Nutr Res Pract.
2014 Oct;8(5):521-525. 10.4162/nrp.2014.8.5.521.
Artemisinin attenuates platelet-derived growth factor BB-induced migration of vascular smooth muscle cells
- Affiliations
-
- 1Department of Medical Science, School of Medicine Konkuk University, Hwayang-dong, Gwangjin-gu, Seoul 143-701, Korea. hhong@kku.ac.kr
- 2Department of Biomedical Laboratory Science, Kyungbok University, Gyeonggi-do 487-717, Korea.
- 3Department of Anatomy, College of Korean Medicine, Dongguk University, Gyeongsangbuk-do 780-714, Korea.
- KMID: 2313773
- DOI: http://doi.org/10.4162/nrp.2014.8.5.521
Abstract
- BACKGROUND/OBJECTIVES
Artemisinin (AT), an active compound in Arternisia annua, is well known as an anti-malaria drug. It is also known to have several effects including anti-oxidant, anti-inflammation, and anti-cancer activities. To date, the effect of AT on vascular disorders has not been studied. In this study, we investigated the effects of AT on the migration and proliferation of vascular smooth muscle cells (VSMC) stimulated by platelet-derived growth factor BB (PDGF-BB).
MATERIALS/METHODS
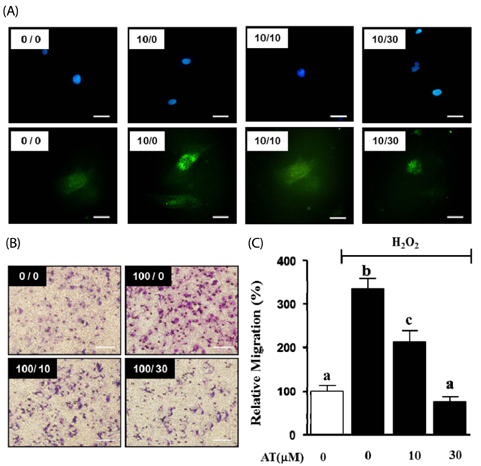
Aortic smooth muscle cells were isolated from Sprague-Dawley rats. PDGF-BB stimulated VSMC migration was measured by the scratch wound healing assay and the Boyden chamber assay. Cell viability was determined by using an EZ-Cytox Cell Viability Assay Kit. The production of reactive oxygen species (ROS) in PDGF-BB stimulated VSMC was measured through H2DCF-DA staining. We also determined the expression levels of signal proteins relevant to ROS, including measures of extracellular signal-regulated kinase (ERK) 1/2 measured by western blot analysis and matrix metalloproteinase (MMP) 9 measured by reverse transcription-polymerase chain reaction (RT-PCR).
RESULTS
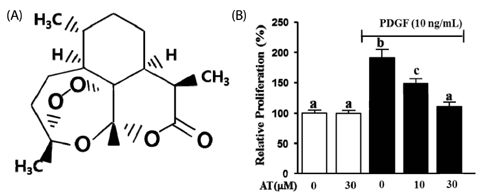
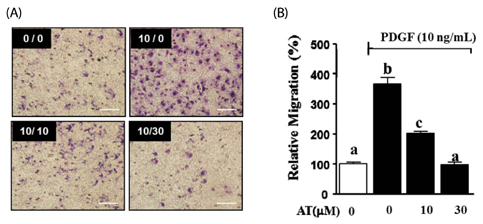
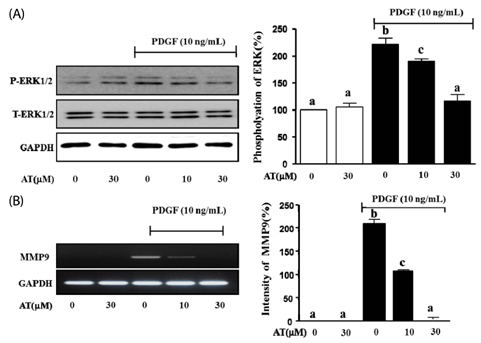
AT (10 microM and 30 microM) significantly reduced the proliferation and migration of PDGF-BB stimulated VSMC in a dose-dependent manner. The production of ROS, normally induced by PDGF-BB, is reduced by treatment with AT at both concentrations. PDGF-BB stimulated VSMC treated with AT (10 microM and 30 microM) have reduced phosphorylation of ERK1/2 and inhibited MMP9 expression compared to untreated PDGF-BB stimulated VSMC.
CONCLUSIONS
We suggest, based on these results, that AT may exert an anti-atherosclerotic effect on PDGF-BB stimulated VSMCs by inhibiting their proliferation and migration through down-regulation of ERK1/2 and MMP9 phosphorylation.
MeSH Terms
Figure
Reference
-
1. Ministry of Health and Welfare, Korea Centers for Disease Control and Prevention. Korea Health Statistics 2012: Korea National Health and Nutrition Examination Survey (KNHANES V-3). Cheongwon: Korea Centers for Disease Control and Prevention;2013.2. Buttari B, Profumo E, Businaro R, Saso L, Capoano R, Salvati B, Riganò R. Oxidized haemoglobin-driven endothelial dysfunction and immune cell activation: novel therapeutic targets for atherosclerosis. Curr Med Chem. 2013; 20:4806–4814.
Article3. Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature. 1993; 362:801–809.
Article4. Shah PK. Inflammation, neointimal hyperplasia, and restenosis: as the leukocytes roll, the arteries thicken. Circulation. 2003; 107:2175–2177.5. Kondo T, Konishi F, Inui H, Inagami T. Differing signal transductions elicited by three isoforms of platelet-derived growth factor in vascular smooth muscle cells. J Biol Chem. 1993; 268:4458–4464.
Article6. Ross R, Raines EW, Bowen-Pope DF. The biology of platelet-derived growth factor. Cell. 1986; 46:155–169.
Article7. Park SS, Kim WJ, Moon SK. Role of decursin in TNF-α-stimulated migration, invasion, and matrix metalloproteinase-9 induction in vascular smooth muscle cells. Food Sci Biotechnol. 2011; 20:913–918.
Article8. Moon CY, Ku CR, Cho YH, Lee EJ. Protocatechuic aldehyde inhibits migration and proliferation of vascular smooth muscle cells and intravascular thrombosis. Biochem Biophys Res Commun. 2012; 423:116–121.
Article9. Lyle AN, Griendling KK. Modulation of vascular smooth muscle signaling by reactive oxygen species. Physiology (Bethesda). 2006; 21:269–280.
Article10. Kyaw M, Yoshizumi M, Tsuchiya K, Kirima K, Tamaki T. Antioxidants inhibit JNK and p38 MAPK activation but not ERK 1/2 activation by angiotensin II in rat aortic smooth muscle cells. Hypertens Res. 2001; 24:251–261.
Article11. Zhou Y, Yan H, Guo M, Zhu J, Xiao Q, Zhang L. Reactive oxygen species in vascular formation and development. Oxid Med Cell Longev. 2013; 2013:374963.
Article12. Park J, Ha H, Seo J, Kim MS, Kim HJ, Huh KH, Park K, Kim YS. Mycophenolic acid inhibits platelet-derived growth factor-induced reactive oxygen species and mitogen-activated protein kinase activation in rat vascular smooth muscle cells. Am J Transplant. 2004; 4:1982–1990.
Article13. Bornfeldt KE, Raines EW, Nakano T, Graves LM, Krebs EG, Ross R. Insulin-like growth factor-I and platelet-derived growth factor-BB induce directed migration of human arterial smooth muscle cells via signaling pathways that are distinct from those of proliferation. J Clin Invest. 1994; 93:1266–1274.
Article14. Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res. 2003; 92:827–839.15. Kohler M, Haerdi W, Christen P, Veuthey JL. Extraction of artemisinin and artemisinic acid from Artemisia annua L. using supercritical carbon dioxide. J Chromatogr A. 1997; 785:353–360.
Article16. Ho WE, Peh HY, Chan TK, Wong WS. Artemisinins: pharmacological actions beyond anti-malarial. Pharmacol Ther. 2014; 142:126–139.
Article17. Won KJ, Lee SC, Lee CK, Lee HM, Lee SH, Fang Z, Choi OB, Jin M, Kim J, Park T, Choi WS, Kim SK, Kim B. Cordycepin attenuates neointimal formation by inhibiting reactive oxygen species-mediated responses in vascular smooth muscle cells in rats. J Pharmacol Sci. 2009; 109:403–412.
Article18. Singh NP, Lai HC. Artemisinin induces apoptosis in human cancer cells. Anticancer Res. 2004; 24:2277–2280.19. Choi OB, Park JH, Lee YJ, Lee CK, Won KJ, Kim J, Lee HM, Kim B. Olibanum extract inhibits vascular smooth muscle cell migration and proliferation in response to platelet-derived growth factor. Korean J Physiol Pharmacol. 2009; 13:107–113.
Article20. Jiang D, Li D, Wu W. Inhibitory effects and mechanisms of luteolin on proliferation and migration of vascular smooth muscle cells. Nutrients. 2013; 5:1648–1659.
Article21. Konneh MK, Rutherford C, Li SR, Anggård EE, Ferns GA. Vitamin E inhibits the intimal response to balloon catheter injury in the carotid artery of the cholesterol-fed rat. Atherosclerosis. 1995; 113:29–39.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Olibanum Extract Inhibits Vascular Smooth Muscle Cell Migration and Proliferation in Response to Platelet-Derived Growth Factor
- Heat shock protein 90 inhibitor AUY922 attenuates platelet-derived growth factor-BB-induced migration and proliferation of vascular smooth muscle cells
- Morphological Analysis of Intimal Hyperplasia in Allografted Aorta of Rat
- Effect of Carvedilol on the Growth of Vascular Smooth Muscle Cells
- Propofol Inhibits Platelet-derived Growth Factor-stimulated Migration in Rat Aortic Smooth Muscle Cells