Nutr Res Pract.
2009 Sep;3(3):192-199.
Cadmium increases ferroportin-1 gene expression in J774 macrophage cells via the production of reactive oxygen species
- Affiliations
-
- 1Department of Food and Nutrition and Research Institute of Science for Human Life, Kyung Hee University, 1 Hoegi-dong, Dongdaemun-gu, Seoul 130-701, Korea. jchung@khu.ac.kr
Abstract
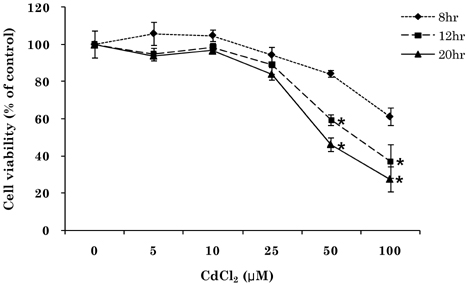
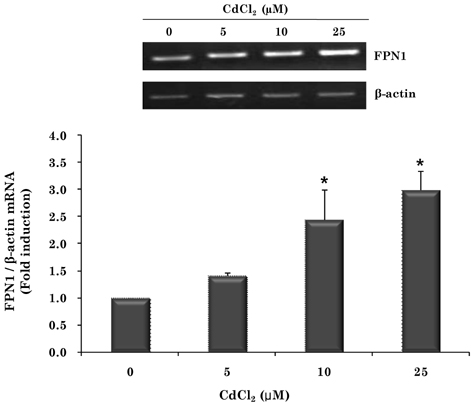
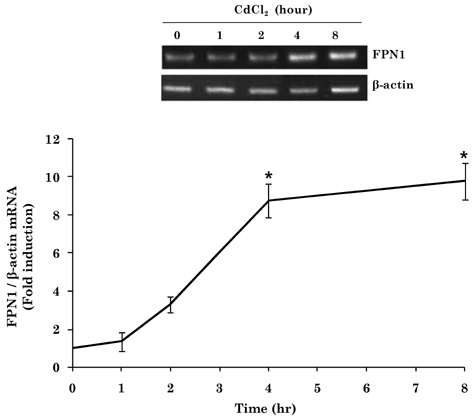
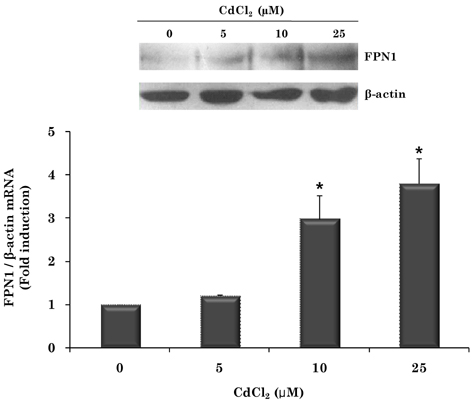
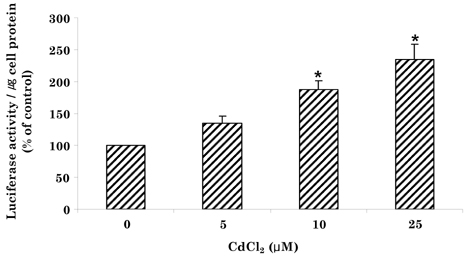
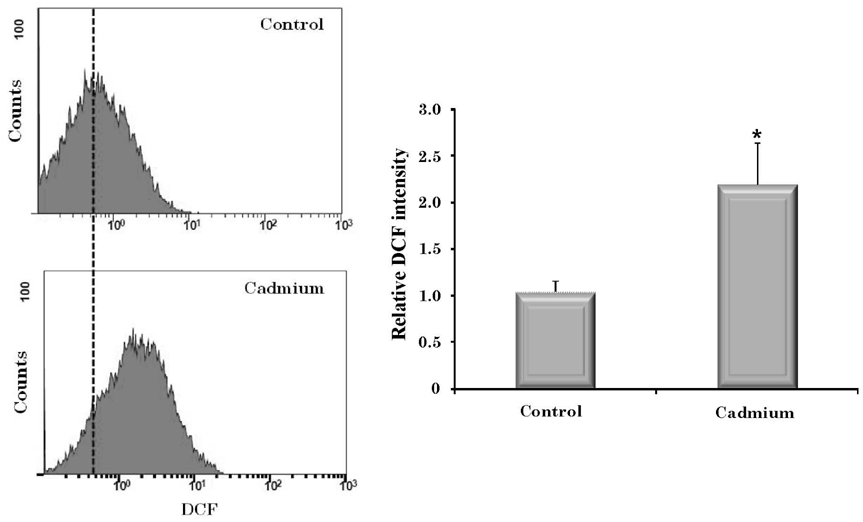
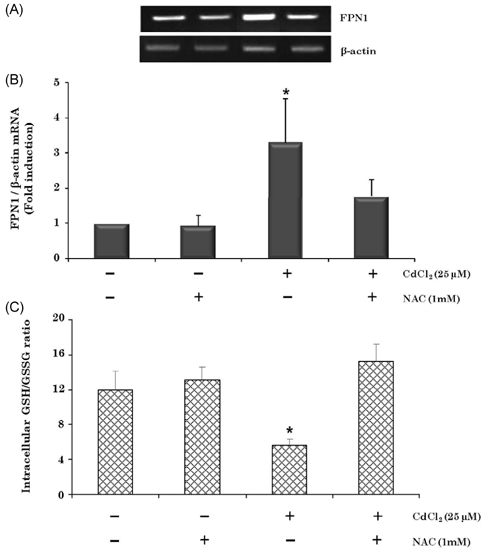
- Cadmium intoxication has been associated with the dysregulation of iron homeostasis. In the present study, we investigated the effect of cadmium on the expression of ferroportin 1 (FPN1), an important iron transporter protein that is involved in iron release from macrophages. When we incubated cadmium with J774 mouse macrophage cells, FPN1 mRNA levels were significantly increased in a dose- and time-dependent manner. Furthermore, the cadmium-induced FPN1 mRNA expression was associated with increased levels of FPN1 protein. On the other hand, cadmium-mediated FPN1 mRNA induction in J774 cells was completely blocked when cells were co-treated with a transcription inhibitor, acitomycin D. Also, cadmium directly stimulated the activity of the FPN1-promoter driven luciferase reporter, suggesting that the cadmium up-regulates FPN1 gene expression in a transcription-dependent manner. Finally, cadmium exposure to J774 macrophages increased intracellular reactive oxygen species (ROS) levels by ~ 2-fold, compared to untreated controls. When J774 cells were co-treated with antioxidant N-acetylcystein, the cadmium-induced FPN1 mRNA induction was significantly attenuated. In summary, the results of this study clearly demonstrated that cadmium increased FPN1 expression in macrophages through a mechanism that involves ROS production, and suggests another important interaction between iron and cadmium metabolism.
Keyword
MeSH Terms
Figure
Reference
-
1. Abboud S, Haile DJ. A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J Biol Chem. 2000. 275:19906–19912.
Article2. Agency for Toxic Substance and Disease Registry. Toxicological Profile for Cadmium. 2003. Atlanta, GA. USA: U.S. Department of Health and Humans Services, Public Health Service, Centers for Disease Control.3. Aydemir F, Jenkitkasemwong S, Gulec S, Knutson MD. Iron loading increases ferroportin heterogeneous nuclear RNA and mRNA levels in murine J774 macrophages. J Nutr. 2009. 139:434–438.
Article4. Bakshi S, Zhang X, Godoy-Tundidor S, Cheng RY, Sartor MA, Medvedovic M, Ho SM. Transcriptome analyses in normal prostate epithelial cells exposed to low-dose cadmium: Oncogenic and immunomodulations involving the action of tumor necrosis factor. Environ Health Perspect. 2008. 116:769–776.
Article5. Bressler JP, Olivi L, Cheong JH, Kim Y, Bannona D. Divalent metal transporter 1 in lead and cadmium transport. Ann N Y Acad Sci. 2004. 1012:142–152.
Article6. Chung J, Haile DJ, Wessling-Resnick M. Copper-induced ferroportin-1 expression in J774 macrophages is associated with increased iron efflux. Proc Natl Acad Sci U S A. 2004. 101:2700–2705.
Article7. Djukic-Cosic D, Curcic Jovanovic M, Plamenac Bulat Z, Ninkovic M, Malicevic Z, Matovic V. Relation between lipid peroxidation and iron concentration in mouse liver after acute and subacute cadmium intoxication. J Trace Elem Med Biol. 2008. 22:66–72.
Article8. Donovan A, Brownlie A, Zhou Y, Shepard J, Pratt SJ, Moynihan J, Paw BH, Drejer A, Barut B, Zapata A, Law TC, Brugnara C, Lux SE, Pinkus GS, Pinkus JL, Kingsley PD, Palis J, Fleming MD, Andrews NC, Zon LI. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature. 2000. 403:776–781.
Article9. Ganz T, Nemeth E. Regulation of iron acquisition and iron distribution in mammals. Biochim Biophys Acta. 2006. 1763:690–699.
Article10. Gaubin Y, Vaissade F, Croute F, Beau B, Soleilhavoup J, Murat J. Implication of free radicals and glutathione in the mechanism of cadmium-induced expression of stress proteins in the A549 human lung cell-line. Biochim Biophys Acta. 2000. 1495:4–13.
Article11. Gunshin H, Mackenzie B, Berger UV, Gunshin Y, Romero MF, Boron WF, Nussberger S, Gollan JL, Hediger MA. Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature. 1997. 388:482–488.
Article12. Hintze KJ, Theil EC. Cellular regulation and molecular interactions of the ferritins. Cell Mol Life Sci. 2006. 63:591–600.
Article13. Horiguchi H. Anemia induced by cadmium intoxication. Nippon Eiseigaku Zasshi. 2007. 62:888–904.
Article14. Im JY, Paik SG, Han PL. Cadmium-induced astroglial death proceeds via glutathione depletion. J Neurosci Res. 2006. 83:301–308.
Article15. International Agency for Research on Cancer. Berrylium, cadmium, mercury and exposures in the glass manufacturing industry. International Agency for Research on Cancer Monographs on the Evaluation of Carcinogenic Risks to Humans. 1993. 58. Lyon. France: IARC Scientific Publications;119–237.16. Iwasaki K, Mackenzie EL, Hailemariam K, Sakamoto K, Tsuji Y. Hemin-mediated regulation of an antioxidant-responsive element of the human ferritin H gene and role of ref-1 duringerythroid differentiation of K562 cells. Mol Cell Biol. 2006. 26:2845–2856.
Article17. Järup L. Hazards of heavy metal contamination. Br Med Bull. 2003. 68:167–182.
Article18. Järup L, Berglund M, Elinder CG, Nordberg G, Vahter M. Health effects of cadmium exposure--a review of the literature and a risk estimate. Scand J Work Environ Health. 1998. 24:Suppl 1. 1–51.19. Knutson MD, Vafa MR, Haile DJ, Wessling-Resnick M. Iron loading and erythrophagocytosis increase ferroportin 1 (FPN1) expression in J774 macrophages. Blood. 2003. 102:4191–4197.
Article20. Koizumi T, Shirakura H, Kumagai H, Tatsumoto H, Suzuki KT. Mechanism of cadmium-induced cytotoxicity in rat hepatocytes: Cadmium-induced active oxygen-related permeability changes of the plasma membrane. Toxicology. 1996. 114:125–134.
Article21. Lee MJ, Nishio H, Ayaki H, Yamamoto M, Sumino K. Upregulation of stress response mRNAs in COS-7 cells exposed to cadmium. Toxicology. 2002. 174:109–117.
Article22. Lopez E, Arce C, Oset-Gasque MJ, Canadas S, Gonzalez MP. Cadmium induces reactive oxygen species generation and lipid peroxidation in cortical neurons in culture. Free Radic Biol Med. 2006. 40:940–951.
Article23. McKie AT, Marciani P, Rolfs A, Brennan K, Wehr K, Barrow D, Miret S, Bomford A, Peters TJ, Farzaneh F, Hediger MA, Hentze MW, Simpson RJ. A novel duodenal iron-regulated transporter, IREG1, implicated in the basolateral transfer of iron to the circulation. Mol Cell. 2000. 5:299–309.
Article24. Pari L, Murugavel P. Diallyl tetrasulfide improves cadmium induced alterations of acetylcholinesterase, ATPases and oxidative stress in brain of rats. Toxicology. 2007. 234:44–50.
Article25. Park BY, Chung J. Effects of various metal ions on the gene expression of iron exporter ferroportin-1 in J774 macrophages. Nutrition Research and Practice. 2008. 2:317–321.
Article26. Pathak N, Khandelwal S. Role of oxidative stress and apoptosis in cadmium induced thymic atrophy and splenomegaly in mice. Toxicol Lett. 2007. 169:95–108.
Article27. Rahman I, Kode A, Biswas SK. Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat Protoc. 2006. 1:3159–3165.
Article28. Ramirez DC, Gimenez MS. Induction of redox changes, inducible nitric oxide synthase and cyclooxygenase-2 by chronic cadmium exposure in mouse peritoneal macrophages. Toxicol Lett. 2003. 145:121–132.
Article29. Ryu DY, Lee SJ, Park DW, Choi BS, Klaassen CD, Park JD. Dietary iron regulates intestinal cadmium absorption through iron transporters in rats. Toxicol Lett. 2004. 152:19–25.
Article30. Satarug S, Baker JR, Urbenjapol S, Haswell-Elkins M, Reilly PE, Williams DJ, Moore MR. A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett. 2003. 137:65–83.
Article31. Schafer SG, Schwegler U, Schumann K. Retention of cadmium in cadmium-naive normal and iron-deficient rats as well as in cadmium-induced iron-deficient animals. Ecotoxicol Environ Saf. 1990. 20:71–81.
Article32. Shaikh ZA, Vu TT, Zaman K. Oxidative stress as a mechanism of chronic cadmium-induced hepatotoxicity and renal toxicity and protection by antioxidants. Toxicol Appl Pharmacol. 1999. 154:256–263.
Article33. Staessen JA, Buchet JP, Ginucchio G, Lauwerys RR, Lijnen P, Roels H, Fagard R. Public health implications of environmental exposure to cadmium and lead: An overview of epidemiological studies in belgium. working groups. J Cardiovasc Risk. 1996. 3:26–41.
Article34. Sureda A, Hebling U, Pons A, Mueller S. Extracellular H2O2 and not superoxide determines the compartment-specific activation of transferrin receptor by iron regulatory protein 1. Free Radic Res. 2005. 39:817–824.
Article35. Thevenod F, Friedmann JM, Katsen AD, Hauser IA. Up-regulation of multidrug resistance P-glycoprotein via nuclear factor-kappaB activation protects kidney proximal tubule cells from cadmium- and reactive oxygen species-induced apoptosis. J Biol Chem. 2000. 275:1887–1896.36. Tsuji Y. JunD activates transcription of the human ferritin H gene through an antioxidant response element during oxidative stress. Oncogene. 2005. 24:7567–7578.
Article37. Tsuji Y, Ayaki H, Whitman SP, Morrow CS, Torti SV, Torti FM. Coordinate transcriptional and translational regulation offerritin in response to oxidative stress. Mol Cell Biol. 2000. 20:5818–5827.
Article38. Yang Z, Yang S, Qian SY, Hong JS, Kadiiska MB, Tennant RW, Waalkes MP, Liu J. Cadmium-induced toxicity in rat primary mid-brain neuroglia cultures: Role of oxidative stress from microglia. Toxicol Sci. 2007. 98:488–494.
Article39. Weiss G. Iron and immunity: A double-edged sword. Eur J Clin Invest. 2002. 32:Suppl 1. 70–78.
Article40. World Health Organization. Chapter 6.3. Cadmium. 2000. Geneva. Switzerland: 1–11.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Copper on the Regulation of Ferroportin-1 Gene Expression
- Effects of various metal ions on the gene expression of iron exporter ferroportin-1 in J774 macrophages
- Rhamnazin inhibits LPS-induced inflammation and ROS/RNS in raw macrophages
- Effect of High Glucose on the Production of Reactive Oxygen Species in Trabecular Meshwork Cells
- Effect of Salicylate on the Monocyte Chemoattractant Protein-1 Expression and Intracellular Reactive Oxygen Species Formation in Human Mesangial Cells