J Clin Neurol.
2009 Jun;5(2):74-80. 10.3988/jcn.2009.5.2.74.
Cerebellar Hypoperfusion during Transient Global Amnesia: An MRI and Oculographic Study
- Affiliations
-
- 1Department of Neurology, Hyoja Geriatric Hospital, Yongin, Korea.
- 2Department of Neurology, Seoul National University College of Medicine, Seoul, Korea. neuroksy@snu.ac.kr
- 3Department of Nuclear Medicine, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 2287657
- DOI: http://doi.org/10.3988/jcn.2009.5.2.74
Abstract
-
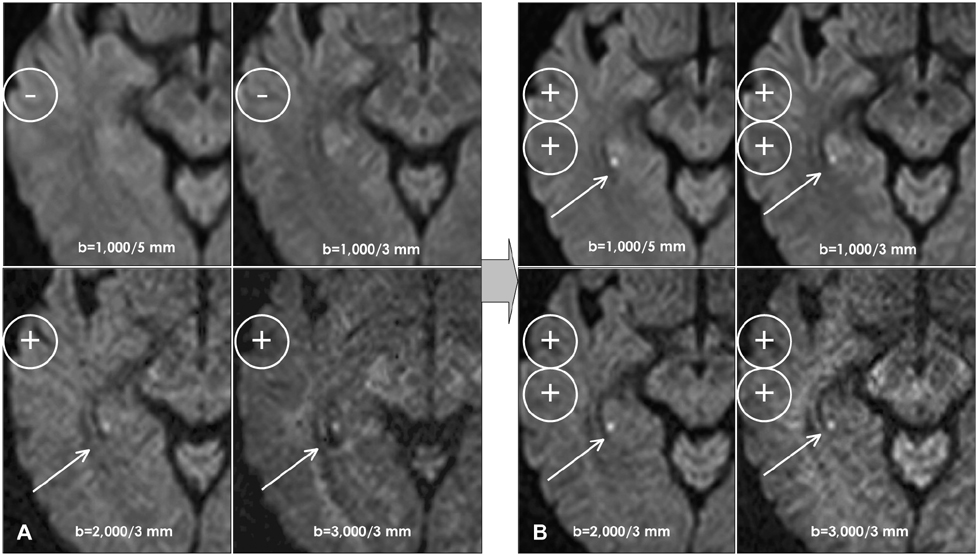
BACKGROUND AND PURPOSE: Transient global amnesia (TGA) is characterized by sudden anterograde and retrograde amnesia lasting for up to 24 hours. Diffusion-weighted magnetic resonance imaging (DWI) in cases of TGA and ischemia demonstrates a high frequency of high signal intensities restricted to the hippocampus, and this has been proposed as an etiology of TGA. The aims of this study were to characterize the DWI and single-photon-emission computed tomography (SPECT) findings during the acute and recovered phases of TGA and to correlate the findings with oculomotor abnormalities.
METHODS
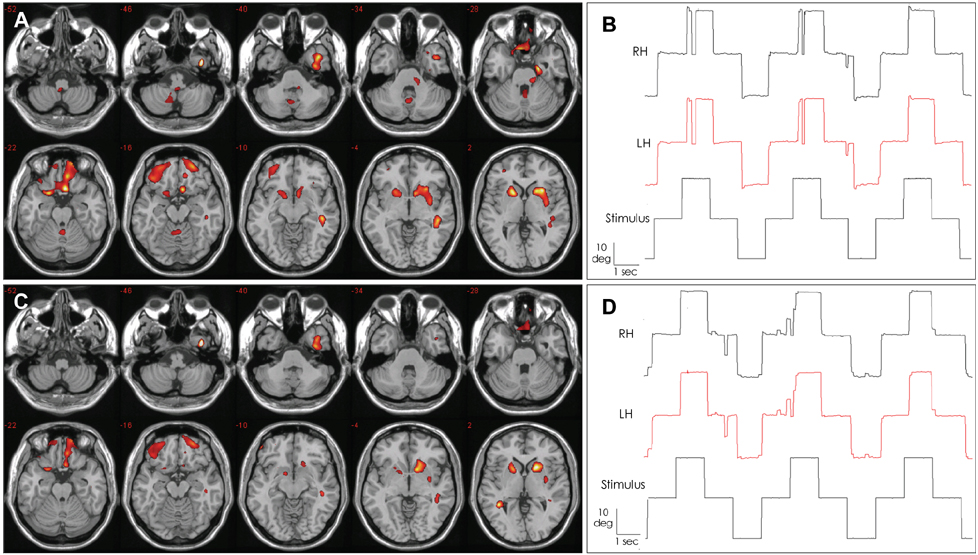
Five consecutive patients with a clinical diagnosis of TGA underwent DWI and SPECT of the brain within 24 hours after symptom onset and again 3 days later. Eye movements were also recorded using three-dimensional video-oculography.
RESULTS
In all patients, DWI disclosed small punctuate (1-3 mm), high-signal lesions in the lateral portion of the hippocampus. The initial SPECT also revealed hypoperfusion in the cerebellar vermis, which had recovered by the follow-up examination. Three patients showed saccadic hypermetria or impaired smooth pursuit only during the acute phase.
CONCLUSIONS
Our patients with TGA showed cerebellar vermian hypoperfusion in addition to ischemic insults to the lateral hippocampus. The oculomotor abnormalities observed in our patients support the occurrence of cerebellar dysfunction during the TGA attack.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Impaired Smooth Pursuit During Transient Global Amnesia
Sung-Hee Kim, Young Ho Park, SangYun Kim, Ji-Soo Kim
J Clin Neurol. 2019;15(3):301-307. doi: 10.3988/jcn.2019.15.3.301.
Reference
-
1. Hodges JR, Warlow CP. Syndromes of transient amnesia: towards a classification. A study of 153 cases. J Neurol Neurosurg Psychiatry. 1990. 53:834–843.
Article2. Hodges JR, Warlow CP. The aetiology of transient global amnesia. A case-control study of 114 cases with prospective follow-up. Brain. 1990. 113:639–657.3. Tong DC, Grossman M. What causes transient global amnesia? New insights from DWI. Neurology. 2004. 62:2154–2155.
Article4. Jovin TG, Vitti RA, McCluskey LF. Evolution of temporal lobe hypoperfusion in transient global amnesia: a serial single photon emission computed tomography study. J Neuroimaging. 2000. 10:238–241.
Article5. Sander K, Sander D. New insights into transient global amnesia: recent imaging and clinical findings. Lancet Neurol. 2005. 4:437–444.
Article6. Yang YS, Kim SY, Kim JH. Ischemic Evidence of Transient Global Amnesia: Location of the Lesion in the Hippocampus. J Clin Neurol. 2008. 4:59–66.
Article7. Pantoni L, Lamassa M, Inzitari D. Transient global amnesia: a review emphasizing pathogenic aspects. Acta Neurol Scand. 2000. 102:275–283.
Article8. Hodges JR. Semantic memory and frontal executive function during transient global amnesia. J Neurol Neurosurg Psychiatry. 1994. 57:605–608.
Article9. Schmidtke K, Reinhardt M, Krause T. Cerebral perfusion during transient global amnesia: findings with HMPAO SPECT. J Nucl Med. 1998. 39:155–159.10. Lampl Y, Sadeh M, Lorberboym M. Transient global amnesia - not always a benign process. Acta Neurol Scand. 2004. 110:75–79.
Article11. Takeuchi R, Matsuda H, Yoshioka K, Yonekura Y. Cerebral blood flow SPET in transient global amnesia with automated ROI analysis by 3DSRT. Eur J Nucl Med Mol Imaging. 2004. 31:578–589.
Article12. Warren JD, Chartteron B, Thompson PD. A SPECT of the anatomy of transient global amnesia. J Clin Neurosci. 2000. 7:57–59.13. Asada T, Matsuda H, Morooka T, Nakano S, Kimura M, Uno M. Quantitative single photon emission tomography analysis for the diagnosis of transient global amnesia: adaptation of statistical parametric mapping. Psychiatry Clin Neurosci. 2000. 54:691–694.
Article14. Ghelarducci B, Sebastiani L. Classical heart rate conditioning and affective behavior: the role of the cerebellar vermis. Arch Ital Biol. 1997. 135:369–384.15. Levitt JJ, McCarley RW, Nestor PG, Petrescu C, Donnino R, Hirayasu Y, et al. Quantitative volumetric MRI study of the cerebellum and vermis in schizophrenia: clinical and cognitive correlates. Am J Psychiatry. 1999. 156:1105–1107.16. Teicher MH, Andersen SL, Polcari A, Anderson CM, Navalta CP, Kim DM. The neurobiological consequences of early stress and childhood maltreatment. Neurosci Biobehav Rev. 2003. 27:33–44.
Article17. Giedd JN, Blumenthal J, Jeffries NO, Rajapakse JC, Vaituzis AC, Liu H, et al. Development of the human corpus callosum during childhood and adolescence: a longitudinal MRI study. Prog Neuropsychopharmacol Biol Psychiatry. 1999. 23:571–588.
Article18. Heath RG. Brain function in epilepsy: midbrain, medullary, and cerebellar interaction with the rostral forebrain. J Neurol Neurosurg Psychiatry. 1976. 39:1037–1051.
Article19. Anderson CM, Teicher MH, Polcari A, Renshaw PF. Abnormal T2 relaxation time in the cerebellar vermis of adults sexually abused in childhood: potential role of the vermis in stress-enhanced risk for drug abuse. Psychoneuroendocrinology. 2002. 27:231–244.
Article20. Schmahmann JD. Joseph A, editor. A new role for the cerebellum: the modulation of cognition and affect. Movement disorders. 1999. 2nd ed. Boston: Blackwell Science.21. Berquin PC, Giedd JN, Jacobsen LK, Hamburger SD, Krain AL, Rapoport JL, et al. Cerebellum in attention-deficit hyperactivity disorder: a morphometric MRI study. Neurology. 1998. 50:1087–1093.
Article22. Loeber RT, Sherwood AR, Renshaw PF, Cohen BM, Yurgelun-Todd DA. Differences in cerebellar blood volume in schizophrenia and bipolar disorder. Schizophr Res. 1999. 37:81–89.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Transient Global Amnesia Developed in a 14-Year Old Girl during Cellular Phone Texting
- Transient Global Amnesia after Gastroscopy
- Recurrent transient amnesia: a case of transient epileptic amnesia misdiag-nosed as transient global amnesia
- Transient Global Amnesia Developed after Zolpidem Intake
- Retrograde Amnesia as a Predominant Symptom of Transient Global Amnesia