Tuberc Respir Dis.
2006 May;60(5):554-563.
The Effects of Air-borne Particulate Matters on the Alveolar Macrophages for the TNF-alpha and IL-1beta Secretion
- Affiliations
-
- 1Konkuk University College of Veterinary Medicine, Seoul, Korea. nojamaji@hanmail.net
- 2Kyungpook National University College of Veterinary Medicine, Korea.
- 3National Veterinary Research and Quarantine Service, Korea.
Abstract
-
BACKGROUND: PM is known to induce various pulmonary diseases, including asthma, cancer, fibrosis and chronic bronchitis. Despite the epidemiological evidence the pathogenesis of PM-related pulmonary diseases is unclear.
METHODS
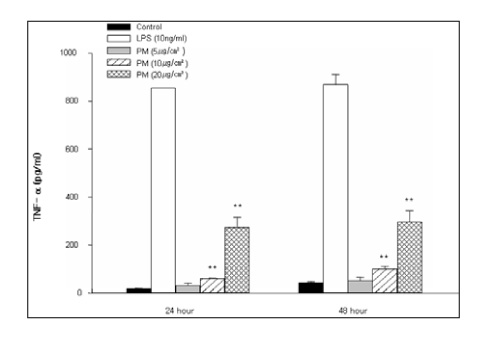
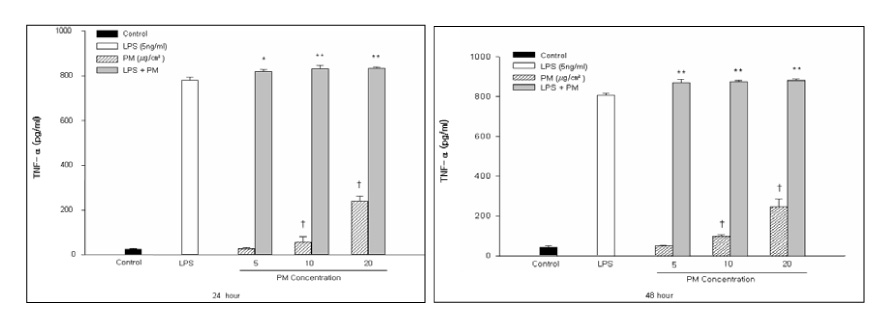
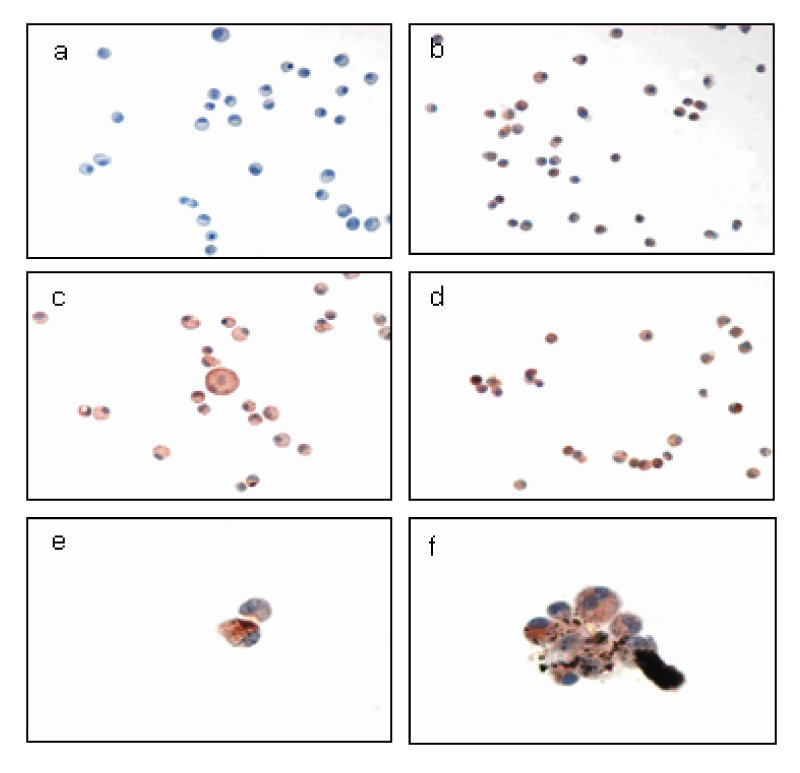
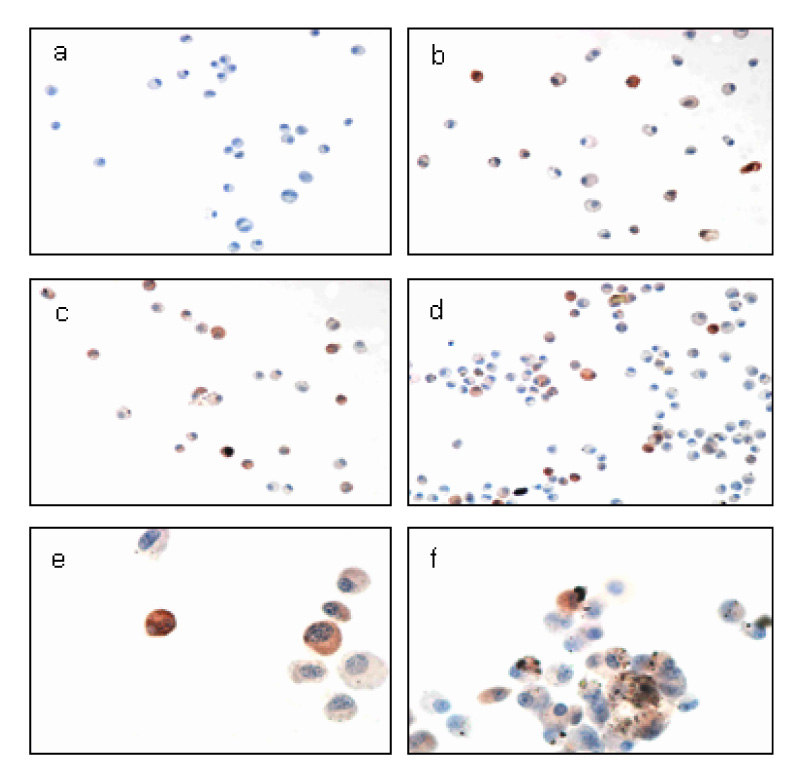
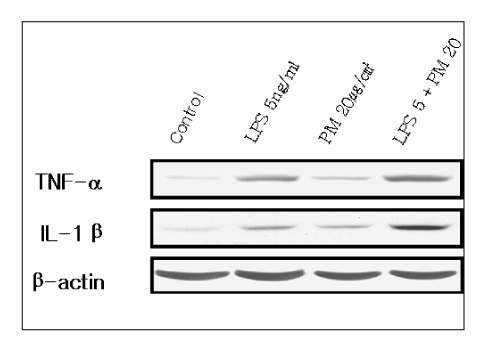
This study examined the effects of PM exposure on the secretion of TNF-alpha and IL-1beta in the cultured alveolar macrophages. The cultured primary alveolar macrophages were treated with the medium, PM (5~20 microgram/cm2), LPS (5ng/ml), and PM with LPS for 24h and 48h respectively. ELISA was used to assay the secreted TNF-alpha and IL-beta in the culture medium. Western blotting was used to identify and determine the level of proteins isolated from the culture cells. The cells cultured in the Lab-Tek(R) chamber slides were stained with immunocytochemical stains.
RESULTS
PM induced TNF-alpha and IL-1beta secretion in the culturing alveolar macrophages, collected from the SPF and inflammatory rats. However, the effects were only dose-dependent in the inflammatory macrophages. When the cells were co-treated with PM and LPS, there was a significant synergistic effect compared with the LPS in the both cell types.
CONCLUSION
PM might be play an important role in the induction and/or potentiation of various lung diseases by oversecretion of TNF-alpha and IL-1beta.
Keyword
MeSH Terms
Figure
Reference
-
1. Harre ES, Price PD, Ayrey RB, Toop LJ, Martin IR, Town GI. Respiratory effects of air populution in chronic obstructive pulmonary disease: a three-month prospective study. Thorax. 1997. 52:1040–1044.2. Lebowitz MD. Epidemiological studies of the respiratory effects of air pollution. Eur Respir J. 1996. 9:1029–1054.3. Pope CA 3rd, Dockery DW, Spengler JD, Raizenne ME. Respiratory health and PM10 pollution: a daily time series analysis. Am Rev Respir Dis. 1991. 144:668–674.4. Committee of the Environmental and Occupational Health Assembly of the American Thoracic Society. Health effects of outdoor air pollution: part 2. Am J Respir Crit Care Med. 1996. 153:477–498.5. Dockery DW, Pope CA 3rd. Acute respiratory effects of particulate air pollution. Annu Rev Public Health. 1994. 15:107–132.6. Pope CA 3rd, Kanner RE. Acute effects of PM10 pollution on pulmonary function of smokers with mild to moderate COPD. Am Rev Respir Dis. 1993. 147:1336–1340.7. Emanuel MB. Hay fever, a post industrial revolution epidemic: a history of its growth during the 19th century. Clin Allergy. 1988. 18:295–304.8. Abbey DE, Hwang BL, Burchette RJ, Vancuren T, Mills PK. Estimated long-term ambient concentrations of PM10 and development of respiratory symptoms in a nonsmoking population. Arch Environ Health. 1995. 50:139–152.9. Dusseldorp A, Kruize H, Brunekreef B, Hofschreuder P, de Meer G, van Oudvorst AB. Associations of PM10 and airbone iron with respiratory health of adults living near a steel factory. Am J Respir Crit Care Med. 1995. 152:1932–1939.10. Fels AO, Cohn ZA. The alveolar macrophage. J Appl Physiol. 1986. 60:353–369.11. Tracey KJ, Cerami A. Metabolic responses to cachectin/TNF. Ann N Y Acad Sci. 1990. 587:325–331.12. Beutler B, Grau GE. Tumor necrosis factor in the pathogenesis of infectious diseases. Crit Care Med. 1993. 21:S423–S435.13. Briscoe DM, Cotran RS, Pober JS. Effect of tumor necrosis factor, lipopolysaccharide, and IL-4 on the expression of vascular cell adhesion molecule-1 in vivo: correlation with CD3+T cell infiltrataion. J Immunol. 1992. 149:2954–2960.14. Gamber JR, Harlan JM, Klebanoff SJ, Vadas MA. Stimulation of the adherence of neutrophils to umbilical vein endothelium by human recombinant tumor necrosis factor. Proc Natl Acad Sci U S A. 1985. 82:8667–8671.15. Boumpas DT, Chrousos GP, Wilder RL, Cupps TR, Balow JE. Glucocorticoid therapy of immune-mediated diseases: basic and clinical correlates. Ann Intern Med. 1993. 119:1198–1208.16. Tracy KJ, Fong Y, Hesse DG, Manogue KR, lee AT, Kuo GC, et al. Anti-cachectin/TNF monoclonal antibodies prevent septic shock dmuring lethal bacteraemia. Nature. 1987. 330:662–664.17. Manel DN, Moore RN, Mergenhagen SE. Macrophages as a sourse of tumoricidal activity (tumor necrotizing factor). Infect Immun. 1980. 30:523–530.18. Kim KA, Lee DW, Lim Y, Yun IG. Effect of asbestos on fibroblast proliferation of rat. Korean J Occup Environ Med. 1996. 8:392–402.19. Lim Y, Kim KA, Kim HN, Lee DW, Cho WS, Yun IG. Cytotoxicity and apoptosis by silica, asbestos and man-made mineral fibers. Korean J Occup Environ Med. 1997. 9:641–649.20. Lim Y, Kim KA, Yun IG. The measurement of IL-1, 8, TNF for the diagnosis of pneumoconiosis. Korean J Occup Environ Med. 1997. 9:17–25.21. Lim Y, Kim SH, Cho YJ, Kim KA, Oh MW, Lee KH. Silica-inducced oxygen radical ge1neration in alveolar macrophage. Ind Health. 1997. 35:380–387.22. Vanhee D, Gosset P, Marquette CH, Wallaert B, Lafitte JJ, Gosselin B, et al. Secretion and mRNA expression of TNF alpha and IL-6 in the lungs of pneumocaniosis patients. Am J Respir Crit Med. 1995. 152:298–306.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Spontaneous and cStimulated Release of the TNF-alpha, IL-1beta, IL-6 and IL-8 of Alveolar Macrophages in the Patients with Pulmonary Tuberculosis
- Expression of TNF-alpha and IL-1beta in Splenic Dendritic Cells and Their Serum Levels in Mouse Sparganosis
- Immunomodulatory effect of beta-glucan derived from Saccharomyces cerevisiae strains
- Effect of bosentan on the production of proinflammatory cytokines in a rat model of emphysema
- Metformin Down-regulates TNF-alpha Secretion via Suppression of Scavenger Receptors in Macrophages