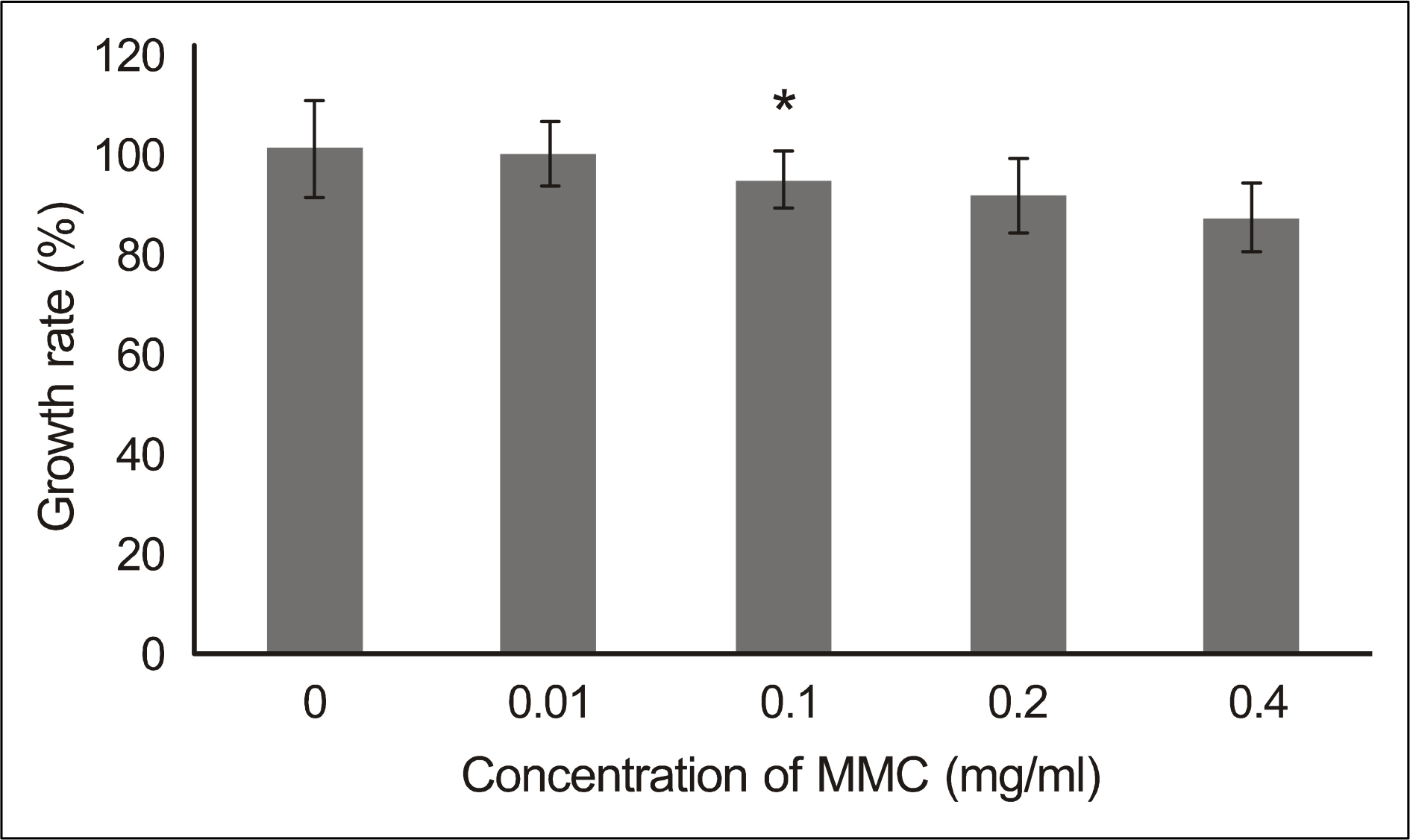
In both experimental groups exposed to MMC and 5-FU, fibroblasts maintained standard spindle shape. The MTT assay showed that both MMC and 5-FU inhibited fibroblast proliferation in a dose dependent manner. AO/HO staining showed apoptotic cells in both experimental groups.
CONCLUSIONS
Both MMC and 5-FU have an antiproliferative effect on fibroblasts in vitro at least through induction of apoptosis. Therefore, adjuvant use of either MMC or 5-FU during endonasal dacryocystorhinostomy may improve the clinical outcome by inhibiting proliferation of the nasal mucosa.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Caldwell GW. Two new operations for the radical cure of obstruction of the nasal duct with preservation of the canalicuoli and an incidental description of a new lacrimal probe. NY Med J. 1893; 57:581.2. McDonogh M, Meiring JH. Endoscopic transnasal dacryocystorhinostomy. J Laryngol Otol. 1989; 103:585–7.
Article3. Woog JJ, Kennedy RH, Custer PL, et al. Endonasal dacryocystorhinostomy: a report by the American Academy of Ophthalmology. Ophthalmology. 2001; 108:2369–77.4. Lee HC, Chung WS. Success rate of endonasal Dacryocystorhinostomy. J Korean Ophthalmol Soc. 1996; 37:211–8.5. Lee SH, Chung WS. Long term surgical efficacy of endonasal dacryocystorhinostomy. J Korean Ophthalmol Soc. 2000; 41:307–13.6. Lee TS, Kim JS, Kim JK. The effect of double silicone tube intubation on surgical outcome of endonasal dacryocystorhinostomy. J Korean Ophthalmol Soc. 2002; 43:2089–94.7. Allen K, Berlin AJ. Dacryocystorhinostomy failure: association with nasolacrimal silicone intubation. Ophthalmic Surg. 1989; 20:486–9.
Article8. Zolli CL, Shannon GM. Dacryocystorhinostomy: a review of 119 cases. Ophthalmic Surg. 1982; 13:905–10.
Article9. McLachlan DL, Shannon GM, Flanagan JC. Results of dacryocystorhinostomy: analysis of the reoperation. Ophthalmic Surg. 1980; 11:427–30.10. Tarbet KJ, Custer PL. External dacryocystorhinostomy. Surgical success, patient satisfaction, and economic cost. Ophthalmology. 1995; 102:1065–70.11. Walland MJ, Rose GE. Factors affecting the success rate of open lacrimal surgery. Br J Ophthalmol. 1994; 78:888–91.
Article12. Kao SC, Liao CL, Tseng JH, et al. Dacryocystorhinostomy with intraoperative mitomycin C. Ophthalmology. 1997; 104:86–91.
Article13. Yalaz SC, Firinciogullari E, Zeren H. Use of mitomycin C and 5-fluorouracil in external dacryocystorhinostomy. Orbit. 1999; 18:239–45.
Article14. Kim JH, Kim KS, Yoon HC, et al. Anti-adhesive effect of GUARDIX-SL® after endoscopic sinus surgery. Korean J Otolaryngol- Head Neck Surg. 2005; 48:1478–83.15. Lee TS, Lee KC. The effect of Mitomycin-C eyedrop on pre-vention of internal ostium obstruction after endonasal dacryocystorhinostomy. J Korean Ophthalmol Soc. 1998; 39:1915–20.16. Ugurbas SH, Zilelioglu G, Sargon MF, et al. Histopathologic effects of mitomycin-C on endoscopic transnasal dacryocystorhinostomy. Ophthalmic Surg Lasers. 1997; 28:300–4.
Article17. Zilelioğ lu G, Uğ urbaş SH, Anadolu Y, et al. Adjunctive use of mitomycin C on endoscopic lacrimal surgery. Br J Ophthalmol. 1998; 82:63–6.18. Roozitalab MH, Amirahmadi M, Namazi MR. Results of the application of intraoperative mitomycin C in dacryocystorhinostomy. Eur J Ophthalmol. 2004; 14:461–3.
Article19. Iliff CE. A simplified dacryocystorhinostomy. 1954-1970. Arch Ophthalmol. 1971; 85:586–91.20. Watts P, Ram AR, Nair R, Williams H. Comparison of external dacryocystorhinostomy and 5-fluorouracil augmented endonasal laser dacryocystorhinostomy. A retrospective review. Indian J Ophthalmol. 2001; 49:169–72.21. Hu D, Sires BS, Tong DC, et al. Effect of brief exposure to mitomycin C on cultured human nasal mucosa fibroblasts. Ophthal Plast Reconstr Surg. 2000; 16:119–25.
Article22. Givens KT, Kitada S, Chen AK, et al. Proliferation of human ocular fibroblasts. An assessment of in vitro colorimetric assays. Invest Ophthalmol Vis Sci. 1990; 31:1856–62.23. Senderoff RI, Weber PA, Smith DR, Sokoloski TD. Evaluation of antiproliferative agents using a cell-culture model. Invest Ophthalmol Vis Sci. 1990; 31:2572–8.24. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983; 65:55–63.
Article25. Mpoke SS, Wolfe J. Differential staining of apoptotic nuclei in living cells: Application to macronuclear elimination in Tetrahymena. J Histochem Cytochem. 1997; 45:675–83.26. Gilman AG, Goodman LS, Gilman A. The Pharmacological Basis of Therapeutics. 6th ed.New York: Macmillan Publishing Co.;1980. p. 1295–300.27. Dushinski R, Pleven E, Heidelberger C. The synthesis of 5 fluoro pyrimidines. J Am Chem Soc. 1957; 79:4559–60.28. Blumenkranz MS, Ophir A, Claflin AJ, Hajek A. Fluorouracil for the treatment of massive periretinal proliferation. Am J Ophthalmol. 1982; 94:458–67.
Article29. Abraham LM, Selva D, Casson R, Leibovitch I. The clinical appli cations of fluorouracil in ophthalmic practice. Drugs. 2007; 67:237–55.30. Yamamoto T, Varani J, Soong HK, Lichter PR. Effects of 5-fluo-rouracil and mitomycin C on cultured rabbit subconjunctival fibroblasts. Ophthalmology. 1990; 97:1204–10.
Article31. Singh K, Mehta K, Shaikh NM, et al. Trabeculectomy with intraoperative mitomycin C versus 5-fluorouracil. Prospective randomized clinical trial. Ophthalmology. 2000; 107:2305–9.32. Suh WJ, Chung WS. Effects of mitomycin C on cultured rabbit osteoblasts. J Korean Ophthalmol Soc. 2001; 42:1464–9.33. Lee JH, Chung WS, Son JH. The effect of mitomycin C on cultured human osteoblasts. J Korean Ophthalmol Soc. 2003; 44:2122–7.34. Huang L, Wong YP, Cai YJ. Low dose 5-fluorouracil induces cell cycle G2 arrest and apoptosis in keloid fibroblasts. Br J Dermatol. 2010; 163:1181–5.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Amniotic Membrane Extract on Cultured Human Nasal Mucosa Fibroblasts
- Comparison of Inhibitory Effects of 5-Fluorouracil and Mitomycin C on Proliferation of Fibroblagts and Retinal Pigment Epithelial Cells
- Evaluation of Antiproliferative Agents in Tissue Culture Model
- The Effect of Topical Mitomycin-C on the Patency of Internal Nasal Ostium of the Experimental Antrorhinostomy in Rabbits
- Comparison of the Effects Between Bevacizumab and Mitomycin C on the Survival of Fibroblasts