J Breast Cancer.
2008 Sep;11(3):133-138. 10.4048/jbc.2008.11.3.133.
Recovery of Ovarian Function with Aromatase Inhibitors: In Young Breast Cancer Patients (<45) with Chemotherapy-induced Amenorrhea
- Affiliations
-
- 1Department of Surgery, Korea Institute of Radiological & Medical Science, Korea Cancer Center Hospital, Seoul, Korea. nohwoo@kcch.re.kr
- 2Department of Surgery, Kang-Won National University College of Medicine, Chuncheon, Korea.
- KMID: 1485106
- DOI: http://doi.org/10.4048/jbc.2008.11.3.133
Abstract
- PURPOSE
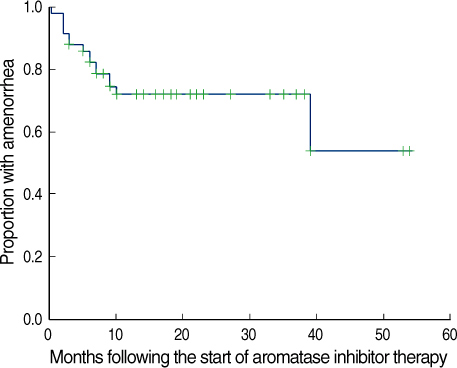
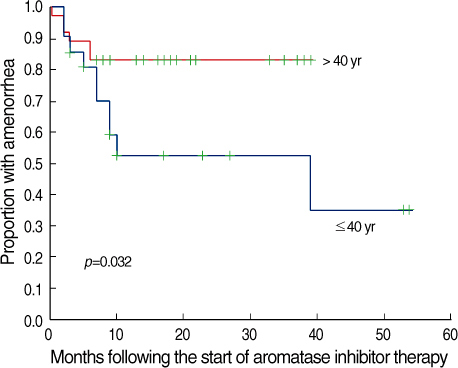
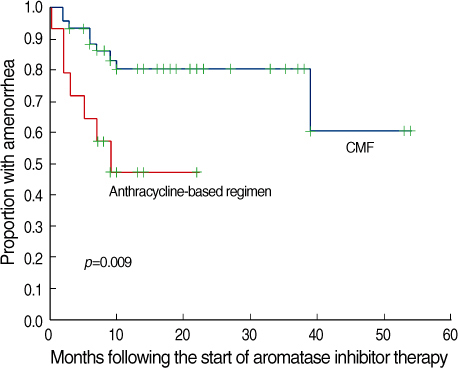
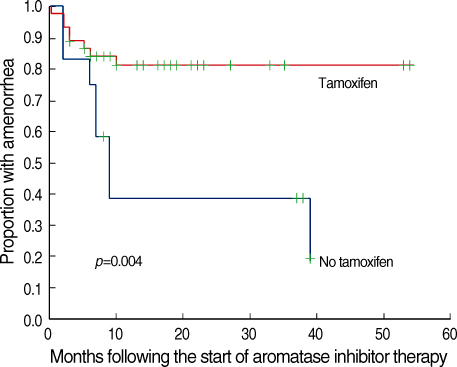
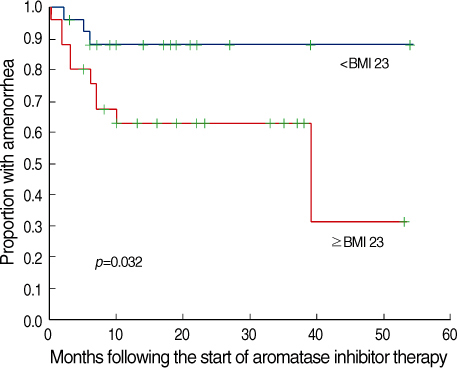
The role and safety of aromatase inhibitors (AIs) in young breast cancer patients with chemotherapy-induced amenorrhea (CIA) has not been established. The goal of this study was to investigate the safety and efficacy of AIs in young breast cancer patients with CIA. METHODS: From December 2000 to December 2006, 58 patients with hormone receptor positive breast cancer under the age of 45 were treated with AIs as adjuvant therapy. All patients had amenorrhea for more than three consecutive months at the time of treatment. We evaluated the rates of recovery of ovarian function during the treatment, and analyzed the association of the recovery of ovarian function with age, body mass index (BMI), chemotherapy regimen, radiation therapy, and the use of tamoxifen. RESULTS: Recovery of ovarian function was observed in 16 patients (27.6%). The univariate analysis showed that ovarian function was more frequently recovered in patients younger than 40 yr of age, treated with chemotherapy regimens other than Cyclophosphamide, Methotrexate, 5-Flurouracil (CMF), without a history of tamoxifen therapy, and with a higher BMI. The multivariate analysis confirmed that the type of chemotherapy (p=0.034) and the history of tamoxifen therapy (p=0.043) were independent factors significantly associated with the restoration of ovarian function. CONCLUSION: The results of this study suggest that AIs should be considered, with caution in young women with CIA; these agents may promote the unwanted recovery of ovarian function. Especially, in those patients who were not treated with CMF chemotherapy or tamoxifen, where the rates of recovery of ovarian function were higher.
Keyword
MeSH Terms
Figure
Reference
-
1. The Korean Breast Cancer Society. Nationwide Korea breast cancer data of 2002. J Breast Cancer. 2004. 7:72–83.2. Adami HO, Malker B, Holmberg L, Persson I, Stone B. The relation between survival and age at diagnosis in breast cancer. N Engl J Med. 1986. 315:559–563.
Article3. Chung M, Chang HR, Bland KI, Wanebo HJ. Younger women with breast carcinoma have a poorer prognosis than older women. Cancer. 1996. 77:97–103.
Article4. Kollias J, Elston CW, Ellis IO, Robertson JF, Blamey RW. Early-onset breast cancer-histopathological and prognostic considerations. Br J Cancer. 1997. 75:1318–1323.
Article5. Baum M, Buzdar A, Cuzick J, Forbes J, Houghton J, Howell A, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: results of the ATAC (Arimidex, Tamoxifen Alone or in Combination) trial efficacy and safety update analyses. Cancer. 2003. 98:1802–1810.
Article6. Breast International Group 1-98 Collaborative Group. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005. 353:2747–2757.7. Coombes RC, Hall E, Gibson LJ, Paridaens R, Jassem J, Delozier T, et al. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med. 2004. 350:1081–1092.
Article8. Boccardo F, Rubagotti A, Puntoni M, Guglielmini P, Amoroso D, Fini A, et al. Switching to anastrozole versus continued tamoxifen treatment of early breast cancer: preliminary results of the Italian tamoxifen anastrozole trial. J Clin Oncol. 2005. 23:5138–5147.
Article9. Jakesz R, Jonat W, Gnant M, Mittlboeck M, Greil R, Tausch C, et al. Switching of postmenopausal women with endocrine responsive early breast cancer to anastrozole after 2 years' adjuvant tamoxifen: Combined results of ABCSG trial 8 and ARNO 95 trial. Lancet. 2005. 366:455–462.
Article10. Goss PE, Ingle JN, Martino S, Robert NJ, Muss HB, Piccart MJ, et al. Randomized trial of letrozole following tamoxifen as extended adjuvant therapy in receptor-positive breast cancer: updated findings from NCIC CTG MA. 17. J Natl Cancer Inst. 2005. 97:1262–1271.
Article11. Winer EP, Hudis C, Burstein HJ, Wolff AC, Pritchard KI, Ingle JN, et al. American society of clinical oncology technology assessment on the use of aromatase inhibitors as adjuvant therapy for postmenopausal women with hormone receptor-positive breast cancer: status report 2004. J Clin Oncol. 2005. 23:619–629.
Article12. Smith IE, Dowsett M. Aromatase inhibitors in breast cancer. N Engl J Med. 2003. 348:2431–2442.
Article13. Mitwally MF, Casper RF. Use of an aromatase inhibitor for induction of ovulation in patients with an inadequate response to clomiphene citrate. Fertil Steril. 2001. 75:305–309.
Article14. Smith IE, Dowsett M, Yap YS, Walsh G, Lonning PE, Santen RJ, et al. Adjuvant aromatase inhibitors for early breast cancer after chemotherapy-induced amenorrhoea: caution and suggested guidelines. J Clin Oncol. 2006. 16:2444–2447.
Article15. Petrek JA, Naughton MJ, Case LD, Paskett ED, Naftalis EZ, Singletary SE, et al. Incidence, time course, and determinants of menstrual bleeding after breast cancer treatment: a prospective study. J Clin Oncol. 2006. 24:1045–1051.
Article16. Goodwin PJ, Ennis M, Pritchard KI, Trudeau M, Hood N. Risk of menopause during the first year after breast cancer diagnosis. J Clin Oncol. 1999. 17:2365–2370.
Article17. Tham YL, Sexton K, Weiss H, Elledge R, Friedman LC, Kramer R. The rates of chemotherapy-induced amenorrhea in patients treated with adjuvant doxorubicin and cyclophosphamide followed by a taxane. Am J Clin Oncol. 2007. 30:126–132.
Article18. Fornier MN, Modi S, Panageas KS, Norton L, Hudis C. Incidence of chemotherapy-induced, long-term amenorrhea in patients with breast carcinoma age 40 years and younger after adjuvant anthracycline and taxane. Cancer. 2005. 104:1575–1579.
Article19. Petrek JA, Naughton MJ, Case LD, Paskett ED, Naftalis EZ, Singletary SE, et al. Incidence, time course, and determinants of menstrual bleeding after breast cancer treatment: a prospective study. J Clin Oncol. 2006. 24:1045–1051.
Article20. Vanhuyse M, Fournier C, Bonneterre J. Chemotherapy-induced amenorrhea: influence on disease-free survival and overall survival in receptor-positive premenopausal early breast cancer patients. Ann Oncol. 2005. 16:1283–1288.
Article21. Metwally M, Li TC, Ledger WL. The impact of obesity on female reproductive function. Obesity Reviews. 2007. 8:515–523.
Article22. Zumoff B, Miller LK, Strain GW. Reversal of the hypogonadotropic hypogonadism of obese men by administration of the aromatase inhibitor testolactone. Metabolism. 2003. 52:1126–1128.
Article23. Mehta RR, Beattie CW, Das Gupta TK. Endocrine profile in breast cancer patients receiving chemotherapy. Breast Cancer Res Treat. 1992. 20:125–132.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Incidence of Chemotherapy-induced Amenorrhea and Recovery in Young (<45-year-old) Breast Cancer Patients
- The Examination of Ovarian Reserve in Premenopausal Patients with Hormone Receptor Positive Breast Cancer
- Role of Ovarian Function Suppression in Premenopausal Women with Early Breast Cancer
- Hormonal Changes during Extended Letrozole Treatment after Completion of 5 Years of Tamoxifen in Premenopausal Patients with Breast Cancer who Became Postmenopausal
- Personalized therapy for advanced breast cancer using molecular signatures