J Breast Cancer.
2009 Mar;12(1):20-26. 10.4048/jbc.2009.12.1.20.
The Incidence of Chemotherapy-induced Amenorrhea and Recovery in Young (<45-year-old) Breast Cancer Patients
- Affiliations
-
- 1Department of Surgery, Korea Cancer Center Hospital, Korea Institute of Radiological & Medical Science, Seoul, Korea. nohwoo@kcch.re.kr
- 2Department of Surgery, Daejeon Hankook Hospital, Daejeon, Korea.
- 3Department of Surgery, College of Medicine, Konkuk University, Seoul, Korea.
- KMID: 2286574
- DOI: http://doi.org/10.4048/jbc.2009.12.1.20
Abstract
-
PURPOSE: Although adjuvant chemotherapy improves the survival of premenopausal breast cancer patients, it could induce the premature menopause. The objective of this study was to investigate the incidence and risk factors of chemotherapy-induced amenorrhea (CIA) and recovery for young (< 45-year-old) breast cancer patients.
METHODS
We examined patients with primary invasive breast cancer who had been treated with surgery and adjuvant chemotherapy from January 2003 to June 2006. All of the patients were younger than 45 year and they had regular menstruation at the time of diagnosis. Amenorrhea was defined as the absence of menstruation for three consecutive months or a serum follicular stimulating hormone level > 30 mIU/mL.
RESULTS
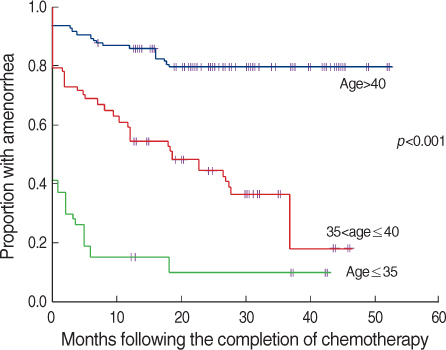
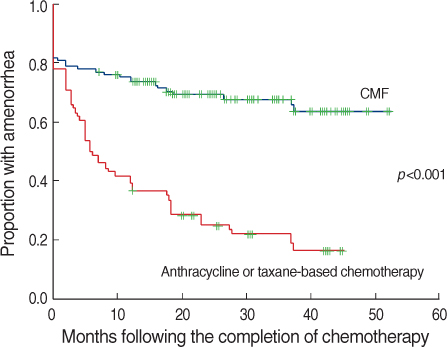
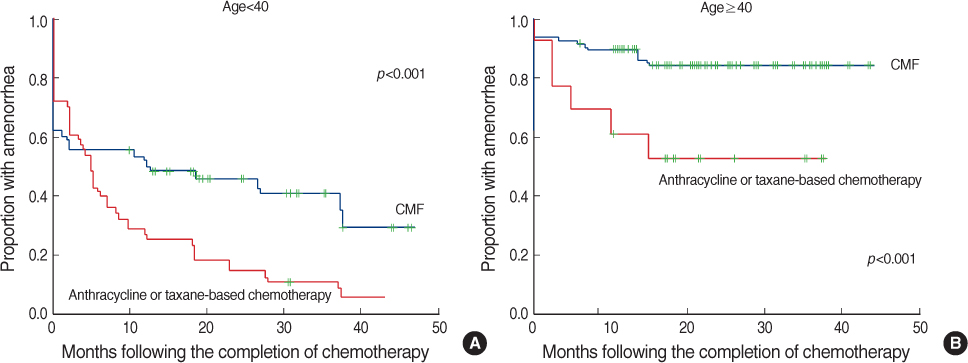
A total of 324 patients were included in this study. Of these patients, 261 patients (80.6%) developed amenorrhea just after the completion of chemotherapy. During follow-up, 77 patients (29.5%) resumed menstruation. Amenorrhea rates at 6, 12, 24, and 36 months after chemotherapy were 72.2%, 66.6%, 58.1%, and 55.5%. Women who recovered from amenorrhea were significantly younger than the women who did not recover (p<0.001). Patients treated with cyclophosphamide, methotrexate and 5-fluorouracil (CMF) less frequently recovered from amenorrhea than patients who were treated with anthracycline or taxane-based chemo- therapy (p<0.001).
CONCLUSION
The age of the patient was the most important factor for the development of CIA. Patients treated with a CMF regimen and older patients showed poor recovery from CIA. These results could be helpful to make decisions about the treatment strategies for premenopausal young women.
MeSH Terms
-
Amenorrhea
Antineoplastic Combined Chemotherapy Protocols
Breast
Breast Neoplasms
Chemotherapy, Adjuvant
Cyclophosphamide
Female
Fluorouracil
Follow-Up Studies
Humans
Incidence
Menopause, Premature
Menstruation
Methotrexate
Risk Factors
Antineoplastic Combined Chemotherapy Protocols
Cyclophosphamide
Fluorouracil
Methotrexate
Figure
Cited by 1 articles
-
Hormonal Changes during Extended Letrozole Treatment after Completion of 5 Years of Tamoxifen in Premenopausal Patients with Breast Cancer who Became Postmenopausal
Ja Young Cho, Hee-Jung Kim, Jong-Won Lee, Jong-Han Yu, Beom-Suk Koh, On Vox Yi, Byung-Ho Son, Sei-Hyun Ahn
J Breast Cancer. 2010;13(4):375-381. doi: 10.4048/jbc.2010.13.4.375.
Reference
-
1. Ahn SH, Yoo KY. The Korean Breast Cancer Society. Chronological changes of clinical characteristics in 31,115 new breast cancer patients among Koreans during 1996-2004. Breast Cancer Res Treat. 2006. 99:209–214.
Article2. Gong GY, Kim MJ, Shim YH, Kang GH, Ahn SH, Ro JY. Nationwide Korean breast cancer data of 2004 using breast cancer registration program. J Breast Cancer. 2006. 9:151–161.
Article3. Hankey BF, Miller B, Curtis R, Kosary C. Trends in breast cancer in younger women in contrast to older women. J Natl Cancer Inst Monogr. 1994. 16:7–14.4. Ahn SH, Son BH, Kim SW, Kim SI, Jeong J, Ko SS, et al. Poor outcome of hormone receptor-positive breast cancer at very young age is due to tamoxifen resistance: nationwide survival data in Korea--a report from the Korean breast cancer society. J Clin Oncol. 2007. 25:2360–2368.
Article5. Xiong Q, Valero V, Kau V, Kau SW, Taylor S, Smith TL, et al. Female patients with breast carcinoma age 30 years and younger have a poor prognosis: the M.D. Anderson cancer center experience. Cancer. 2001. 92:2523–2528.
Article6. de la Rochefordiere A, Asselain B, Campana F, Scholl SM, Fenton J, Vilcoq JR, et al. Age as prognostic factor in premenopausal breast carcinoma. Lancet. 1993. 341:1039–1043.
Article7. Nixon AJ, Neuberg D, Hayes DF, Gelman R, Connolly JL, Schnitt S, et al. Relationship of patient age to pathologic features of the tumor and prognosis for patients with stage I or II breast cancer. J Clin Oncol. 1994. 12:888–894.
Article8. Bland KI, Menck HR, Scott-Conner CE, Morrow M, Winchester DJ, Winchester DP. The national cancer data base 10-year survey of breast carcinoma treatment at hospitals in the United States. Cancer. 1998. 83:1262–1273.
Article9. Walker RA, Lees E, Webb MB, Dearing SJ. Breast carcinomas occurring in young women (< 35 years) are different. Br J Cancer. 1996. 74:1796–1800.
Article10. Early breast cancer trialists' collaborative group. Effects of adjuvant tamoxifen and of cytotoxic therapy on mortality in early breast cancer. An overview of 61 randomized trials among 28,896 women. N Engl J Med. 1988. 319:1681–1692.11. Goldhirsch A, Glick JH, Gelber RD, Coates AS, Thurlimann B, Senn HJ, et al. Meeting highlights: international expert consensus on the primary therapy of early breast cancer 2005. Ann Oncol. 2005. 16:1569–1583.
Article12. Minton SE, Munster PN. Chemotherapy-induced amenorrhea and fertility in women undergoing adjuvant treatment for breast cancer. Cancer Control. 2002. 9:466–472.
Article13. Di Cosimo S, Alimonti A, Ferretti G, Sperduti I, Carlini P, Papaldo P, et al. Incidence of chemotherapy-induced amenorrhea depending on the timing of treatment by menstrual cycle phase in women with early breast cancer. Ann Oncol. 2004. 15:1065–1071.
Article14. Bines J, Oleske DM, Cobleigh MA. Ovarian function in premenopausal women treated with adjuvant chemotherapy for breast cancer. J Clin Oncol. 1996. 14:1718–1729.
Article15. Parulekar WR, Day AG, Ottaway JA, Shepherd LE, Trudeau ME, Bramwell V, et al. Incidence and prognostic impact of amenorrhea during adjuvant therapy in high-risk premenopausal breast cancer: analysis of a national cancer institute of canada clinical trials group study--NCIC CTG MA.5. J Clin Oncol. 2005. 23:6002–6008.
Article16. Goldhirsch A, Gelber RD, Castiglione M. The magnitude of endocrine effects of adjuvant chemotherapy for premenopausal breast cancer patients. the international breast cancer study group. Ann Oncol. 1990. 1:183–188.
Article17. Tham YL, Sexton K, Weiss H, Elledge R, Friedman LC, Kramer R. The rates of chemotherapy-induced amenorrhea in patients treated with adjuvant doxorubicin and cyclophosphamide followed by a taxane. Am J Clin Oncol. 2007. 30:126–132.
Article18. Bonadonna G, Moliterni A, Zambetti M, Daidone MG, Pilotti S, Gianni L, et al. 30 years' follow up of randomised studies of adjuvant CMF in operable breast cancer: cohort study. BMJ. 2005. 330:217.
Article19. Padmanabhan N, Howell A, Rubens RD. Mechanism of action of adjuvant chemotherapy in early breast cancer. Lancet. 1986. 2:411–414.
Article20. Goodwin PJ, Ennis M, Pritchard KI, Trudeau M, Hood N. Risk of menopause during the first year after breast cancer diagnosis. J Clin Oncol. 1999. 17:2365–2370.
Article21. Petrek JA, Naughton MJ, Case LD, Paskett ED, Naftalis EZ, Singletary SE, et al. Incidence, time course, and determinants of menstrual bleeding after breast cancer treatment: a prospective study. J Clin Oncol. 2006. 24:1045–1051.
Article22. Cobleigh MA, Bines J, Harris D. Amenorrhea following adjuvant chemotherapy for breast cancer. Proc Am Soc Clin Oncol. 1995. 14:A158.23. Vanhuyse M, Fournier C, Bonneterre J. Chemotherapy-induced amenorrhea: influence on disease-free survival and overall survival in receptor-positive premenopausal early breast cancer patients. Ann Oncol. 2005. 16:1283–1288.
Article24. Walshe JM, Denduluri N, Swain SM. Amenorrhea in premenopausal women after adjuvant chemotherapy for breast cancer. J Clin Oncol. 2006. 24:5769–5779.
Article25. Baum M, Hackshaw A, Houghton J, Rutqvist , Fornander T, Nordenskjold B, et al. Adjuvant goserelin in pre-menopausal patients with early breast cancer: results from the ZIPP study. Eur J Cancer. 2006. 42:895–904.
Article26. Kaufmann M, Jonat W, Blamey R, Cuzick J, Namer M, Fogelman I, et al. Survival analyses from the ZEBRA study. goserelin (zoladex) versus CMF in premenopausal women with node-positive breast cancer. Eur J Cancer. 2003. 39:1711–1717.27. Thomson CS, Twelves CJ, Mallon EA, Leake RE. Scottish Cancer Trials Breast Group. Scottish Cancer Therapy Network. Adjuvant ovarian ablation vs CMF chemotherapy in premenopausal breast cancer patients: trial update and impact of immunohistochemical assessment of ER status. Breast. 2002. 11:419–429.
Article28. Jonat W, Kaufmann M, Sauerbrei W, Blamey R, Cuzick J, Namer M, et al. Goserelin versus cyclophosphamide, methotrexate, and fluorouracil as adjuvant therapy in premenopausal patients with nodepositive breast cancer: the zoladex early breast cancer research association study. J Clin Oncol. 2002. 20:4628–4635.
Article29. Warne GL, Fairley KF, Hobbs JB, Martin FI. Cyclophosphamide-induced ovarian failure. N Engl J Med. 1973. 289:1159–1162.
Article30. Fisher B, Brown AM, Dimitrov NV, Poisson R, Redmond C, Margolese RG, et al. Two months of doxorubicin-cyclophosphamide with and without interval reinduction therapy compared with 6 months of cyclophosphamide, methotrexate, and fluorouracil in positive-node breast cancer patients with tamoxifen-nonresponsive tumors: results from the national surgical adjuvant breast and bowel project B-15. J Clin Oncol. 1990. 8:1483–1496.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Recovery of Ovarian Function with Aromatase Inhibitors: In Young Breast Cancer Patients (<45) with Chemotherapy-induced Amenorrhea
- Menopausal Symptoms and Quality of Life Among Breast Cancer Patients with Chemotherapy-induced Amenorrhea
- Factors of Occurrence of Amenorrhea and Climacteric Symptoms in Breast Cancer Patients Underwent Chemotherapy
- Risperidone-Induced Amenorrhea in a Premenopausal Woman with Schizophrenia
- Chemotherapy in Breast Cancer