Lab Anim Res.
2010 Jun;26(2):203-209. 10.5625/lar.2010.26.2.203.
The Effect of Quercetin on the BK(Ca) in Umbilical Cord Vein-derived Mesenchymal Stem Cells
- Affiliations
-
- 1Department of Physiology, College of Medicine, Seoul National University, Seoul, Korea.
- 2Department of Physiology, College of Medicine, Chungbuk National University, Cheongju, Korea. yangmik@chungbuk.ac.kr
- KMID: 2312066
- DOI: http://doi.org/10.5625/lar.2010.26.2.203
Abstract
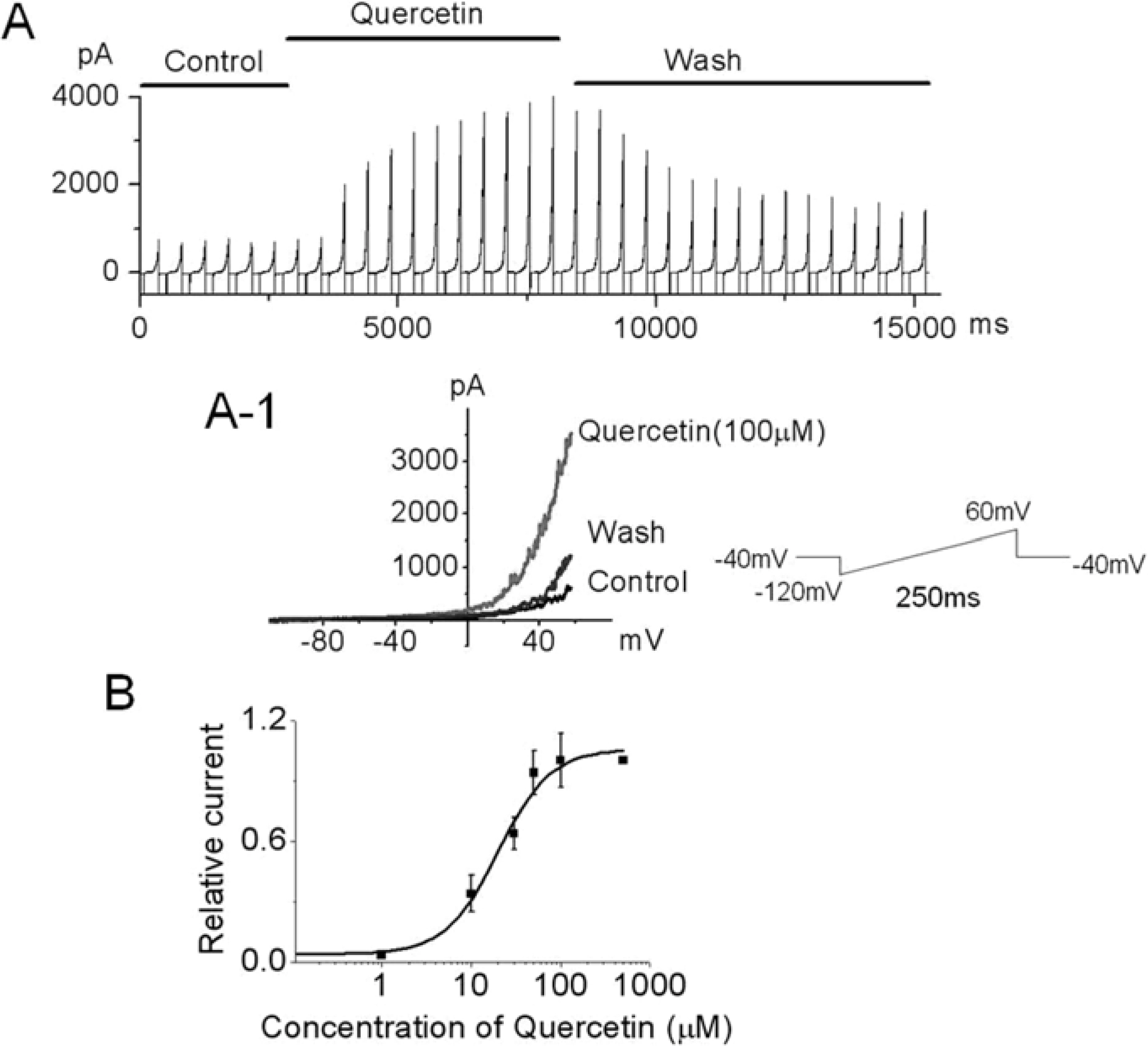
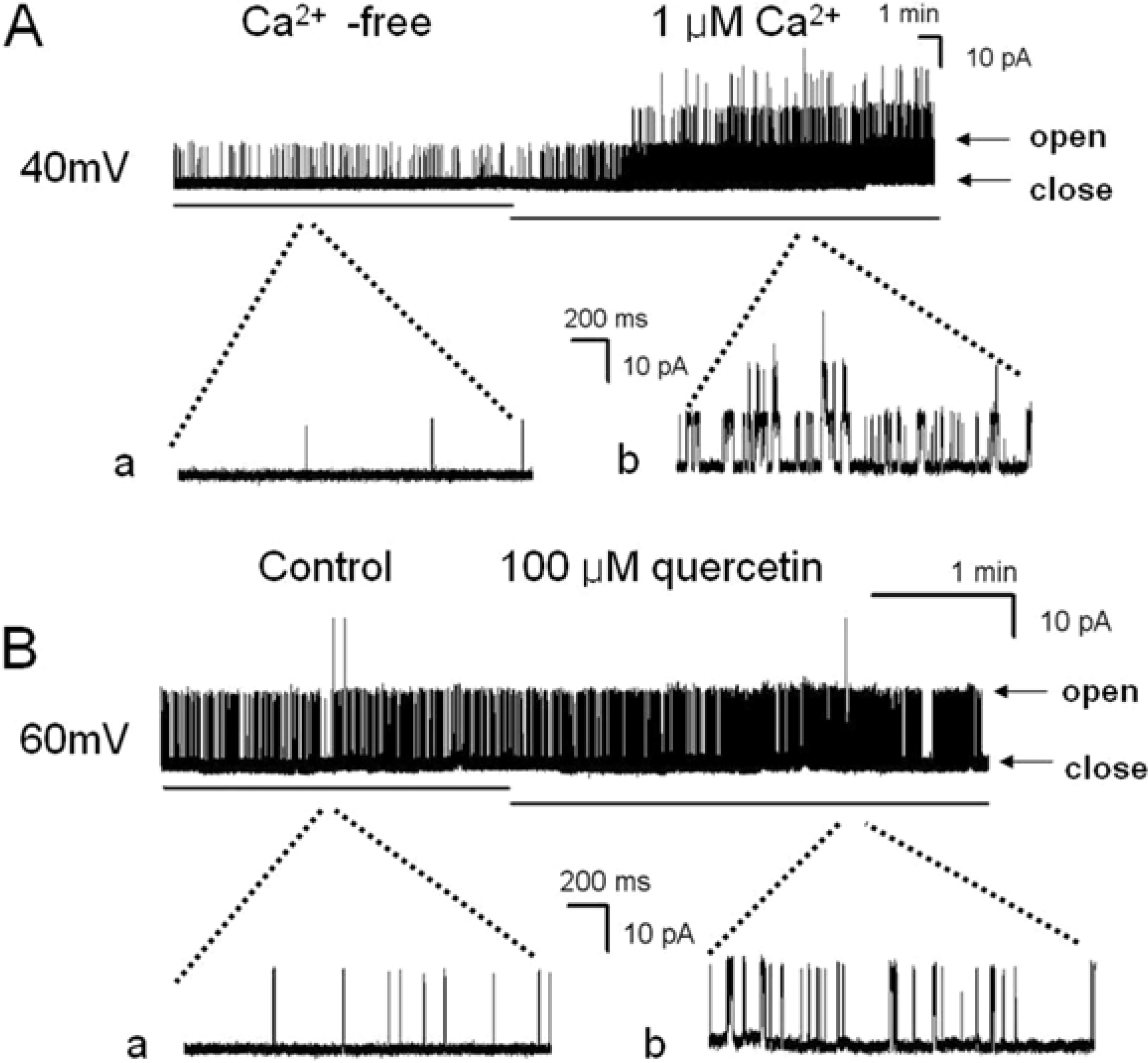
- Recently, we demonstrated that a large conductance Ca2+-activated K+ channel (BK(Ca)) in human umbilical cord vein-derived mesenchymal cells (hUC-MSCs). In this study, we studied effects of quercetin, the flavonoid used for the modulator of cell cycle and the treatment of anti-tumor, on BK(Ca) in hUC-MSCs using single channel and/or whole cell configuration. Single channel conductance was 277.8+/-36.2 pS in hUC-MSCs and the value is similar to that of typical BK(Ca) channel reported in other cells. The single channel current was activated by increasing of intracellular Ca2+. Quercetin (30~100 micrometer) increased BK(Ca) in whole cell patch configuration whereas EGCG was not affected on the channel. The activation effect of quercetin on BK(Ca) channel was also observed in inside-out configuration. The quercetin-induced BK(Ca) was increased a concentration-dependent manner with an IC50 value of 19.9 micrometer. Taken together, BK(Ca) in hUC-MSCs can be an important target for the action of quercetin and the channels are partly may contribute to maintaining homeostasis by regulation of K ion flux in hUC-MSCs.
MeSH Terms
Figure
Reference
-
Cermak R.., Follmer U.., Wolffram S.1998. ).Dietary flavonol quercetin induces chloride secretion in rat colon. Am. J. Physiol. 275(5 Pt 1):G1166–1172.Choi J. A.., Kim J. Y.., Lee J. Y.., Kang C. M.., Kwon H. J.., Yoo Y. D.., Kim T. W.., Lee Y. S.., Lee S. J.2001. Induction of cell cycle arrest and apoptosis in human breast cancer cells by quercetin. Int. J. Oncol. 19(4):837–844.
ArticleCogolludo A.., Frazziano G.., Briones A. M.., Cobeno L.., Moreno L.., Lodi F.., Salaices M.., Tamargo J.., Perez-Vizcaino F.2007. The dietary flavonoid quercetin activates BKCa currents in coronary arteries via production of H2O2. Role in vasodilatation. Cardiovasc. Res. 73(2):424–431.Duarte J.., Perez-Palencia R.., Vargas F.., Ocete M. A.., Perez-Vizcaino F.., Zarzuelo A.., Tamargo J.2001. Antihypertensive effects of the flavonoid quercetin in spontaneously hypertensive rats. Br. J. Pharmacol. 133(1):117–124.
ArticleHertog M. G.., Hollman P. C.1996. Potential health effects of the dietary flavonol quercetin. Eur. J. Clin. Nutr. 50(2):63.Kuhlmann C. R.., Schaefer C. A.., Kosok C.., Abdallah Y.., Walther S.., Ludders D. W.., Neumann T.., Tillmanns H.., Schafer C.., Piper H. M.., Erdogan A.2005. Quercetin-induced induction of the NO/cGMP pathway depends on Ca2+-activated K+ channel-induced hyperpolarization-mediated Ca2+-entry into cultured human endothelial cells. Planta Med. 71(6):520–524.Lee E. H.., Meissner G.., Kim D. H.2002a. Effects of quercetin on single Ca2+ release channel behavior of skeletal muscle. Biophys. J. 82(3):1266–1277.Lee L. T.., Huang Y. T.., Hwang J. J.., Lee P. P.., Ke F. C.., Nair M. P.., Kanadaswam C.., Lee M. T.2002b. Blockade of the epidermal growth factor receptor tyrosine kinase activity by quercetin and luteolin leads to growth inhibition and apoptosis of pancreatic tumor cells. Anticancer Res. 22(3):1615–1627.Lin R. W.., Chen C. H.., Wang Y. H.., Ho M. L.., Hung S. H.., Chen I. S.., Wang G. J.2009. (-)-Epigallocatechin gallate inhibition of osteoclastic differentiation via NF-kappaB. Biochem. Biophys. Res. Commun. 379(4):1033–1037.Park K. S.., Jung K. H.., Kim S. H.., Kim K. S.., Choi M. R.., Kim Y.., Chai Y. G.2007. Functional expression of ion channels in mesenchymal stem cells derived from umbilical cord vein. Stem Cells. 25(8):2044–2052.
ArticlePark K. S.., Jung K. H.., Chai Y. G.., Kim C. H.., Shin E. Y.., Kim Y.2008. Riluzole-induced activation of BKCa channels and inhibition of voltage-gated Na+ channel in human umbilical cord vein-derived mesenchymal cells. Lab. Anim. Res. 24(3):287–292.Rassi C. M.., Lieberherr M.., Chaumaz G.., Pointillart A.., Cournot G.2005. Modulation of osteoclastogenesis in porcine bone marrow cultures by quercetin and rutin. Cell Tissue Res. 319(3):383–393.
ArticleRoger S.., Potier M.., Vandier C.., Le Guennec J. Y.., Besson P.2004. Description and role in proliferation of iberiotoxin-sensitive currents in different human mammary epithelial normal and cancerous cells. Biochim. Biophys. Acta 1667. (2):190–199.Sanchez M.., Galisteo M.., Vera R.., Villar I. C.., Zarzuelo A.., Tamargo J.., Perez-Vizcaino F.., Duarte J.2006. Quercetin downregulates NADPH oxidase, increases eNOS activity and prevents endothelial dysfunction in spontaneously hypertensive rats. J. Hypertens. 24(1):75–84.Saponara S.., Sgaragli G.., Fusi F.2002. Quercetin as a novel activator of L-type Ca2+ channels in rat tail artery smooth muscle cells. Br. J. Pharmacol. 135(7):1819–1827.Shin D. W.., Kim S. N.., Lee S. M.., Lee W.., Song M. J.., Park S. M.., Lee T. R.., Baik J. H.., Kim H. K.., Hong J. H.., Noh M.2009. (-)-Catechin promotes adipocyte differentiation in human bone marrow mesenchymal stem cells through PPAR gamma transactivation. Biochem. Pharmacol. 77(1):125–133.Wang S.., Degroff V. L.., Clinton S. K.2003. Tomato and soy polyphenols reduce insulin-like growth factor-I-stimulated rat prostate cancer cell proliferation and apoptotic resistance in vitro via inhibition of intracellular signaling pathways involving tyrosine kinase. J. Nutr. 133(7):2367–2376.
ArticleYang L.., Ma J. H.., Zhang P. H.., Zou A. R.., Tu D. N.2009. Quercetin activates human Kv1.5 channels by a residue I502 in the S6 segment. Clin. Exp. Pharmacol. Physiol. 36(2):154–161.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Differentiation of Osteoblast Progenitor Cells from Human Umbilical Cord Blood
- Differential Potential of Stem Cells Following Their Origin: Subacromial Bursa, Bone Marrow, Umbilical Cord Blood
- Percutaneous transplantation of human umbilical cord-derived mesenchymal stem cells in a dog suspected to have fibrocartilaginous embolic myelopathy
- Difference in HLA-DR Expression of Human Umbilical Cord Blood Derived Mesenchymal Stem Cells after Tri-lineage Differentiation
- Difference in Cell Characteristics among the Monoclonal Cell Populations Obtained from the Human Umbilical Cord Blood Derived Mesenchymal Stem Cell Population