J Korean Ophthalmol Soc.
2015 Dec;56(12):1939-1944. 10.3341/jkos.2015.56.12.1939.
Effect of Amniotic Membrane Extract on Cultured Human Nasal Mucosa Fibroblasts
- Affiliations
-
- 1Department of Ophthalmology, Catholic University of Daegu School of Medicine, Daegu, Korea. kimkh@cu.ac.kr
- KMID: 2148758
- DOI: http://doi.org/10.3341/jkos.2015.56.12.1939
Abstract
- PURPOSE
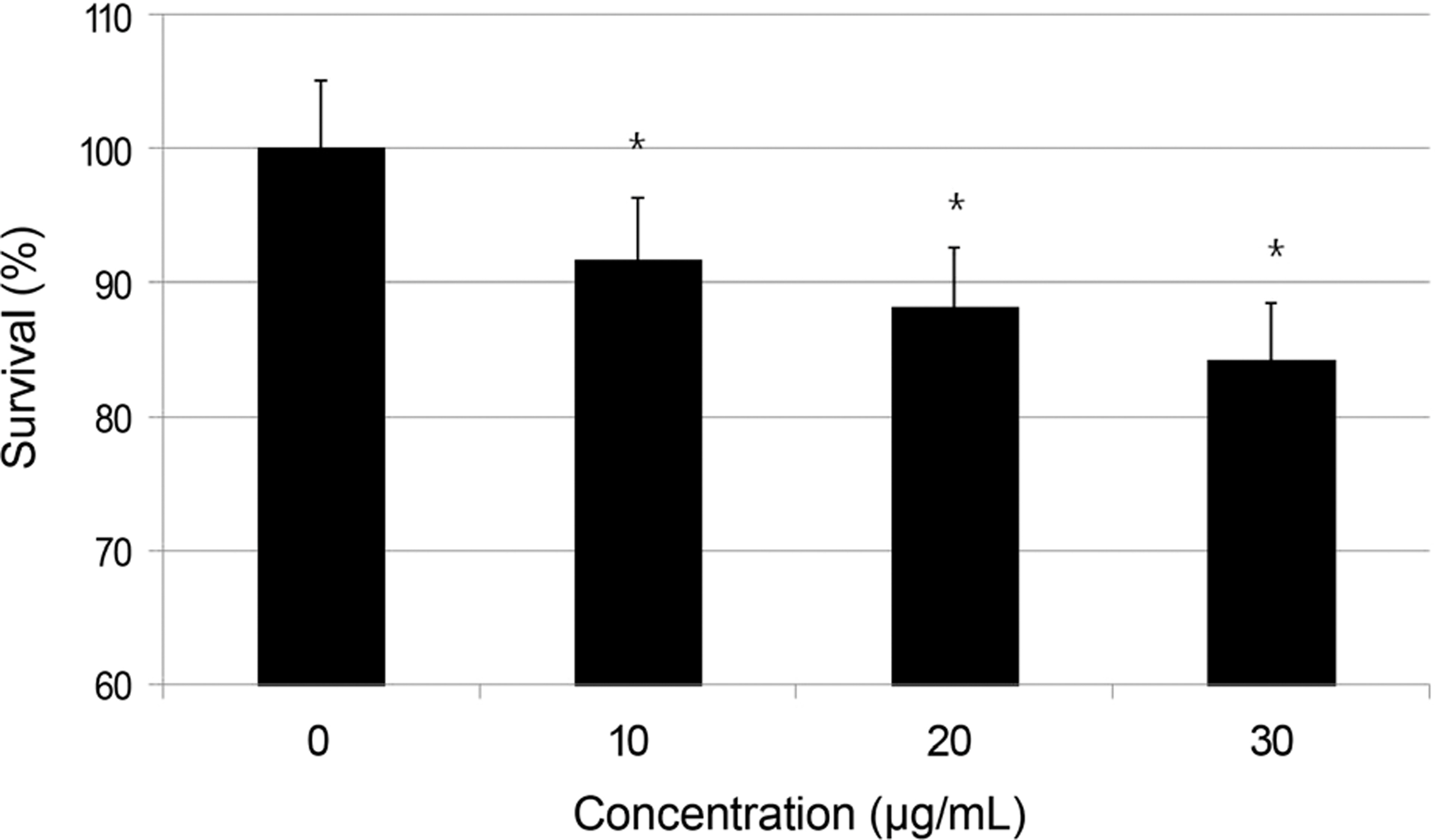
To investigate the effects of amniotic membrane extract (AME) on the survival of cultured human nasal mucosa fibroblasts.
METHODS
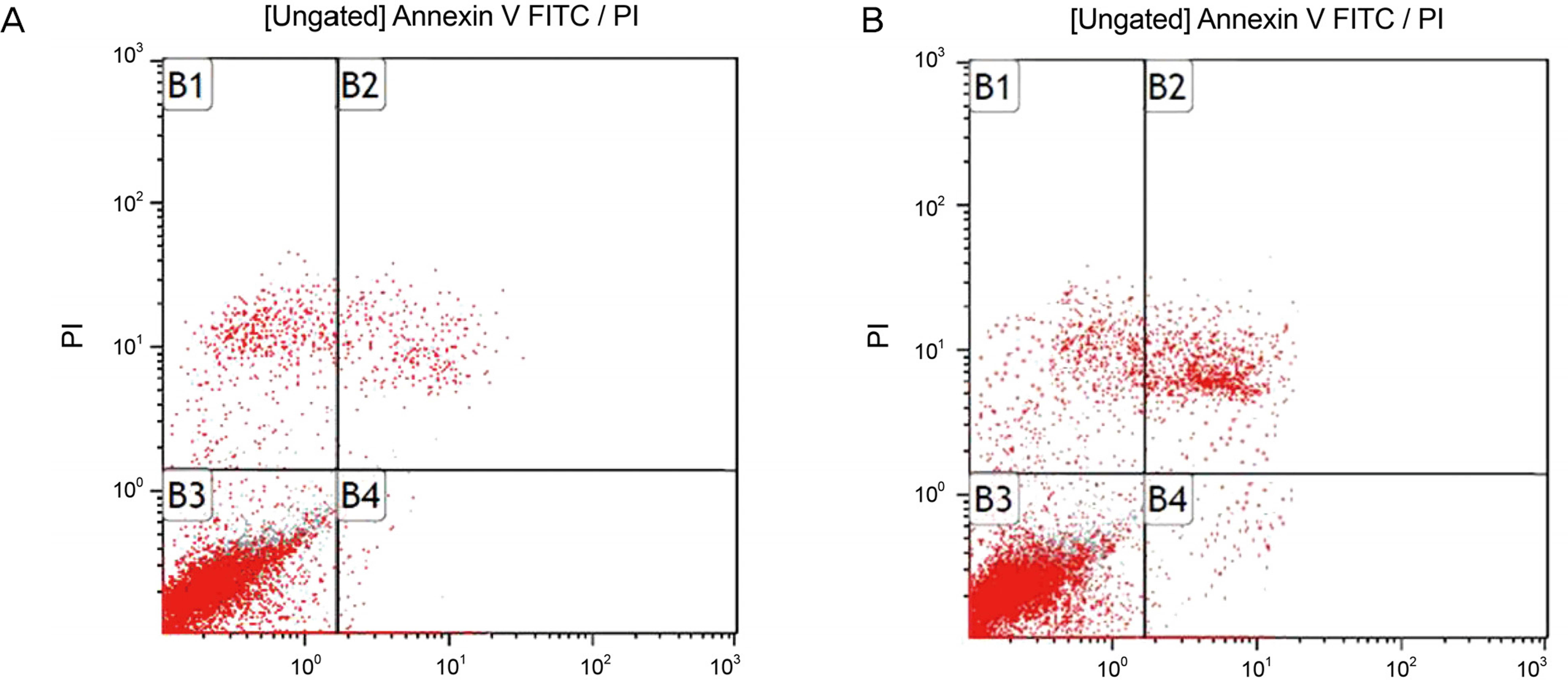
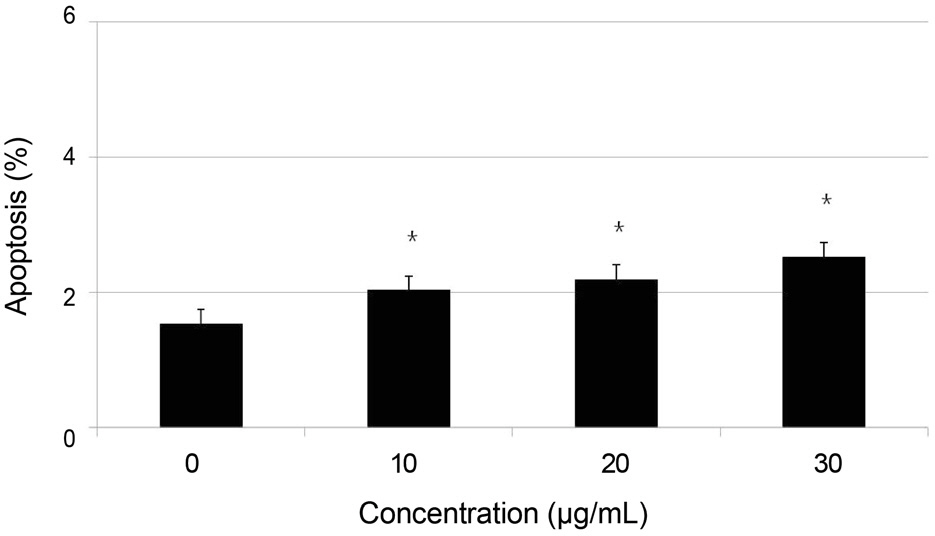
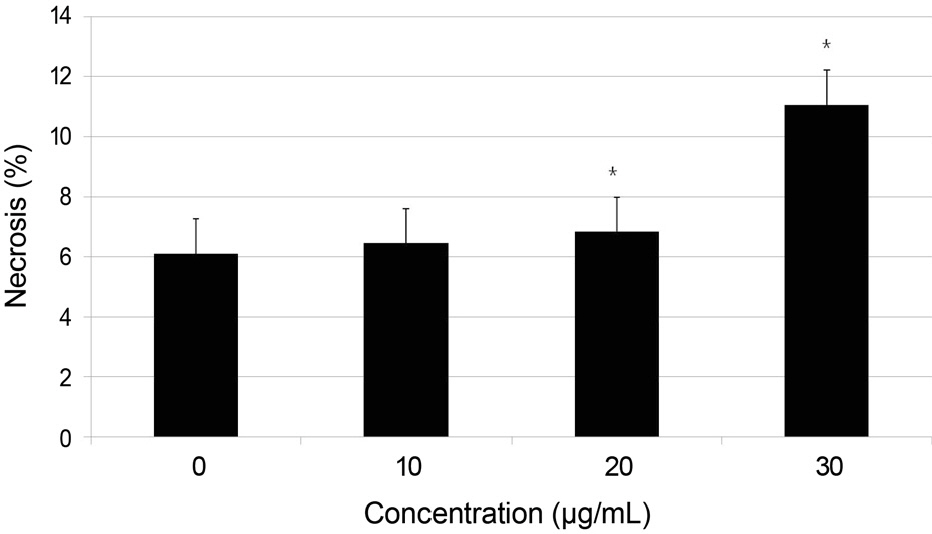
Primary cultured human nasal mucosa fibroblasts were exposed to 0, 10, 20, or 30 microg/mL AME for 3 days. The survival of the human nasal mucosa fibroblasts was measured using the MTT assay and apoptosis was evaluated with flow cytometry using annexin-V/propidium iodide double staining.
RESULTS
AME decreased significantly in fibroblast proliferation after exposure to 10 microg/mL (p = 0.000), and caused significant apoptosis of the fibroblasts after exposure to 10 microg/mL (p = 0.024).
CONCLUSIONS
AME decreased fibroblast proliferation in vitro at least through induction of apoptosis. Therefore, adjuvant use of AME during endonasal dacryocystorhinostomy may improve clinical outcomes.
Figure
Reference
-
References
1. Allen K, Berlin AJ. Dacryocystorhinostomy failure: association with nasolacrimal silicone intubation. Ophthalmic Surg. 1989; 20:486–9.
Article2. Zolli CL, Shannon GM. Dacryocystorhinostomy: a review of 119 cases. Ophthalmic Surg. 1982; 13:905–10.
Article3. McLachlan DL, Shannon GM, Flanagan JC. Results of dacryocys-torhinostomy: analysis of the reoperations. Ophthalmic Surg. 1980; 11:427–30.4. Tarbet KJ, Custer PL. External dacryocystorhinostomy, Surgical success, patient satisfaction, and economic cost. Ophthalmology. 1995; 102:1065–70.5. Walland MJ, Rose GE. Factors affecting the success rate of open lacrimal surgery. Br J Ophthalmol. 1994; 78:888–91.
Article6. Kao SC, Liao CL, Tseng JH. . Dacryocystorhinostomy with in-traoperative mitomycin C. Ophthalmology. 1997; 104:86–91.
Article7. Yalaz M, Firinciogullari E, Zeren H. Use of mitomycin C and 5-flu-orouracil in external dacryocystorhinostomy. Orbit. 1999; 18:239–45.
Article8. Kim JH, Kim KS, Yoon HC. . Anti-adhesive effect of GUARDIX-SL(R) after endoscopic sinus surgery. Korean J Otolaryngol-Head Neck Surg. 2005; 48:1478–83.9. Choi YJ, Hwang SJ, Lee TS. Short-term clinical results of amniotic membrane application to endonasal dacryocystorhinostomy. J Korean Ophthalmol Soc. 2008; 49:384–9.
Article10. De Rötth A. Plastic repair of conjunctival defects with fetal membranes. Arch Ophthalmol. 1940; 23:522–5.11. Tseng SC, Prabhasawat P, Lee SH. Amniotic membrane trans-plantation for conjunctival surface reconstruction. Am J Ophthalmol. 1997; 124:765–74.
Article12. Houlihan JM, Biro PA, Harper HM. . The human amnion is a site of MHC class Ib expression: evidence for the expression of HLA-E and HLA-G. J Immunol. 1995; 154:5665–74.13. Kim JC, Tseng SC. The effects on inhibition of corneal neo-vascularization after human amniotic membrane transplantation in severely damaged rabbit corneas. Korean J Ophthalmol. 1995; 9:32–46.
Article14. Prabhasawat P, Barton K, Burkett G, Tseng SC. Comparison of conjunctival autografts, amniotic membrane grafts, and primary closure for pterygium excision. Ophthalmology. 1997; 104:974–85.
Article15. Tseng SC, Prabhasawat P, Barton K. . Amniotic membrane transplantation with or without limbal allografts for corneal sur-face reconstruction in patients with limbal stem cell deficiency. Arch Ophthalmol. 1998; 116:431–41.
Article16. Lee SH, Tseng SC. Amniotic membrane transplantation for persis-tent epithelial defects with ulceration. Am J Ophthalmol. 1997; 123:303–12.
Article17. Mosmann T. Rapid colorimetric assay for cellular growth and sur-vival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983; 65:55–63.
Article18. Davis JW. Skin transplantation with a review of 550 cases at the Johns Hopkins Hospital. Johns Hopkins Med J. 1910; 15:307.19. Meller D, Pires RT, Tseng SC. Ex vivo preservation and expansion of human limbal epithelial stem cells on amniotic membrane cultures. Br J Ophthalmol. 2002; 86:463–71.
Article20. Meller D, Tseng SC. Conjunctival epithelial cell differentiation on amniotic membrane. Invest Ophthalmol Vis Sci. 1999; 40:878–86.21. Hao Y, Ma DH, Hwang DG. . Identification of antiangiogenic and antiinflammatory proteins in human amniotic membrane. Cornea. 2000; 19:348–52.
Article22. van Herendael BJ, Oberti C, Brosens I. Microanatomy of the hu-man amniotic membranes. A light microscopic, transmission, and scanning electron microscopic study. Am J Obstet Gynecol 1978; 131. 872–80.23. Dua HS, Azuara-Blanco A. Amniotic membrane transplantation. Br J Ophthalmol. 1999; 83:748–52.24. Li DQ, Lee SB, Tseng SC. Differential expression and regulation of TGF-beta1, TGF-beta2, TGF-beta3, TGF-betaRI, TGF-betaRII and TGF-betaRIII in cultured human corneal, limbal, and con-junctival fibroblasts. Curr Eye Res. 1999; 19:154–61.25. Tseng SC, Li DQ, Ma X. Suppression of transforming growth fac-tor-beta isoforms, TGF-beta receptor type II, and myofibroblast differentiation in cultured human corneal and limbal fibroblasts by amniotic membrane matrix. J Cell Physiol. 1999; 179:325–35.26. Lee KH, Lee DW, Kim IC. The clinical effect of amniotic mem-brane transplantation for various ocular surface diseases. J Korean Ophthalmol Soc. 2007; 48:7–12.27. Oh DH, Kwon MS, Kim JC. Five layered reinforcing amniotic membrane transplantation for treatment of deep corneal ulcer or perforation. J Korean Ophthalmol Soc. 2011; 52:1232–7.28. Jang JH, Choi TH. The effect of amniotic membrane trans-plantation for pterygium excision. J Korean Ophthalmol Soc 2005; 46. 597–604.29. Lee DY, Park WC, Rho SH. The effect of amniotic membrane oint-ment application on photore fractive keratectomized cornea in rabbits. J Korean Ophthalmol Soc. 2000; 41:2069–77.30. Portera-Cailliau C, Hedreen JC, Price DL, Koliatsos VE. Evidence for apoptotic cell death in Huntington disease and excitotoxic ani-mal models. J Neurosci. 1995; 15:(5 Pt 2). 3775–87.
Article31. Charriaut-Marlangue C, Aggoun-Zouaoui D, Represa A, Ben-Ari Y. Apoptotic features of selective neuronal death in ischemia, epi-lepsy and gp 120 toxicity. Trends Neurosci. 1996; 19:109–14.32. Leist M, Gantner F, Bohlinger I. . Tumor necrosis factor-in-duced hepatocyte apoptosis precedes liver failure in experimental murine shock models. Am J Pathol. 1995; 146:1220–34.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Mitomycin C and 5-Fluorouracil on Cultured Human Nasal Mucosa Fibroblasts
- Effect of Amniotic Membrane Extract on the Tenon's Capsule Fibroblasts
- Therapeutic Effect of Amniotic Membrane Extract on Keratitis Following Corneal Alkali Burn
- The Differential Gene Expression in Corneal Fibroblast Cultured with Amniotic Membrane Components
- Effect of Amniotic Membrane to Reduce Postlaminectomy Epidural Adhesion on a Rat Model