Tuberc Respir Dis.
2014 Aug;77(2):65-72. 10.4046/trd.2014.77.2.65.
Effects of Morus alba L. and Natural Products Including Morusin on In Vivo Secretion and In Vitro Production of Airway MUC5AC Mucin
- Affiliations
-
- 1Department of Pharmacology, Chungnam National University School of Medicine, Daejeon, Korea. LCJ123@cnu.ac.kr
- 2Department of Pharmacy, College of Pharmacy, Chosun University, Gwangju, Korea.
- 3Department of Pharmacy, College of Pharmacy and Research Institute of Pharmaceutical Sciences, Seoul National University, Seoul, Korea.
- 4Pulmonology Section, Department of Internal Medicine, Chungnam National University Hospital, Chungnam National University School of Medicine, Daejeon, Korea.
- KMID: 2320541
- DOI: http://doi.org/10.4046/trd.2014.77.2.65
Abstract
- BACKGROUND
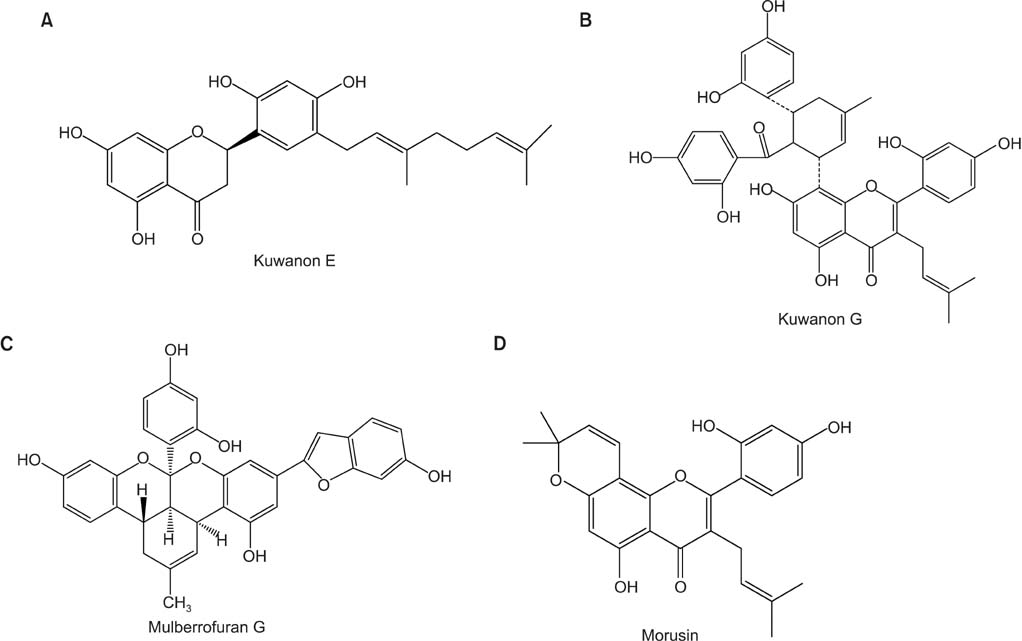
It is valuable to find the potential activity of regulating the excessive mucin secretion by the compounds derived from various medicinal plants. We investigated whether aqueous extract of the root bark of Morus alba L. (AMA), kuwanon E, kuwanon G, mulberrofuran G, and morusin significantly affect the secretion and production of airway mucin using in vivo and in vitro experimental models.
METHODS
Effect of AMA was examined on hypersecretion of airway mucin in sulfur dioxide-induced acute bronchitis in rats. Confluent NCI-H292 cells were pretreated with ethanolic extract, kuwanon E, kuwanon G, mulberrofuran G, or morusin for 30 minutes and then stimulated with phorbol 12-myristate 13-acetate (PMA) for 24 hours. The MUC5AC mucin secretion and production were measured by enzyme-linked immunosorbent assay.
RESULTS
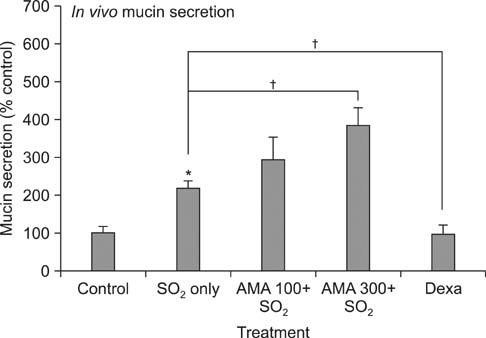
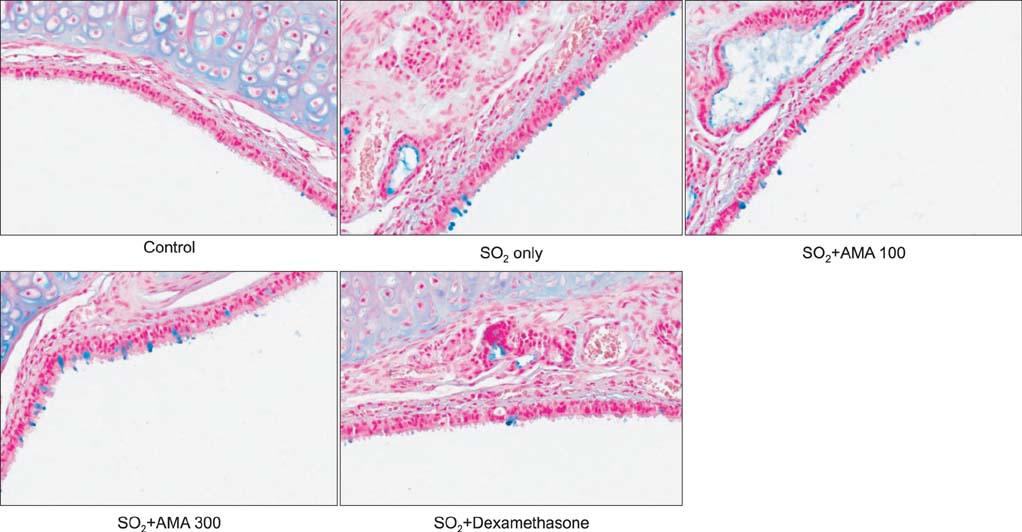
AMA stimulated the secretion of airway mucin in sulfur dioxide-induced bronchitis rat model; aqueous extract, ethanolic extract, kuwanon E, kuwanon G, mulberrofuran G and morusin inhibited the production of MUC5AC mucin induced by PMA from NCI-H292 cells, respectively.
CONCLUSION
These results suggest that extract of the root bark and the natural products derived from Morus alba L. can regulate the secretion and production of airway mucin and, at least in part, explains the folk use of extract of Morus alba L. as mucoregulators in diverse inflammatory pulmonary diseases.
Keyword
MeSH Terms
Figure
Reference
-
1. Lee CJ, Paik SH, Ko KH, Kim KC. Effects of polycationic peptides on mucin release from airway goblet cells: relationship between polymer size and activity. Inflamm Res. 2002; 51:490–494.2. Voynow JA, Rubin BK. Mucins, mucus, and sputum. Chest. 2009; 135:505–512.3. Heo HJ, Lee HJ, Kim YS, Kang SS, Son KH, Seok JH, et al. Effects of baicalin and wogonin on mucin release from cultured airway epithelial cells. Phytother Res. 2007; 21:1130–1134.4. Heo HJ, Lee SY, Lee MN, Lee HJ, Seok JH, Lee CJ. Genistein and curcumin suppress epidermal growth factor-induced MUC5AC mucin production and gene expression from human airway epithelial cells. Phytother Res. 2009; 23:1458–1461.5. Lee HJ, Lee SY, Lee MN, Kim JH, Chang GT, Seok JH, et al. Inhibition of secretion, production and gene expression of mucin from cultured airway epithelial cells by prunetin. Phytother Res. 2011; 25:1196–1200.6. Jang IM. Treatise on Asian herbal medicines. Seoul: Haksul-pyunsu-kwan in Research Institute of Natural Products of Seoul National University;2003.7. Chi YS, Jong HG, Son KH, Chang HW, Kang SS, Kim HP. Effects of naturally occurring prenylated flavonoids on enzymes metabolizing arachidonic acid: cyclooxygenases and lipoxygenases. Biochem Pharmacol. 2001; 62:1185–1191.8. Geng CA, Ma YB, Zhang XM, Yao SY, Xue DQ, Zhang RP, et al. Mulberrofuran G and isomulberrofuran G from Morus alba L.: anti-hepatitis B virus activity and mass spectrometric fragmentation. J Agric Food Chem. 2012; 60:8197–8202.9. Lee JC, Won SJ, Chao CL, Wu FL, Liu HS, Ling P, et al. Morusin induces apoptosis and suppresses NF-kappaB activity in human colorectal cancer HT-29 cells. Biochem Biophys Res Commun. 2008; 372:236–242.10. Mihara S, Hara M, Nakamura M, Sakurawi K, Tokura K, Fujimoto M, et al. Non-peptide bombesin receptor antagonists, kuwanon G and H, isolated from mulberry. Biochem Biophys Res Commun. 1995; 213:594–599.11. Pon DJ, van Staden CJ, Boulet L, Rodger IW. Hyperplastic effects of aerosolized sodium metabisulfite on rat airway mucus-secretory epithelial cells. Can J Physiol Pharmacol. 1994; 72:1025–1030.12. Li JD, Dohrman AF, Gallup M, Miyata S, Gum JR, Kim YS, et al. Transcriptional activation of mucin by Pseudomonas aeruginosa lipopolysaccharide in the pathogenesis of cystic fibrosis lung disease. Proc Natl Acad Sci U S A. 1997; 94:967–972.13. Shao MX, Ueki IF, Nadel JA. Tumor necrosis factor alpha-converting enzyme mediates MUC5AC mucin expression in cultured human airway epithelial cells. Proc Natl Acad Sci U S A. 2003; 100:11618–11623.14. Takeyama K, Dabbagh K, Lee HM, Agusti C, Lausier JA, Ueki IF, et al. Epidermal growth factor system regulates mucin production in airways. Proc Natl Acad Sci U S A. 1999; 96:3081–3086.15. Hano Y, Fukai T, Nomura T, Uzawa J, Fukushima K. Structure of mulberrofuran I, a novel 2-arylbenzofuran derivatives from the cultivated mulberry tree (Morus bombycis Koidz). Chem Pharm Bull. 1984; 32:1260–1263.16. Hano Y, Hirakura K, Nomura T, Terada S, Fukushima K. Components of root bark of morus lhou1 1. Structures of two new natural diels-alder adducts, kuwanons N and o. Planta Med. 1984; 50:127–130.17. Nomura T, Fukai T, Narita T. Hypotensive constituent, kuwanon H, a new flavone derivative from the root bark of the cultivated mulberry tree (Morus alba L.). Heterocycles. 1980; 14:1943–1951.18. Nomura T, Fukai T. Constituents of the cultivated mulberry tree. Planta Med. 1981; 42:79–88.19. Burgel PR, Montani D, Danel C, Dusser DJ, Nadel JA. A morphometric study of mucins and small airway plugging in cystic fibrosis. Thorax. 2007; 62:153–161.20. Rogers DF, Barnes PJ. Treatment of airway mucus hypersecretion. Ann Med. 2006; 38:116–125.21. Hong DH, Petrovics G, Anderson WB, Forstner J, Forstner G. Induction of mucin gene expression in human colonic cell lines by PMA is dependent on PKC-epsilon. Am J Physiol. 1999; 277:G1041–G1047.22. Hewson CA, Edbrooke MR, Johnston SL. PMA induces the MUC5AC respiratory mucin in human bronchial epithelial cells, via PKC, EGF/TGF-alpha, Ras/Raf, MEK, ERK and Sp1-dependent mechanisms. J Mol Biol. 2004; 344:683–695.23. Park SJ, Kang SY, Kim NS, Kim HM. Phosphatidylinositol 3-kinase regulates PMA-induced differentiation and superoxide production in HL-60 cells. Immunopharmacol Immunotoxicol. 2002; 24:211–226.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Homogentisic Acid and Natural Products Derived from Pinellia ternata on Secretion, Production and Gene Expression of MUC5AC Mucin from Cultured Airway Epithelial Cells
- Effects of Cynaroside, Cynarin and Linarin on Secretion, Production and Gene Expression of Airway MUC5AC Mucin in NCI-H292 Cells
- Growth Factor- and Phorbol Ester-induced Production and Gene Expression of MUC5AC Mucin in Human Airway Epithelial NCI-H292 Cells Were Inhibited by Afzelin and Natural Products Derived from Houttuynia Cordata
- Effects of Lobetyolin, Lobetyol and Methyl linoleate on Secretion, Production and Gene Expression of MUC5AC Mucin from Airway Epithelial Cells
- Effects of Caffeic Acid, Myristicin and Rosemarinic Acid on the Gene Expression and Production of Airway MUC5AC Mucin