J Korean Soc Hypertens.
2012 Sep;18(3):117-125.
Cardiac Mitochondrial Integrity Is Regulated by CR6-interacting Factor 1 in the Heart
- Affiliations
-
- 1Department of Internal Medicine, Cardiovascular Center, Chungnam National University Hospital, Chungnam National University School of Medicine, Daejeon, Korea. jojeong@cnu.ac.kr
Abstract
- BACKGROUND
The major cause of metabolic syndrome and diabetes is reduced cellular performances in fuel metabolism, but the underlying pathways and mechanisms are not completely understood. Dysregulation of energy homeostasis can lead to metabolic disturbances and it predisposes diabetes, cardiovascular disease, aging, and cancer. CR6-interacting factor 1 (CRIF1) contacts coiled-coil domain that is required for both genomic stability and mitochondrial integrity. We performed this study to determine the role of CRIF1 on the mice hearts.
METHODS
CRIF1-deficient mouse was embryonic lethal and we made heart specific CRIF1-deficient mouse using Cre-loxP system. We made thoracotomy and directly injected adeno-Cre virus into the heart of CRIF1-loxP mice. Beta-gal virus was used as a control.
RESULTS
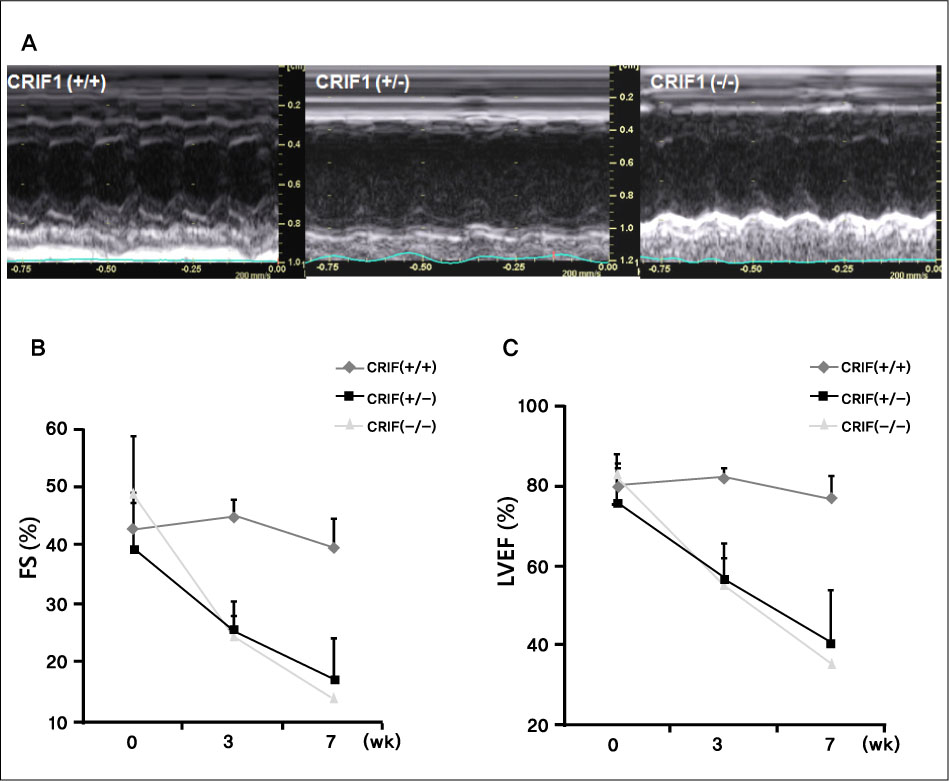
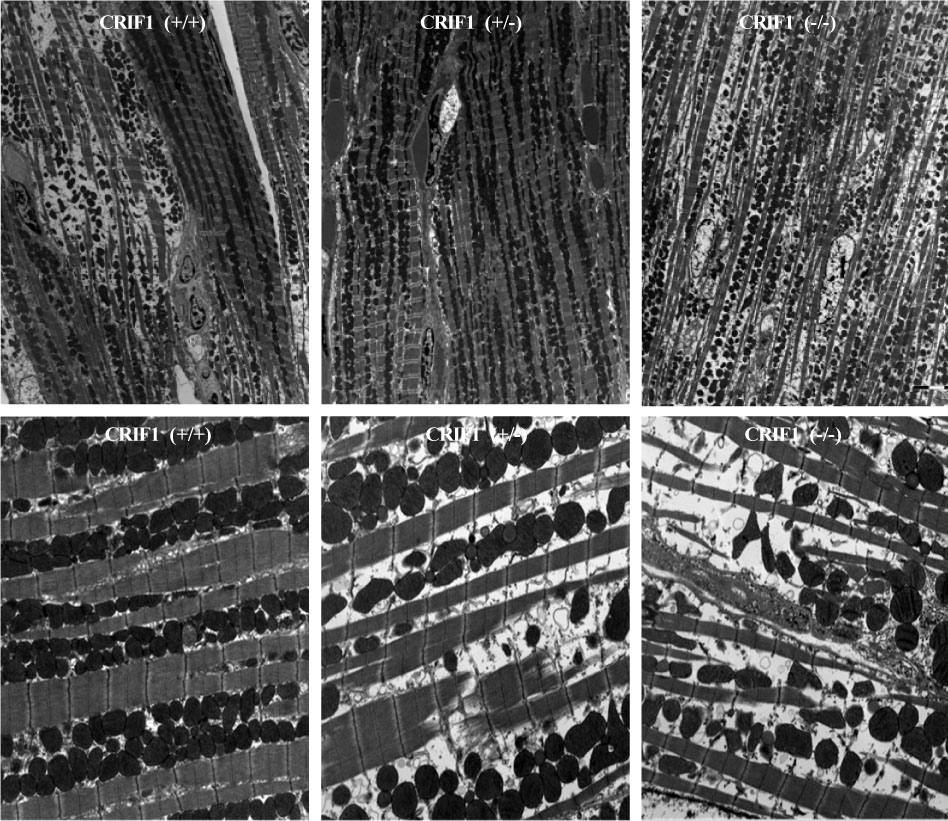
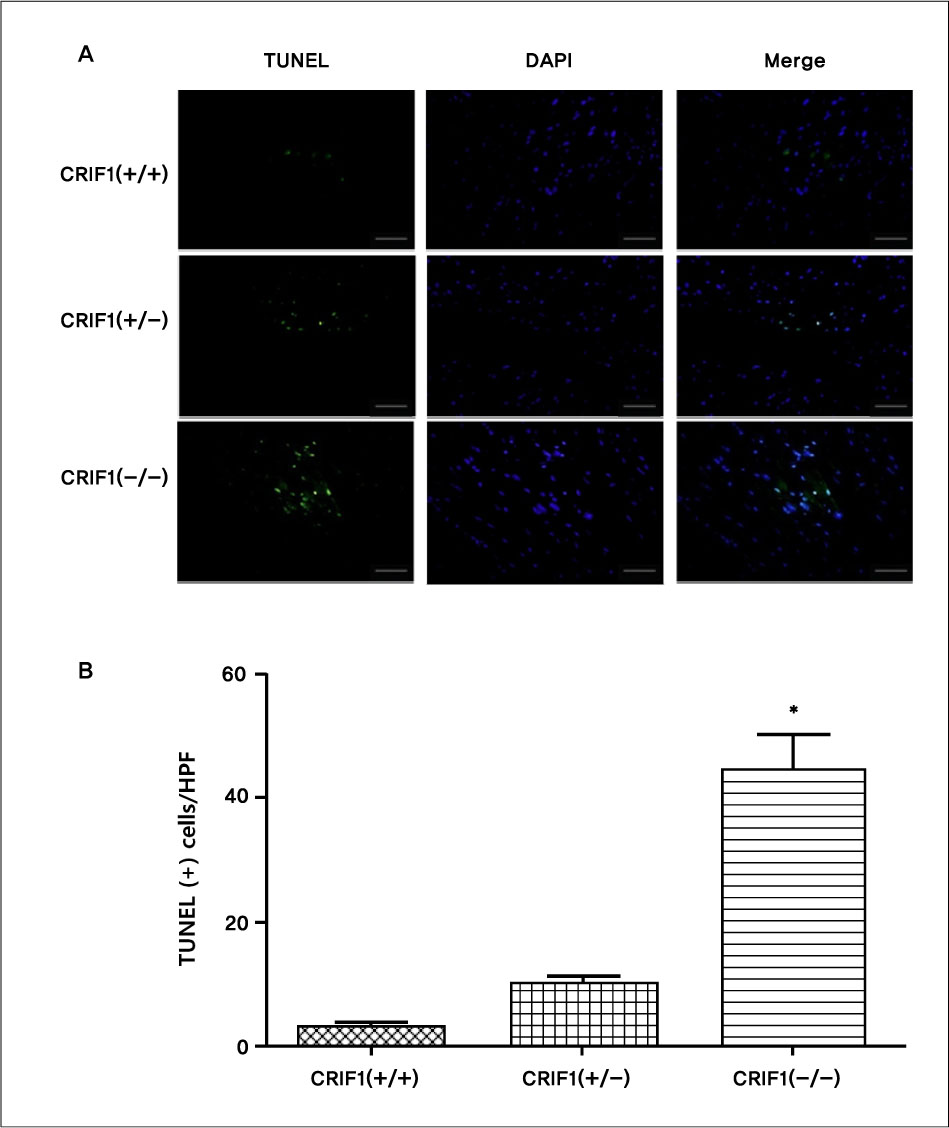
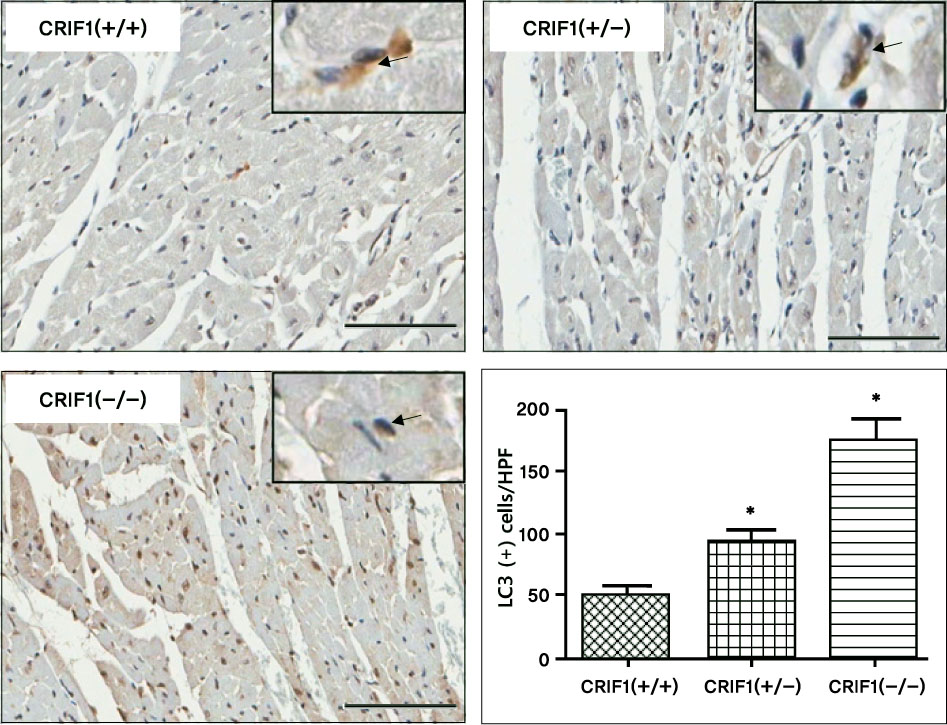
Serial echocardiography showed decreased left ventricular ejection fraction and fractional shortening in the CRIF1-deficient mice at four and seven weeks later compared to wild type mice (p < 0.05). H&E showed increased myocardial inflammation in the CRIF1-deficient mice. Terminal deoxynucleotidyl transferase biotin-dUTP nick end labeling staining and LC3 staining showed increased apoptosis and autophage in CRIF1-deficient mice compared with wild type (p < 0.01). Electron microscopy revealed that the mitochondria in CRIF1-deficient cardiomyocytes showed abnormal morphogenesis. For example, the cells showed excessively fragmented mitochondria, intracristal swelling, and thinning of myocardial fiber. The stability of mitochondrial complexes in CRIF1-deficient cells showed marked derangements.
CONCLUSIONS
CRIF1 is required for maintenance of normal mitochondrial function and modulate apoptosis and autophagy in the heart.
MeSH Terms
-
Aging
Animals
Apoptosis
Autophagy
Cardiovascular Diseases
Cell Cycle Proteins
DNA Nucleotidylexotransferase
Echocardiography
Genomic Instability
Heart
Heart Failure
Homeostasis
Inflammation
Mice
Microscopy, Electron
Mitochondria
Mitochondria, Heart
Morphogenesis
Myocytes, Cardiac
Stroke Volume
Thoracotomy
Viruses
Cell Cycle Proteins
DNA Nucleotidylexotransferase
Figure
Reference
-
1. Madamanchi NR, Runge MS. Mitochondrial dysfunction in atherosclerosis. Circ Res. 2007. 100:460–473.
Article2. Chung HK, Yi YW, Jung NC, Kim D, Suh JM, Kim H, et al. CR6-interacting factor 1 interacts with Gadd45 family proteins and modulates the cell cycle. J Biol Chem. 2003. 278:28079–28088.
Article3. Kwon MC, Koo BK, Moon JS, Kim YY, Park KC, Kim NS, et al. Crif1 is a novel transcriptional coactivator of STAT3. EMBO J. 2008. 27:642–653.
Article4. Park KC, Song KH, Chung HK, Kim H, Kim DW, Song JH, et al. CR6-interacting factor 1 interacts with orphan nuclear receptor Nur77 and inhibits its transactivation. Mol Endocrinol. 2005. 19:12–24.
Article5. Lee TY. Reconsideration of hypertension awareness, treatment, and control. J Korean Soc Hypertens. 2009. 15:1–6.6. Rosenberg P. Mitochondrial dysfunction and heart disease. Mitochondrion. 2004. 4:621–628.
Article7. Cha MJ, Lee HY, Ahn SV, Han KR, Park JB, Lim SJ, et al. Prevalence and clinical characteristics of metabolic syndrome in Korean hypertensive patients. J Korean Soc Hypertens. 2009. 15:37–44.8. Ballinger SW. Mitochondrial dysfunction in cardiovascular disease. Free Radic Biol Med. 2005. 38:1278–1295.
Article9. Coskun P, Wyrembak J, Schriner SE, Chen HW, Marciniack C, Laferla F, et al. A mitochondrial etiology of Alzheimer and Parkinson disease. Biochim Biophys Acta. 2012. 1820:553–564.
Article10. Mitchell T, Darley-Usmar V. Metabolic syndrome and mitochondrial dysfunction: insights from preclinical studies with a mitochondrially targeted antioxidant. Free Radic Biol Med. 2012. 52:838–840.
Article11. Wallace DC, Lott MT, Shoffner JM, Brown MD. Diseases resulting from mitochondrial DNA point mutations. J Inherit Metab Dis. 1992. 15:472–479.
Article12. Tompkins AJ, Burwell LS, Digerness SB, Zaragoza C, Holman WL, Brookes PS. Mitochondrial dysfunction in cardiac ischemia-reperfusion injury: ROS from complex I, without inhibition. Biochim Biophys Acta. 2006. 1762:223–231.
Article13. Lesnefsky EJ, Moghaddas S, Tandler B, Kerner J, Hoppel CL. Mitochondrial dysfunction in cardiac disease: ischemia--reperfusion, aging, and heart failure. J Mol Cell Cardiol. 2001. 33:1065–1089.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Mitochondrial Dynamics in the Heart as a Novel Therapeutic Target for Cardioprotection
- Resveratrol attenuates aging-induced mitochondrial dysfunction and mitochondria-mediated apoptosis in the rat heart
- Modulation of the caveolin-3 localization to caveolae and STAT3 to mitochondria by catecholamine-induced cardiac hypertrophy in H9c2 cardiomyoblasts
- Positioning Metabolism as a Central Player in the Diabetic Heart
- Mitochondrial Quality Control in the Heart: New Drug Targets for Cardiovascular Disease