Lab Med Online.
2012 Oct;2(4):188-196.
Measurement of Serum Levels of 25-Hydroxyvitamin D3 and 25-Hydroxyvitamin D2 Using Diels-Alder Derivatization and Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry
- Affiliations
-
- 1Department of Laboratory Medicine, Seoul National University College of Medicine, Seoul, Korea. songjhcp@snu.ac.kr
- 2Department of Laboratory Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
Abstract
- BACKGROUND
Vitamin D has been recently shown to play important roles in the functioning of various systems. Most of the current analytical methods for measuring vitamin D levels are based on immunoassays. We simultaneously measured the levels of 25-hydroxyvitamin D3 [ 25(OH)D3 ] and 25-hydroxyvitamin D2 [ 25(OH)D2 ] in human serum by performing ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) after Diels-Alder derivatization with 4-phenyl-1,2,4-triazoline-3,5-dione (PTAD) and evaluated the performance of our method.
METHODS
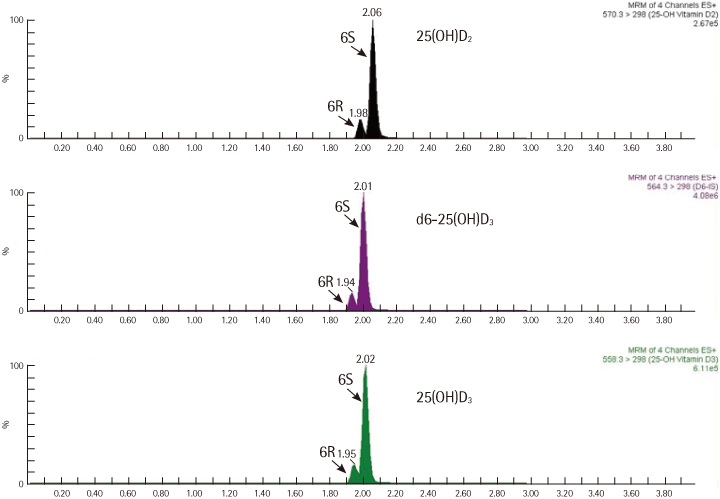
After liquid-liquid extraction, samples were dried under N2 at 50degrees C for 1 hr followed by Diels-Alder derivatization with ethyl acetate containing 0.1 mg/mL PTAD. The samples were resuspended in 60 microL of methanol:10 mM ammonium formate solution (1:1, V/V). C18 UPLC column and positive ion multiple reaction monitoring transitions such as m/z 558.35-->298.1, 25(OH)D3; m/z 570.35-->298.1, 25(OH)D2; and m/z 564.35-->298.1, hexadeuterated-25(OH)D3 were used for UPLC-MS/MS.
RESULTS
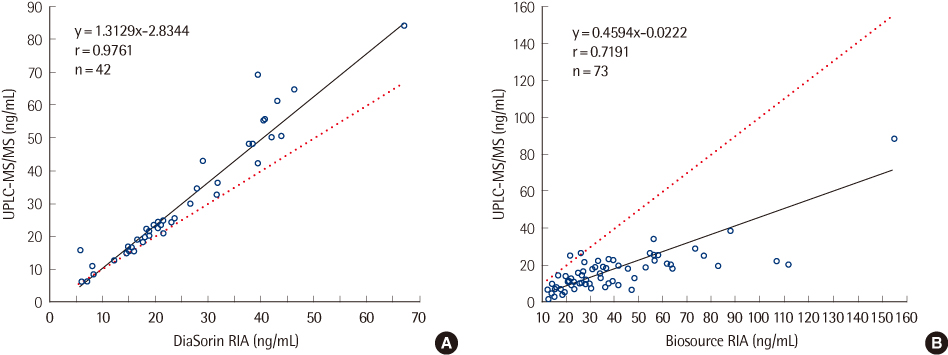
The within-run imprecision (CVs) for 25(OH)D3 and 25(OH)D2 were 3.5-4.0% and 3.8-4.2%, respectively, and the corresponding between-run CVs were 3.3-5.5% and 4.7-5.8%. The lower limit of quantification for 25(OH)D3 and 25(OH)D2 were 0.5 and 1.0 ng/mL, respectively. The curve for interassay calibration variability data obtained over concentrations of 0-120 ng/mL for 25(OH)D3 and 0-90 ng/mL for 25(OH)D2 was linear and reproducible [ 25(OH)D3, R2=0.993; 25(OH)D2, R2=0.998]. The total 25(OH)D levels in Koreans (average, 18.7 ng/mL) were lower than those in American Caucasians, and the percentage of people with total 25(OH)D levels under 10 ng/mL was 8.1%.
CONCLUSIONS
Our method to measure 25(OH)D3 and 25(OH)D2 levels by performing UPLC-MS/MS after PTAD derivatization showed good performance as a sensitive and reproducible method for routine analysis of vitamin D status.
Keyword
MeSH Terms
Figure
Reference
-
1. Norman AW. From vitamin D to hormone D: fundamentals of the vitamin D endocrine system essential for good health. Am J Clin Nutr. 2008. 88(2):491S–499S.
Article2. Dusso AS, Brown AJ, Slatopolsky E. Vitamin D. Am J Physiol Renal Physiol. 2005. 289:F8–F28.
Article3. Holick MF. Vitamin D deficiency. N Engl J Med. 2007. 357:266–281.
Article4. Rajakumar K, Greenspan SL, Thomas SB, Holick MF. SOLAR ultraviolet radiation and vitamin D: a historical perspective. Am J Public Health. 2007. 97:1746–1754.5. Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004. 80:6 Suppl. 1678S–1688S.
Article6. Melamed ML, Muntner P, Michos ED, Uribarri J, Weber C, Sharma J, et al. Serum 25-hydroxyvitamin D levels and the prevalence of peripheral arterial disease: results from NHANES 2001 to 2004. Arterioscler Thromb Vasc Biol. 2008. 28:1179–1185.
Article7. Holick MF. Vitamin D status: measurement, interpretation, and clinical application. Ann Epidemiol. 2009. 19:73–78.
Article8. Zhang R, Naughton DP. Vitamin D in health and disease: current perspectives. Nutr J. 2010. 9:65.
Article9. Shimada K, Mitamura K, Higashi T. Gas chromatography and high-performance liquid chromatography of natural steroids. J Chromatogr A. 2001. 935:141–172.
Article10. Hollis BW. Measuring 25-hydroxyvitamin D in a clinical environment: challenges and needs. Am J Clin Nutr. 2008. 88:2. 507S–510S.
Article11. Binkley N, Krueger D, Cowgill CS, Plum L, Lake E, Hansen KE, et al. Assay variation confounds the diagnosis of hypovitaminosis D: a call for standardization. J Clin Endocrinol Metab. 2004. 89:3152–3157.
Article12. Glendenning P, Taranto M, Noble JM, Musk AA, Hammond C, Goldswain PR, et al. Current assays overestimate 25-hydroxyvitamin D3 and underestimate 25-hydroxyvitamin D2 compared with HPLC: need for assay-specific decision limits and metabolite-specific assays. Ann Clin Biochem. 2006. 43:23–30.
Article13. Roth HJ, Schmidt-Gayk H, Weber H, Niederau C. Accuracy and clinical implications of seven 25-hydroxyvitamin D methods compared with liquid chromatography-tandem mass spectrometry as a reference. Ann Clin Biochem. 2008. 45:153–159.
Article14. Lee J, Vasikaran S. Current recommendations for laboratory testing and use of bone turnover markers in management of osteoporosis. Ann Lab Med. 2012. 32:105–112.
Article15. de la Hunty A, Wallace AM, Gibson S, Viljakainen H, Lamberg-Allardt C, Ashwell M. UK Food Standards Agency Workshop Consensus Report: the choice of method for measuring 25-hydroxyvitamin D to estimate vitamin D status for the UK National Diet and Nutrition Survey. Br J Nutr. 2010. 104:612–619.
Article16. Clinical and Laboratory Standards Institute. Document EP6-A. Evaluation of the linearity of quantitative measurement procedures: a statistical approach; Approved guideline. 2003. Wayne, PA: Clinical and Laboratory Standards Institute.17. Bonfiglio R, King RC, Olah TV, Merkle K. The effects of sample preparation methods on the variability of the electrospray ionization response for model drug compounds. Rapid Commun Mass Spectrom. 1999. 13:1175–1185.
Article18. Aronov PA, Hall LM, Dettmer K, Stephensen CB, Hammock BD. Metabolic profiling of major vitamin D metabolites using Diels-Alder derivatization and ultra-performance liquid chromatography-tandem mass spectrometry. Anal Bioanal Chem. 2008. 391:1917–1930.
Article19. Ding S, Schoenmakers I, Jones K, Koulman A, Prentice A, Volmer DA. Quantitative determination of vitamin D metabolites in plasma using UHPLC-MS/MS. Anal Bioanal Chem. 2010. 398:779–789.
Article20. Lind C, Chen J, Byrjalsen I. Enzyme immunoassay for measuring 25-hydroxyvitamin D3 in serum. Clin Chem. 1997. 43:943–949.
Article21. Jones G. Assay of vitamins D2 and D3, and 25-hydroxyvitamins D2 and D3 in human plasma by high-performance liquid chromatography. Clin Chem. 1978. 24:287–298.
Article22. Lensmeyer GL, Wiebe DA, Binkley N, Drezner MK. HPLC method for 25-hydroxyvitamin D measurement: comparison with contemporary assays. Clin Chem. 2006. 52:1120–1126.
Article23. Shimada K, Mitamura K, Kitama N. Quantitative determination of 25-hydroxyvitamin D3 3-sulphate in human plasma using high performance liquid chromatography. Biomed Chromatogr. 1995. 9:229–232.
Article24. Maunsell Z, Wright DJ, Rainbow SJ. Routine isotope-dilution liquid chromatography-tandem mass spectrometry assay for simultaneous measurement of the 25-hydroxy metabolites of vitamins D2 and D3. Clin Chem. 2005. 51:1683–1690.
Article25. Saenger AK, Laha TJ, Bremner DE, Sadrzadeh SM. Quantification of serum 25-hydroxyvitamin D(2) and D(3) using HPLC-tandem mass spectrometry and examination of reference intervals for diagnosis of vitamin D deficiency. Am J Clin Pathol. 2006. 125:914–920.
Article26. Chen H, McCoy LF, Schleicher RL, Pfeiffer CM. Measurement of 25-hydroxyvitamin D3 (25OHD3) and 25-hydroxyvitamin D2 (25OHD2) in human serum using liquid chromatography-tandem mass spectrometry and its comparison to a radioimmunoassay method. Clin Chim Acta. 2008. 391:6–12.
Article27. Højskov CS, Heickendorff L, Møller HJ. High-throughput liquid-liquid extraction and LCMSMS assay for determination of circulating 25(OH) vitamin D3 and D2 in the routine clinical laboratory. Clin Chim Acta. 2010. 411:114–116.
Article28. Kushnir MM, Ray JA, Rockwood AL, Roberts WL, La'ulu SL, Whittington JE, et al. Rapid analysis of 25-hydroxyvitamin D(2) and D(3) by liquid chromatography-tandem mass spectrometry and association of vitamin D and parathyroid hormone concentrations in healthy adults. Am J Clin Pathol. 2010. 134:148–156.
Article29. Vogeser M. Quantification of circulating 25-hydroxyvitamin D by liquid chromatography-tandem mass spectrometry. J Steroid Biochem Mol Biol. 2010. 121:565–573.
Article30. Schleicher RL, Encisco SE, Chaudhary-Webb M, Paliakov E, McCoy LF, Pfeiffer CM. Isotope dilution ultra performance liquid chromatography-tandem mass spectrometry method for simultaneous measurement of 25-hydroxyvitamin D2, 25-hydroxyvitamin D3 and 3-epi-25-hydroxyvitamin D3 in human serum. Clin Chim Acta. 2011. 412:1594–1599.
Article31. Higashi T, Suzuki M, Hanai J, Inagaki S, Min JZ, Shimada K, et al. A specific LC/ESI-MS/MS method for determination of 25-hydroxyvitamin D3 in neonatal dried blood spots containing a potential interfering metabolite, 3-epi-25-hydroxyvitamin D3. J Sep Sci. 2011. 34:725–732.
Article32. Wang Z, Senn T, Kalhorn T, Zheng XE, Zheng S, Davis CL, et al. Simultaneous measurement of plasma vitamin D(3) metabolites, including 4 β,25-dihydroxyvitamin D(3), using liquid chromatography-tandem mass spectrometry. Anal Biochem. 2011. 418:126–133.
Article33. Armas LA, Hollis BW, Heaney RP. Vitamin D2 is much less effective than vitamin D3 in humans. J Clin Endocrinol Metab. 2004. 89:5387–5391.34. Houghton LA, Vieth R. The case against ergocalciferol (vitamin D2) as a vitamin supplement. Am J Clin Nutr. 2006. 84:694–697.
Article35. Adamec J, Jannasch A, Huang J, Hohman E, Fleet JC, Peacock M, et al. Development and optimization of an LC-MS/MS-based method for simultaneous quantification of vitamin D2, vitamin D3, 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3. J Sep Sci. 2011. 34:11–20.
Article36. Singh RJ. Are clinical laboratories prepared for accurate testing of 25-hydroxy vitamin D? Clin Chem. 2008. 54:221–223.
Article37. Stepman HC, Vanderroost A, Van Uytfanghe K, Thienpont LM. Candidate reference measurement procedures for serum 25-hydroxyvitamin D3 and 25-hydroxyvitamin D2 by using isotope-dilution liquid chromatography-tandem mass spectrometry. Clin Chem. 2011. 57:441–448.
Article38. Carter GD, Jones JC. Use of a common standard improves the performance of liquid chromatography-tandem mass spectrometry methods for serum 25-hydroxyvitamin-D. Ann Clin Biochem. 2009. 46:79–81.
Article39. Yates AM, Bowron A, Calton L, Heynes J, Field H, Rainbow S, et al. Interlaboratory variation in 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3 is significantly improved if common calibration material is used. Clin Chem. 2008. 54:2082–2084.
Article40. Ginde AA, Liu MC, Camargo CA Jr. Demographic differences and trends of vitamin D insufficiency in the US population, 1988-2004. Arch Intern Med. 2009. 169:626–632.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Efficacy of the Measurement of 25-Hydroxyvitamin D2 and D3 Levels by Using PerkinElmer Liquid Chromatography-Tandem Mass Spectrometry Vitamin D Kit Compared With DiaSorin Radioimmunoassay Kit and Elecsys Vitamin D Total Assay
- Performance Evaluation of Method for Detecting Serum 25-Hydroxyvitamin D2 and 25-Hydroxyvitamin D3 by Using PerkinElmer MSMS Vitamin D Kit
- Vitamin D Levels and Prevalence of Vitamin D Deficiency Associated with Sex, Age, Region, and Season in Koreans
- Correlation between serum 25-hydroxyvitamin D3 and the severity of atopic dermatitis in children with allergic or nonallergenic sensitization
- Efficacy of plain cholecalciferol versus ergocalciferol in raising serum vitamin D level in Thai female healthcare workers