J Bacteriol Virol.
2016 Jun;46(2):84-92. 10.4167/jbv.2016.46.2.84.
Bacillus spp. or Bacillus spp.-Derived Membrane Vesicles Induce the Intrinsic Pathways of Apoptosis of Human Colon Cancer Cell Lines
- Affiliations
-
- 1Department of Microbiology, of Medical Science, Cancer Research Institute, School of Medicine, Chungnam National University, Daejeon, Korea. jekpark@cnu.ac.kr
- KMID: 2327903
- DOI: http://doi.org/10.4167/jbv.2016.46.2.84
Abstract
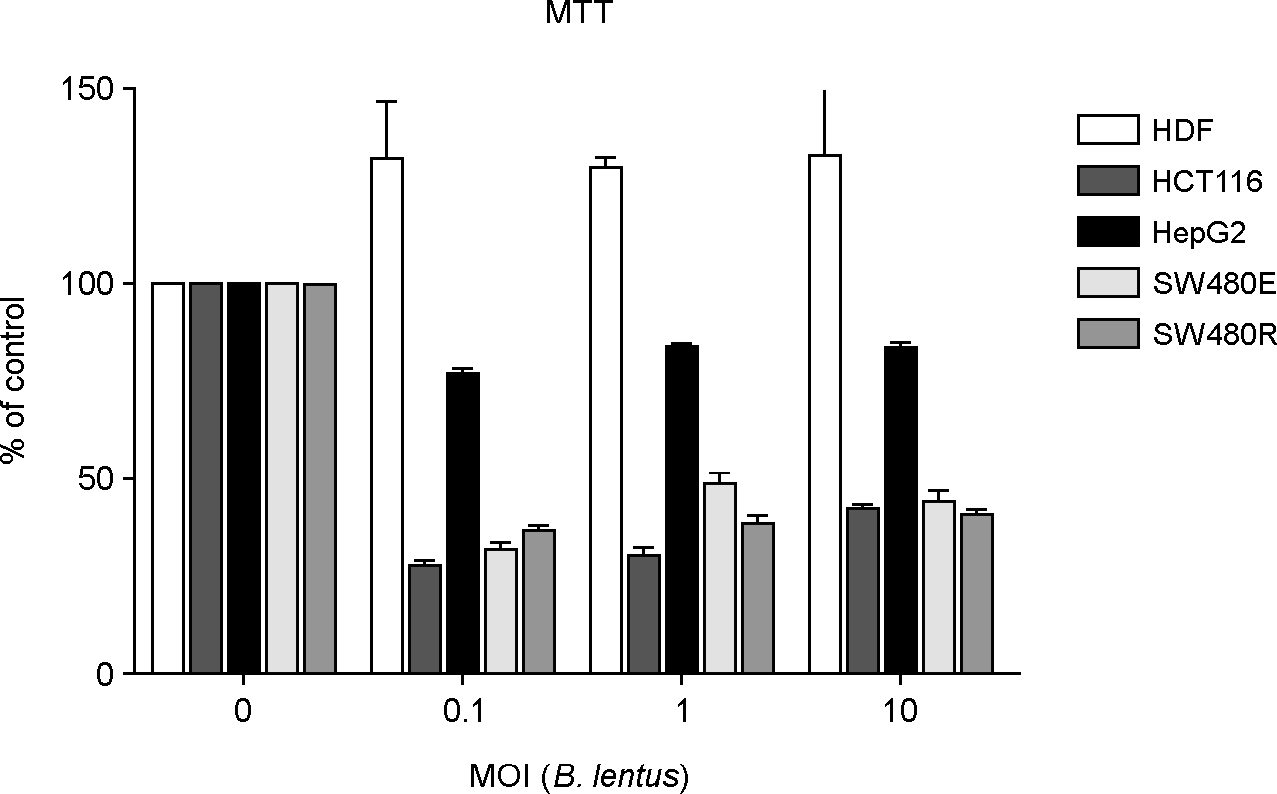
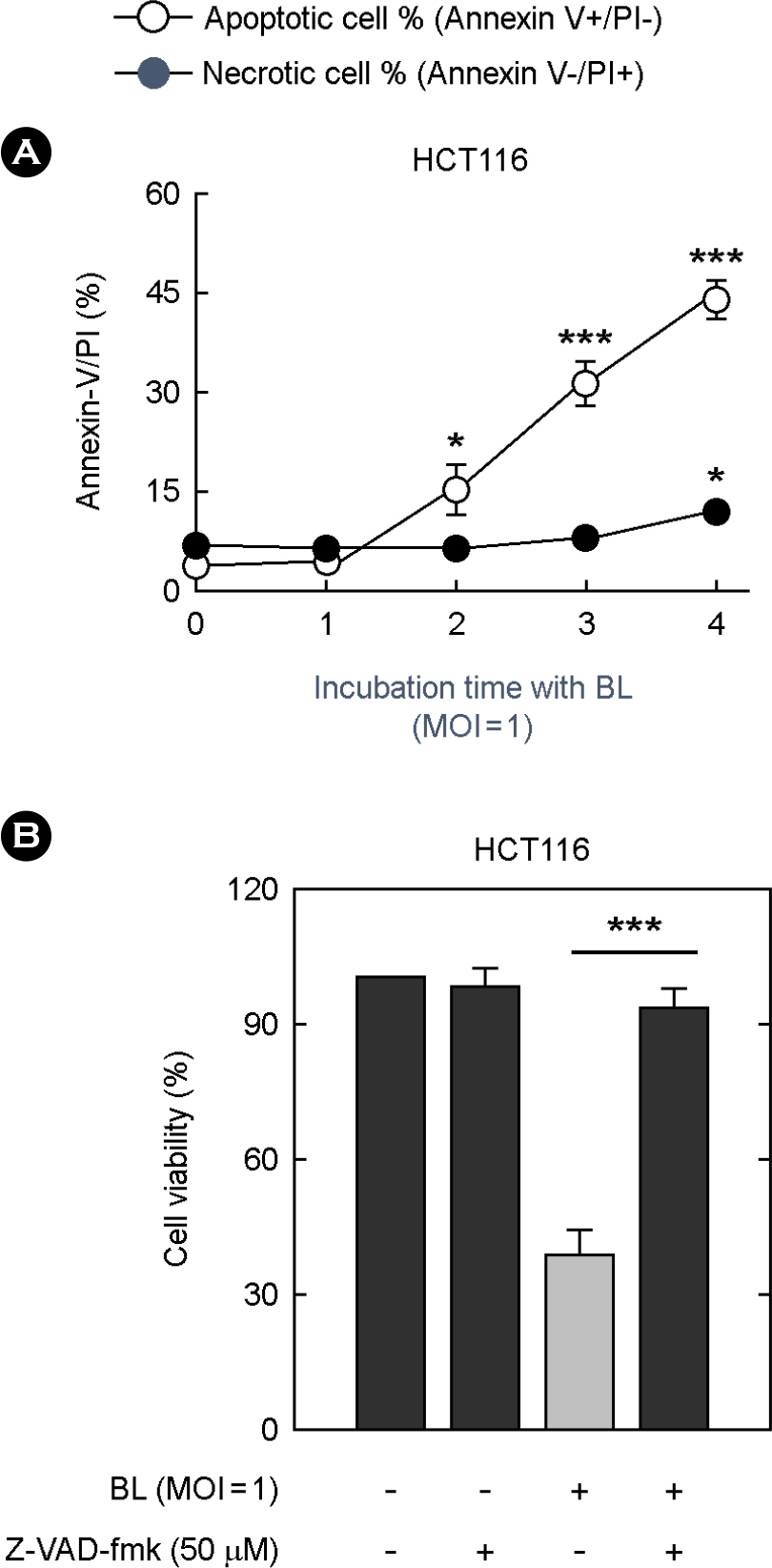
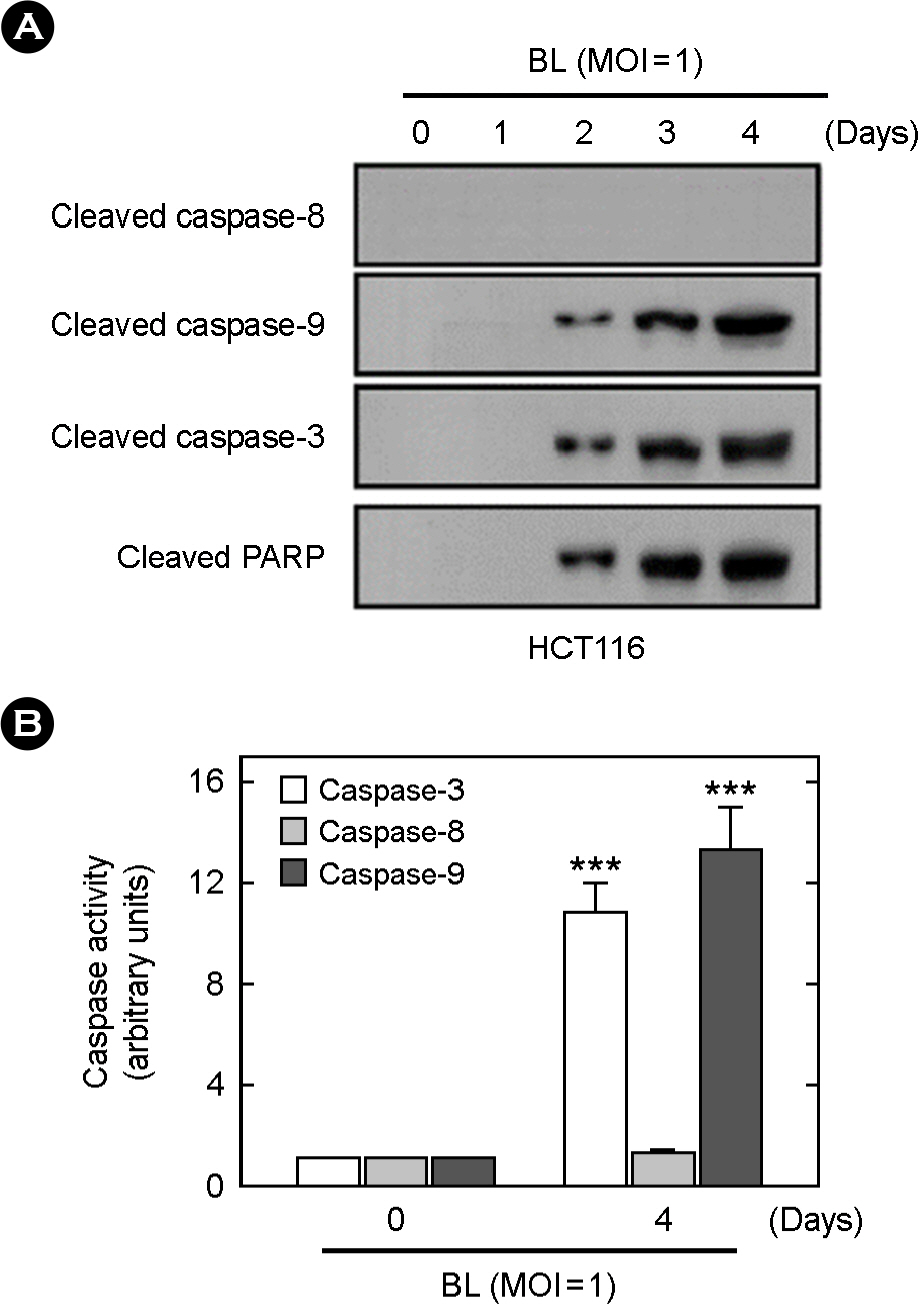
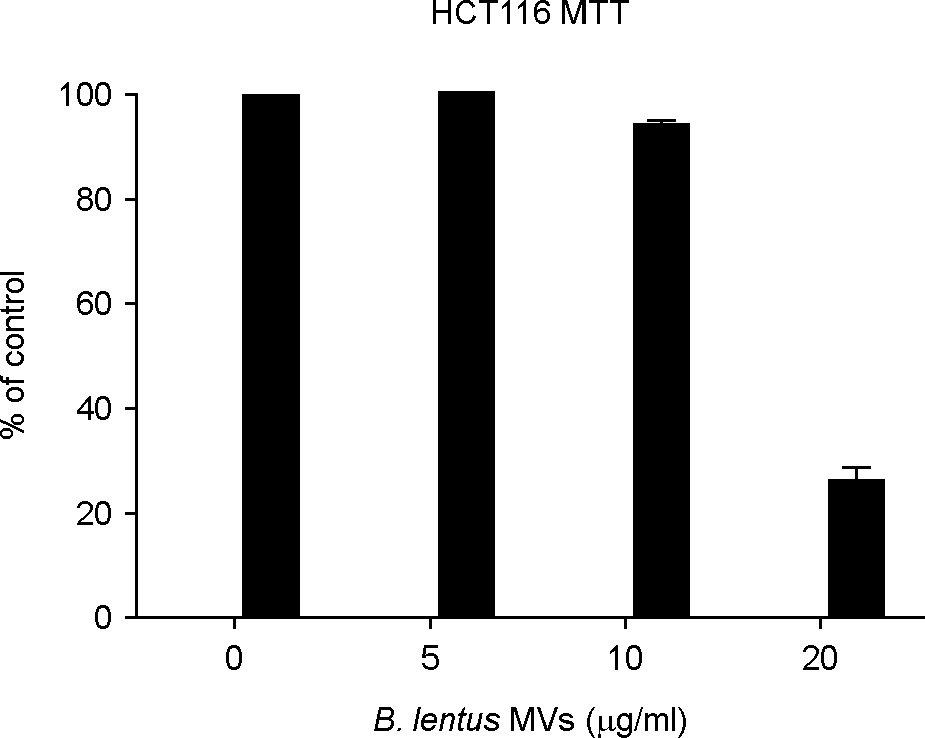
- The striking increase in colorectal cancer (CRC) has shown the great fatality in Korea for more than 15 years. The leading edge of this rising incidence rate is mainly due to the people's dietary changes in Korea. Some studies have reported that the dietary fiber does not have significant cytotoxic effects on CRC cells, which contrasts to the effects of probiotics. It gives a positive evaluation that the nonpathogenic spore-forming Bacillus species among the probiotics including fermented bacteria might have optimistic effects on CRC incidence rate. Recently, we isolated Bacillus lentus (BL) from Korean soybean fermented food. BL showed the cytotoxic effect on human colon carcinoma cell lines HCT116 and SW480. Interestingly, BL did not have effect on human dermal fibroblast cells and human hepatoma cell line HepG2. It suggested that BL has the target cell-specific cytotoxicity toward human colon carcinoma cells. To clarify the death signaling pathway underlying the BL-induced apoptosis in cancer cells, we analyzed the expression of caspases, Bax and Bcl-2 by western blotting. The apoptotic effects by cytotoxic elements were executed by direct BL contact or membrane-derived vesicles isolated from BL. Treatment of HCT116 with BL activated caspase-9, -3 and increased cleavage form of poly (ADP-ribose) polymerase (PARP). However, caspase-8 activity was not increased by BL. BL-activated intrinsic pathway increased the pro-apoptotic Bax, decreased the anti-apoptotic Bcl-2 proteins on mitochondria, disrupted the mitochondrial membrane potential, and then released the cytochrome c from mitochondria. The membrane-derived vesicles (MVs) from BL induced apoptosis of the HCT116. Here, we propose that BL as a strong candidate for the development of apoptosis-specific anti-tumor agent will give great contribution to the understandings of the tumor-microbe interdisciplinary areas.
MeSH Terms
-
Apoptosis*
Bacillus*
Bacteria
Blotting, Western
Carcinoma, Hepatocellular
Caspase 8
Caspase 9
Caspases
Cell Line*
Colon*
Colonic Neoplasms*
Colorectal Neoplasms
Cytochromes c
Dietary Fiber
Fibroblasts
Humans*
Incidence
Korea
Membrane Potential, Mitochondrial
Membranes*
Mitochondria
Probiotics
Soybeans
Strikes, Employee
Caspase 8
Caspase 9
Caspases
Cytochromes c
Figure
Reference
-
1). Jung KW, Won YJ, Oh CM, Kong HJ, Cho H, Lee JK, et al. Prediction of Cancer Incidence and Mortality in Korea, 2016. Cancer Res Treat. 2016; 48:451–7.
Article2). Raskov H, Pommergaard HC, Burcharth J, Rosenberg J. Colorectal carcinogenesis–update and perspectives. World J Gastroenterol. 2014; 20:18151–64.
Article3). zur Hausen H. Red meat consumption and cancer: reasons to suspect involvement of bovine infectious factors in colorectal cancer. Int J Cancer. 2012; 130:2475–83.
Article4). Zhu Y, Michelle Luo T, Jobin C, Young HA. Gut microbiota and probiotics in colon tumorigenesis. Cancer Lett. 2011; 309:119–27.
Article5). O'Keefe SJ, Ou J, Aufreiter S, O'Connor D, Sharma S, Sepulveda J, et al. Products of the colonic microbiota mediate the effects of diet on colon cancer risk. J Nutr. 2009; 139:2044–8.6). Akhter M, Inoue M, Kurahashi N, Iwasaki M, Sasazuki S, Tsugane S. Japan Public Health Center-Based prospective Study Group. Dietary soy and isoflavone intake and risk of colorectal cancer in the Japan public health center-based prospective study. Cancer Epidemiol Biomarkers Prev. 2008; 17:2128–35.7). Kim MK, Park JH. Conference on "Multidisciplinary approaches to nutritional problems". Symposium on "Nutrition and health". Cruciferous vegetable intake and the risk of human cancer: epidemiological evidence. Proc Nutr Soc. 2009; 68:103–10.8). Fotiadis CI, Stoidis CN, Spyropoulos BG, Zografos ED. Role of probiotics, prebiotics and synbiotics in chemoprevention for colorectal cancer. World J Gastroenterol. 2008; 14:6435–57.
Article9). Lim YC, Cho KW, Kwon HC, Kang SU, Pyun JH, Lee MH, et al. Growth Inhibition and Apoptosis with H31 Metabolites from Marine Bacillus SW31 in Head and Neck Cancer Cells. Clin Exp Otorhinolaryngol. 2010; 3:217–25.10). Jeong SY, Park SY, Kim YH, Kim M, Lee SJ. Cytotoxicity and apoptosis induction of Bacillus vallismortis BIT-33 metabolites on colon cancer carcinoma cells. J Appl Microbiol. 2008; 104:796–807.11). Ma EL, Choi YJ, Choi J, Pothoulakis C, Rhee SH, Im E. The anticancer effect of probiotic Bacillus polyfermenticus on human colon cancer cells is mediated through ErbB2 and ErbB3 inhibition. Int J Cancer. 2010; 127:780–90.12). Kim J, Park S, Nam BH. The risk of colorectal cancer is associated with the frequency of meat consumption in a population-based cohort in Korea. Asian Pac J Cancer Prev. 2011; 12:2371–6.13). Choi JK, Kim DW, Shin SY, Park EC, Kang JG. Effect of Ulcerative Colitis on Incidence of Colorectal Cancer: Results from the Nationwide Population-Based Cohort Study (2003-2013). J Cancer. 2016; 7:681–6.
Article14). Jackson CS, Oman M, Patel AM, Vega KJ. Health disparities in colorectal cancer among racial and ethnic minorities in the United States. J Gastrointest Oncol. 2016; 7:S32–43.15). Gagnière J, Raisch J, Veziant J, Barnich N, Bonnet R, Buc E, et al. Gut microbiota imbalance and colorectal cancer. World J Gastroenterol. 2016; 22:501–18.
Article16). Hammond WA, Swaika A, Mody K. Pharmacologic resistance in colorectal cancer: a review. Ther Adv Med Oncol. 2016; 8:57–84.
Article17). Rabeneck L, Horton S, Zauber AG, Earle C. Colorectal Cancer. Gelband H, Jha P, Sankaranarayanan R, Horton S, editors. Cancer: Disease Control Priorities. 3rd ed.Washington D.C.: The International Bank for Reconstruction and Development/The World Bank;2015. p. 101–19.
Article18). Oostindjer M, Alexander J, Amdam GV, Andersen G, Bryan NS, Chen D, et al. The role of red and processed meat in colorectal cancer development: a perspective. Meat Sci. 2014; 97:583–96.
Article19). Fotiadis CI, Stoidis CN, Spyropoulos BG, Zografos ED. Role of probiotics, prebiotics and synbiotics in chemoprevention for colorectal cancer. World J Gastroenterol. 2008; 14:6453–7.
Article20). Jung JY, Lee SH, Kim JM, Park MS, Bae JW, Hahn Y, et al. Metagenomic analysis of kimchi, a traditional Korean fermented food. Appl Environ Microbiol. 2011; 77:2264–74.
Article21). Song YR, Song NE, Kim JH, Nho YC, Baik SH. Exopolysaccharide produced by Bacillus licheniformis strains isolated from Kimchi. J Gen Appl Microbiol. 2011; 57:169–75.22). Kim TW, Lee JH, Kim SE, Park MH, Chang HC, Kim HY. Analysis of microbial communities in doenjang, a Korean fermented soybean paste, using nested PCR-denaturing gradient gel electrophoresis Int J Food Microbiol. 2009; 131:265–71.23). Kwon GH, Lee HA, Park JY, Kim JS, Lim J, Park CS, et al. Development of a RAPD-PCR method for identification of Bacillus species isolated from Cheonggukjang. Int J Food Microbiol. 2009; 129:282–7.24). Lee CH. Engineering bacteria toward tumor targeting for cancer treatment: current state and perspectives. Appl Microbiol Biotechnol. 2012; 93:517–23.
Article25). Moushumi Priya A, Jayachandran S. Induction of apoptosis and cell cycle arrest by Bis (2-ethylhexyl) phthalate produced by marine Bacillus pumilus MB 40. Chem Biol Interact. 2012; 195:133–43.26). Lee EY, Choi DY, Kim DK, Kim JW, Park JO, Kim S, et al. Gram-positive bacteria produce membrane vesicles: proteomics-based characterization of Staphylococcus aureus-derived membrane vesicles. Proteomics. 2009; 9:5425–36.27). Lim YJ, Jo YH, Kim HJ, Park JK. The synergistic effects of antimicrobial peptides on the growth inhibition of Salmonella Typhimurium through Imd pathway in Drosophila intestine. J Bacteriol Virol. 2013; 43:120–30.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Distributions of Listeria spp., Bacillus spp., Enterococcus spp., Staphylococcus spp., and Coliforms Isolated from Agricultural Herb Products from the Market
- Distributions of Bacillus cereus, Pseudomonas, Enterococcus, and coliforms Isolated from Agricultural Products
- Alterations of the Apoptosis Genes and Their Products in Non-small Cell Lung Cancer Tissues
- 16S rRNA gene-based metagenomic analysis reveals differences in bacteria-derived extracellular vesicles in the urine of pregnant and non-pregnant women
- The Effect of Transfection of RET9 and RET51 on Apoptosis in SW48 and RKO Colon Cancer Cells