Korean J Physiol Pharmacol.
2013 Aug;17(4):315-320. 10.4196/kjpp.2013.17.4.315.
Radicicol Inhibits iNOS Expression in Cytokine-Stimulated Pancreatic Beta Cells
- Affiliations
-
- 1DNA Damage Response Network Center, Chosun University, Kwangju 501-709, Korea. yjjeon@chosun.ac.kr
- 2Department of Pharmacology, School of Medicine, Chosun University, Gwangju 501-759, Korea.
- 3Department of Anatomy, School of Medicine, Chosun University, Gwangju 501-759, Korea.
- 4Department of Anatomy, School of Medicine, Jeju National University, Jeju 690-756, Korea.
- KMID: 2285466
- DOI: http://doi.org/10.4196/kjpp.2013.17.4.315
Abstract
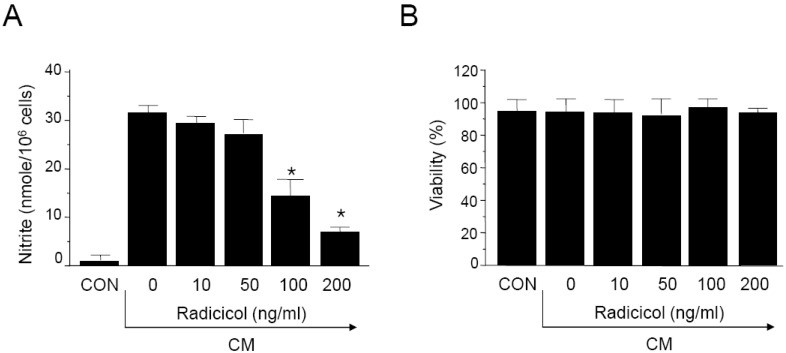
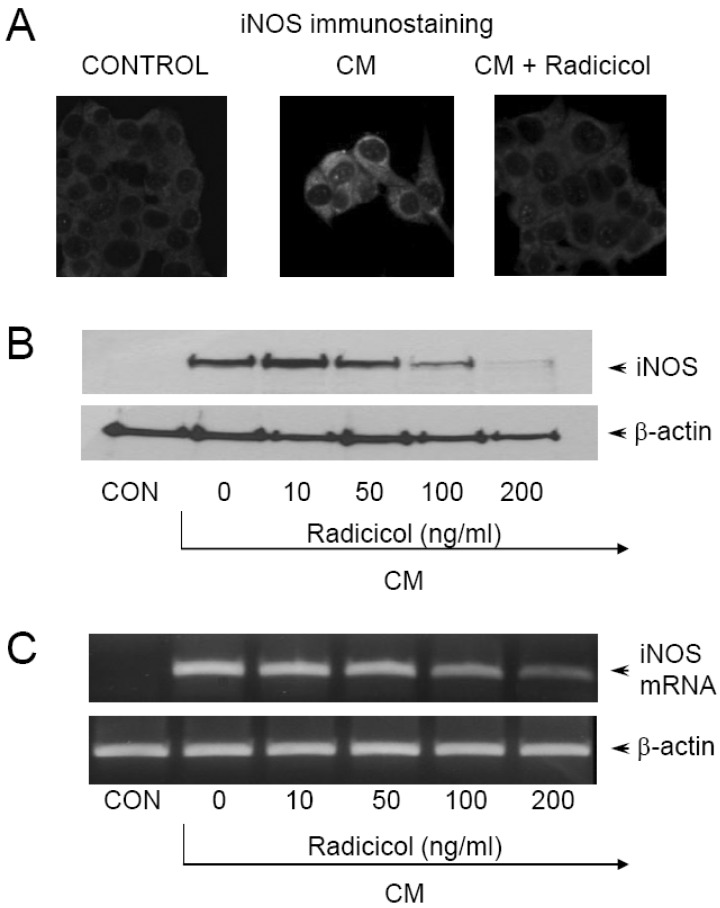
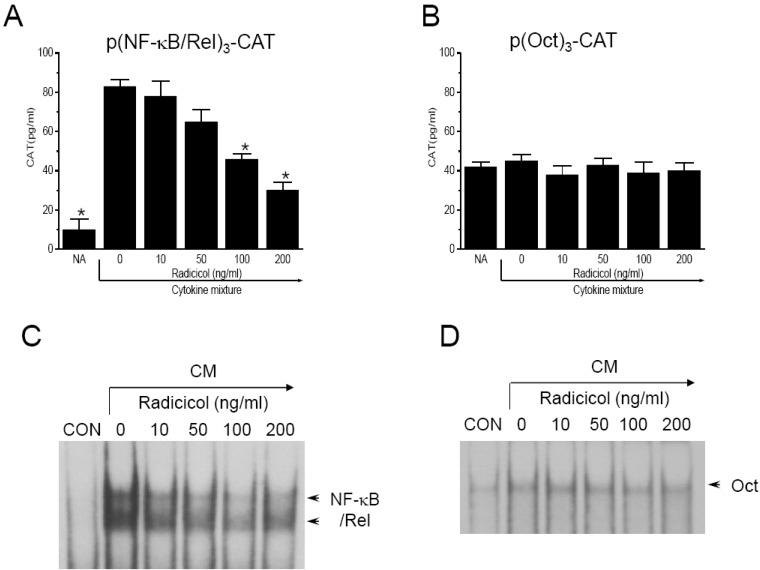
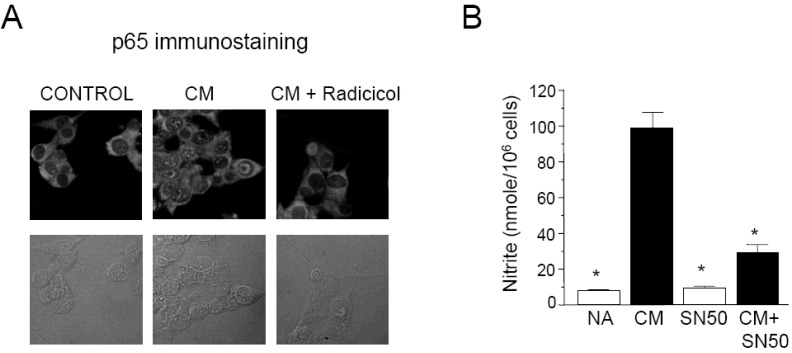
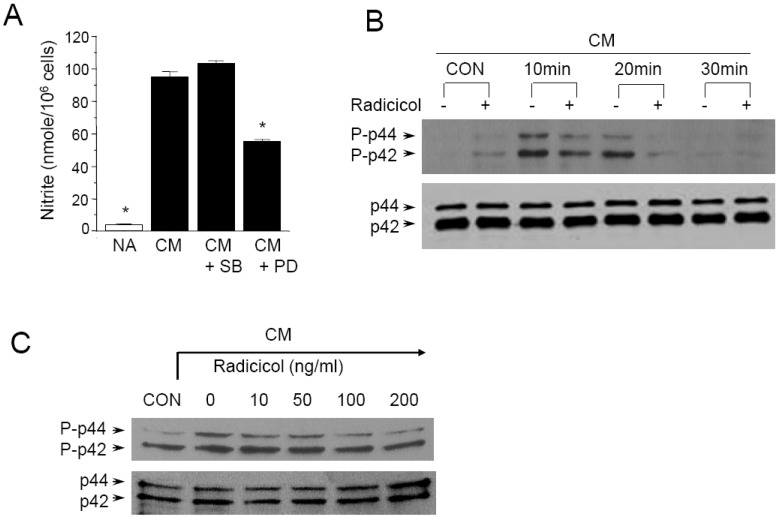
- Here, we show that radicicol, a fungal antibiotic, resulted in marked inhibition of inducible nitric oxide synthase (iNOS) transcription by the pancreatic beta cell line MIN6N8a in response to cytokine mixture (CM: TNF-alpha, IFN-gamma, and IL-1beta). Treatment of MIN6N8a cells with radicicol inhibited CM-stimulated activation of NF-kappaB/Rel, which plays a critical role in iNOS transcription, in a dose-related manner. Nitrite production in the presence of PD98059, a specific inhibitor of the extracellular signal-regulated protein kinase-1 and 2 (ERK1/2) pathway, was dramatically diminished, suggesting that the ERK1/2 pathway is involved in CM-induced iNOS expression. In contrast, SB203580, a specific inhibitor of p38, had no effect on nitrite generation. Collectively, this series of experiments indicates that radicicol inhibits iNOS gene expression by blocking ERK1/2 signaling. Due to the critical role that NO release plays in mediating destruction of pancreatic beta cells, the inhibitory effects of radicicol on iNOS expression suggest that radicicol may represent a useful anti-diabetic activity.
Keyword
- beta cells; ERK1/2; iNOS; NO
MeSH Terms
Figure
Cited by 2 articles
-
Silymarin Inhibits Morphological Changes in LPS-Stimulated Macrophages by Blocking NF-κB Pathway
Eun Jeong Kim, Min Young Lee, Young Jin Jeon
Korean J Physiol Pharmacol. 2015;19(3):211-218. doi: 10.4196/kjpp.2015.19.3.211.Inhibition of ERK1/2 by silymarin in mouse mesangial cells
Cha Kyung Youn, Sung Il Cho, Min Young Lee, Young Jin Jeon, Seog Ki Lee
Korean J Physiol Pharmacol. 2017;21(1):117-124. doi: 10.4196/kjpp.2017.21.1.117.
Reference
-
1. Cetkovic-Cvrlje M, Eizirik DL. TNF-alpha and IFN-gamma potentiate the deleterious effects of IL-1 beta on mouse pancreatic islets mainly via generation of nitric oxide. Cytokine. 1994; 6:399–406. PMID: 7948748.2. Darville MI, Eizirik DL. Regulation by cytokines of the inducible nitric oxide synthase promoter in insulin-producing cells. Diabetologia. 1998; 41:1101–1108. PMID: 9754830.
Article3. Eizirik DL, Sandler S, Welsh N, Cetkovic-Cvrlje M, Nieman A, Geller DA, Pipeleers DG, Bendtzen K, Hellerström C. Cytokines suppress human islet function irrespective of their effects on nitric oxide generation. J Clin Invest. 1994; 93:1968–1974. PMID: 7514190.
Article4. Yamada K, Otabe S, Inada C, Takane N, Nonaka K. Nitric oxide and nitric oxide synthase mRNA induction in mouse islet cells by interferon-gamma plus tumor necrosis factor-alpha. Biochem Biophys Res Commun. 1993; 197:22–27. PMID: 7504484.5. Takamura T, Kato I, Kimura N, Nakazawa T, Yonekura H, Takasawa S, Okamoto H. Transgenic mice overexpressing type 2 nitric-oxide synthase in pancreatic beta cells develop insulin-dependent diabetes without insulitis. J Biol Chem. 1998; 273:2493–2496. PMID: 9446547.6. Flodström M, Tyrberg B, Eizirik DL, Sandler S. Reduced sensitivity of inducible nitric oxide synthase-deficient mice to multiple low-dose streptozotocin-induced diabetes. Diabetes. 1999; 48:706–713. PMID: 10102685.
Article7. Fukuhara Y, Turner RJ. Cation dependence of renal outer cortical brush border membrane L-glutamate transport. Am J Physiol. 1985; 248:F869–F875. PMID: 2860810.
Article8. Oikawa T, Ito H, Ashino H, Toi M, Tominaga T, Morita I, Murota S. Radicicol, a microbial cell differentiation modulator, inhibits in vivo angiogenesis. Eur J Pharmacol. 1993; 241:221–227. PMID: 7694864.9. Kwon HJ, Yoshida M, Abe K, Horinouchi S, Beppu T. Radicicol, an agent inducing the reversal of transformed phenotypes of src-transformed fibroblasts. Biosci Biotechnol Biochem. 1992; 56:538–539. PMID: 1368339.10. Kwon HJ, Yoshida M, Fukui Y, Horinouchi S, Beppu T. Potent and specific inhibition of p60v-src protein kinase both in vivo and in vitro by radicicol. Cancer Res. 1992; 52:6926–6930. PMID: 1458481.11. Soga S, Kozawa T, Narumi H, Akinaga S, Irie K, Matsumoto K, Sharma SV, Nakano H, Mizukami T, Hara M. Radicicol leads to selective depletion of Raf kinase and disrupts K-Ras-activated aberrant signaling pathway. J Biol Chem. 1998; 273:822–828. PMID: 9422737.
Article12. Chanmugam P, Feng L, Liou S, Jang BC, Boudreau M, Yu G, Lee JH, Kwon HJ, Beppu T, Yoshida M, Xia Y, Wilson CB, Hwand D. Radicicol, a protein tyrosine kinase inhibitor, suppresses the expression of mitogen-inducible cyclooxygenase in macrophages stimulated with lipopolysaccharide and in experimental glomerulonephritis. J Biol Chem. 1995; 270:5418–5426. PMID: 7890656.
Article13. Jeon YJ, Kim YK, Lee M, Park SM, Han SB, Kim HM. Radicicol suppresses expression of inducible nitric-oxide synthase by blocking p38 kinase and nuclear factor-kappaB/Rel in lipopolysaccharide-stimulated macrophages. J Pharmacol Exp Ther. 2000; 294:548–554. PMID: 10900231.14. Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem. 1982; 126:131–138. PMID: 7181105.15. Huong PT, Lee MY, Lee KY, Chang IY, Lee SK, Yoon SP, Lee DC, Jeon YJ. Synergistic induction of iNOS by IFN-γ and glycoprotein isolated from dioscorea batatas. Korean J Physiol Pharmacol. 2012; 16:431–436. PMID: 23269906.16. Jeon YJ, Yang KH, Pulaski JT, Kaminski NE. Attenuation of inducible nitric oxide synthase gene expression by delta 9-tetrahydrocannabinol is mediated through the inhibition of nuclear factor-kappa B/Rel activation. Mol Pharmacol. 1996; 50:334–341. PMID: 8700141.17. Li MH, Kothandan G, Cho SJ, Huong PT, Nan YH, Lee KY, Shin SY, Yea SS, Jeon YJ. Magnolol inhibits LPS-induced NF-κB/Rel activation by blocking p38 kinase in murine macrophages. Korean J Physiol Pharmacol. 2010; 14:353–358. PMID: 21311674.
Article18. Xie QW, Whisnant R, Nathan C. Promoter of the mouse gene encoding calcium-independent nitric oxide synthase confers inducibility by interferon gamma and bacterial lipopolysaccharide. J Exp Med. 1993; 177:1779–1784. PMID: 7684434.
Article19. Jeon YJ, Han SH, Lee YW, Yea SS, Yang KH. Inhibition of NF-kappa B/Rel nuclear translocation by dexamethasone: mechanism for the inhibition of iNOS gene expression. Biochem Mol Biol Int. 1998; 45:435–441. PMID: 9679644.20. Dunnett M. A multiple comparison procedure for comparing several treatments with a control. J Am Statistics Assoc. 1955; 50:1096–1121.
Article21. Xie QW, Kashiwabara Y, Nathan C. Role of transcription factor NF-kappa B/Rel in induction of nitric oxide synthase. J Biol Chem. 1994; 269:4705–4708. PMID: 7508926.
Article22. Lin YZ, Yao SY, Veach RA, Torgerson TR, Hawiger J. Inhibition of nuclear translocation of transcription factor NF-kappa B by a synthetic peptide containing a cell membrane-permeable motif and nuclear localization sequence. J Biol Chem. 1995; 270:14255–14258. PMID: 7782278.23. Heimberg H, Heremans Y, Jobin C, Leemans R, Cardozo AK, Darville M, Eizirik DL. Inhibition of cytokine-induced NF-kappaB activation by adenovirus-mediated expression of a NF-kappaB super-repressor prevents beta-cell apoptosis. Diabetes. 2001; 50:2219–2224. PMID: 11574401.24. Baker MS, Chen X, Cao XC, Kaufman DB. Expression of a dominant negative inhibitor of NF-kappaB protects MIN6 beta-cells from cytokine-induced apoptosis. J Surg Res. 2001; 97:117–122. PMID: 11341786.25. Eldor R, Yeffet A, Baum K, Doviner V, Amar D, Ben-Neriah Y, Christofori G, Peled A, Carel JC, Boitard C, Klein T, Serup P, Eizirik DL, Melloul D. Conditional and specific NF-kappaB blockade protects pancreatic beta cells from diabetogenic agents. Proc Natl Acad Sci USA. 2006; 103:5072–5077. PMID: 16551748.26. Schmitz ML, Baeuerle PA. The p65 subunit is responsible for the strong transcription activating potential of NF-kappa B. EMBO J. 1991; 10:3805–3817. PMID: 1935902.
Article27. Roe SM, Prodromou C, O'Brien R, Ladbury JE, Piper PW, Pearl LH. Structural basis for inhibition of the Hsp90 molecular chaperone by the antitumor antibiotics radicicol and geldanamycin. J Med Chem. 1999; 42:260–266. PMID: 9925731.
Article28. Larsen L, Strling J, Darville M, Eizirik DL, Bonny C, Billestrup N, Mandrup-Poulsen T. Extracellular signal-regulated kinase is essential for interleukin-1-induced and nuclear factor kappaB-mediated gene expression in insulin-producing INS-1E cells. Diabetologia. 2005; 48:2582–2590. PMID: 16283237.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Silymarin Inhibits Cytokine-Stimulated Pancreatic Beta Cells by Blocking the ERK1/2 Pathway
- The stimulatory effect of IL-1beta on the insulin secretion of rat pancreatic islet is not related with iNOS pathway
- Docosahexaenoic Acid Inhibits Cytokine Expression by Reducing Reactive Oxygen Species in Pancreatic Stellate Cells
- Inducible Nitric Oxide Synthase (iNOS) Expression in the Hypoxic Injury to Pancreatic Beta (MIN6) Cells
- Androgen Hormone Inhibits Expression of iNOS and COX-2 Protein in Rat Vascular Smooth Muscle Cell