Korean J Physiol Pharmacol.
2009 Dec;13(6):483-489. 10.4196/kjpp.2009.13.6.483.
Knockdown of RCAN1.4 Increases Susceptibility to FAS-mediated and DNA-damage-induced Apoptosis by Upregulation of p53 Expression
- Affiliations
-
- 1Department of Pharmacology, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea. haechee@catholic.ac.kr
- 2Department of Biochemistry, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea.
- 3Cell Death Disease Research Center, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea.
- KMID: 2285385
- DOI: http://doi.org/10.4196/kjpp.2009.13.6.483
Abstract
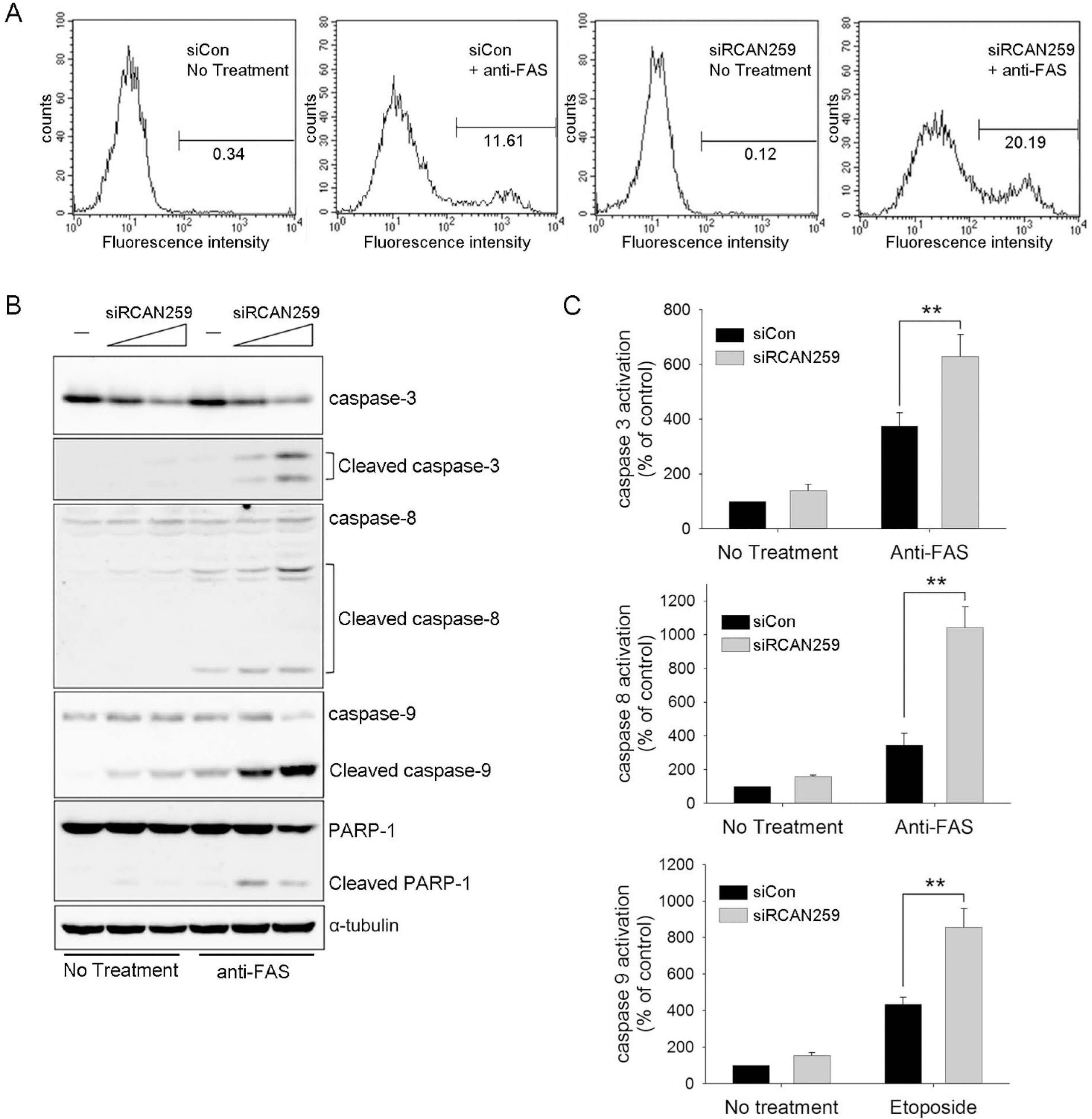
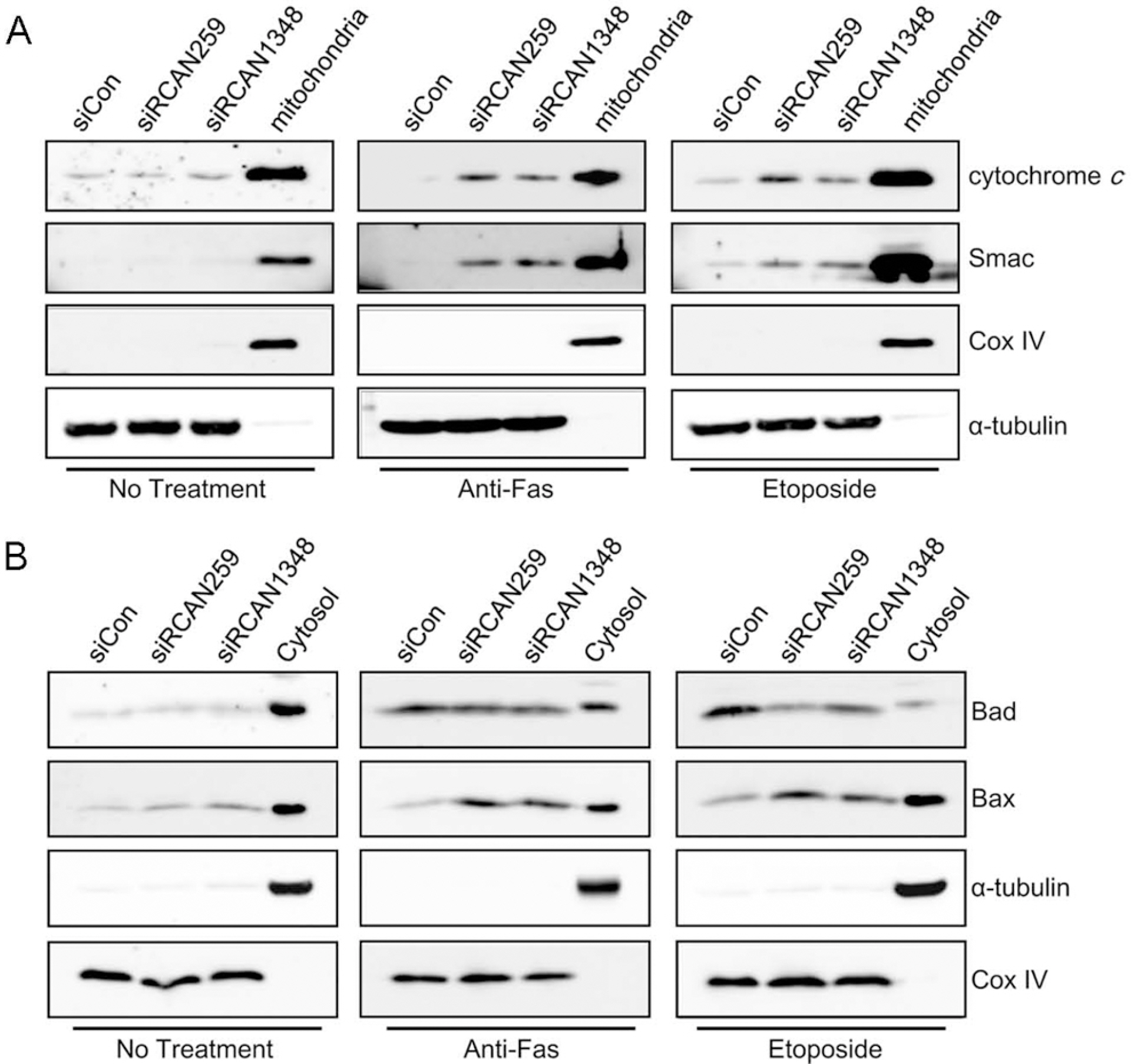
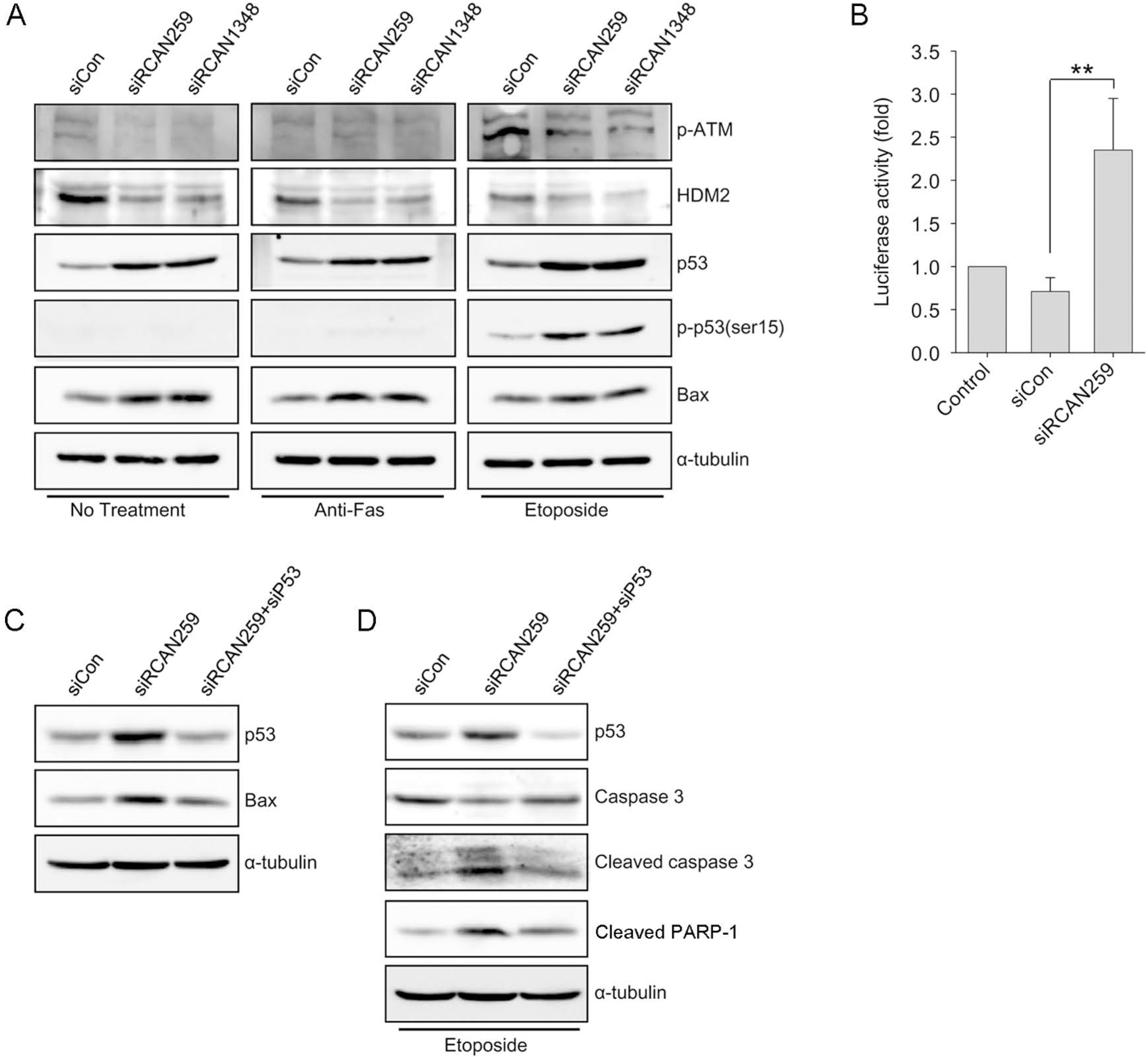
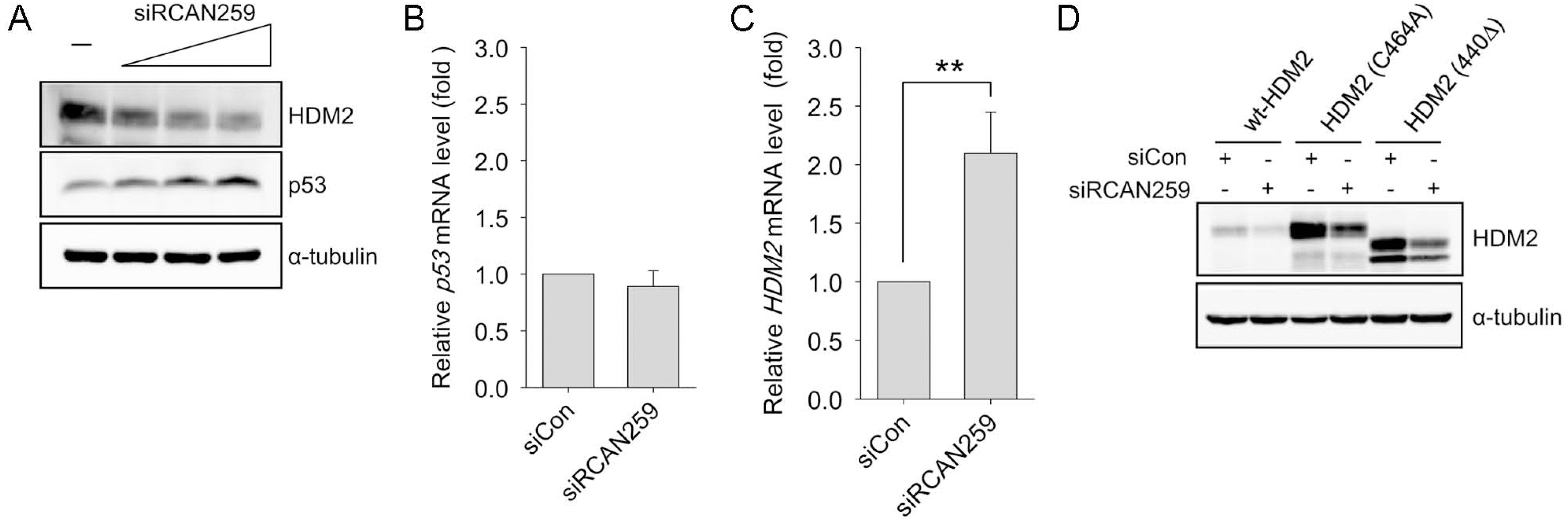
- Despite the potential importance of the human regulator of calcineurin 1 (RCAN-1) gene in the modulation of cell survival under stress, little is known about its role in death-inducing signal pathways. In this study, we addressed the effects of RCAN1.4 knockdown on cellular susceptibility to apoptosis and the activation of death pathway proteins. Transfection of siRNAs against RCAN1.4 resulted in enhanced Fas- and etoposide-induced apoptosis, which was associated with increased expression and translocation of Bax to mitochondria. Our results suggest that enhanced expression and activation of p53 was responsible for the upregulation of Bax and the increased sensitivity to apoptosis, which could be reversed by p53 knockdown. To explain the observed upregulation of p53, we propose a downregulation of the ubiquitin ligase HDM2, probably translationally. These findings show the importance of appropriate RCAN1.4 expression in the modulation of cell survival and reveal a link between RCAN1.4 and p53.
MeSH Terms
Figure
Cited by 2 articles
-
Melatonin Induces Apoptotic Cell Death via p53 in LNCaP Cells
Chi Hyun Kim, Yeong-Min Yoo
Korean J Physiol Pharmacol. 2010;14(6):365-369. doi: 10.4196/kjpp.2010.14.6.365.Regulator of Calcineurin 1 Isoform 4 (RCAN1.4) Is Overexpressed in the Glomeruli of Diabetic Mice
Chorong Jang, Ji Hee Lim, Cheol Whee Park, Young-Jin Cho
Korean J Physiol Pharmacol. 2011;15(5):299-305. doi: 10.4196/kjpp.2011.15.5.299.
Reference
-
References
Banin S., Moyal L., Shieh S., Taya Y., Anderson CW., Chessa L., Smorodinsky NI., Prives C., Reiss Y., Shiloh Y., Ziv Y. Enhanced phosphorylation of p53 by ATM in response to DNA damage. Science. 281:1674–1677. 1998.
ArticleBarak Y., Juven T., Haffner R., Oren M. Mdm2 expression is induced by wild type p53 activity. EMBO J. 12:461–468. 1993.
ArticleCanman CE., Lim DS., Cimprich KA., Taya Y., Tamai K., Sakaguchi K., Appella E., Kastan MB., Siliciano JD. Activation of the ATM kinase by ionizing radiation and phosphorylation of p53. Science. 281:1677–1679. 1998.
ArticleDe Benedetti A., Graff JR. eIF-4E expression and its role in malignancies and metastases. Oncogene. 23:3189–3199. 2004.
ArticleErmak G., Harris CD., Davies KJ. The DSCR1 (Adapt78) isoform 1 protein calcipressin 1 inhibits calcineurin and protects against acute calcium-mediated stress damage, including transient oxidative stress. FASEB J. 16:814–824. 2002.Ermak G., Morgan TE., Davies KJ. Chronic overexpression of the calcineurin inhibitory gene DSCR1 (Adapt78) is associated with Alzheimer's disease. J Biol Chem. 276:38787–38794. 2001.
ArticleFang S., Jensen JP., Ludwig RL., Vousden KH., Weissman AM. Mdm2 is a RING finger-dependent ubiquitin protein ligase for itself and p53. J Biol Chem. 275:8945–8951. 2000.
ArticleFuentes JJ., Genesca L., Kingsbury TJ., Cunningham KW., Perez-Riba M., Estivill X., de la Luna S. DSCR1, overexpressed in Down syndrome, is an inhibitor of calcineurin-mediated signaling pathways. Hum Mol Genet. 9:1681–1690. 2000.
ArticleFuentes JJ., Pritchard MA., Estivill X. Genomic organization, alternative splicing, and expression patterns of the DSCR1 (Down syndrome candidate region 1) gene. Genomics. 44:358–361. 1997.Fuentes JJ., Pritchard MA., Planas AM., Bosch A., Ferrer I., Estivill X. A new human gene from the Down syndrome critical region encodes a proline-rich protein highly expressed in fetal brain and heart. Hum Mol Genet. 4:1935–1944. 1995.
ArticleHarris CD., Ermak G., Davies KJ. Multiple roles of the DSCR1 (Adapt78 or RCAN1) gene and its protein product calcipressin 1 (or RCAN1) in disease. Cell Mol Life Sci. 62:2477–2486. 2005.
ArticleHead E., Lott IT., Patterson D., Doran E., Haier RJ. Possible compensatory events in adult Down syndrome brain prior to the development of Alzheimer disease neuropathology: targets for nonpharmacological intervention. J Alzheimers Dis. 11:61–76. 2007.
ArticleHsu YT., Wolter KG., Youle RJ. Cytosol-to-membrane redistribution of Bax and Bcl-X(L) during apoptosis. Proc Natl Acad Sci U S A. 94:3668–3672. 1997.
ArticleKubbutat MH., Jones SN., Vousden KH. Regulation of p53 stability by Mdm2. Nature. 387:299–303. 1997.
ArticleKubbutat MH., Ludwig RL., Ashcroft M., Vousden KH. Regulation of Mdm2-directed degradation by the C terminus of p53. Mol Cell Biol. 18:5690–5698. 1998.
ArticleLee HJ., Kim YS., Sato Y., Cho YJ. RCAN1–4 knockdown attenuates cell growth through the inhibition of Ras signaling. FEBS Lett. 583:2557–2564. 2009.
ArticleMiyashita T., Krajewski S., Krajewska M., Wang HG., Lin HK., Liebermann DA., Hoffman B., Reed JC. Tumor suppressor p53 is a regulator of bcl-2 and bax gene expression in vitro and in vivo. Oncogene. 9:1799–1805. 1994.Porta S., Serra SA., Huch M., Valverde MA., Llorens F., Estivill X., Arbones ML., Marti E. RCAN1 (DSCR1) increases neuronal susceptibility to oxidative stress: a potential pathogenic process in neurodegeneration. Hum Mol Genet. 16:1039–1050. 2007.
ArticlePrives C. Signaling to p53: breaking the MDM2-p53 circuit. Cell. 95:5–8. 1998.Rothermel B., Vega RB., Yang J., Wu H., Bassel-Duby R., Williams RS. A protein encoded within the Down syndrome critical region is enriched in striated muscles and inhibits calcineurin signaling. J Biol Chem. 275:8719–8725. 2000.
ArticleRyeom S., Baek KH., Rioth MJ., Lynch RC., Zaslavsky A., Birsner A., Yoon SS., McKeon F. Targeted deletion of the calcineurin inhibitor DSCR1 suppresses tumor growth. Cancer Cell. 13:420–431. 2008.
ArticleRyeom S., Greenwald RJ., Sharpe AH., McKeon F. The threshold pattern of calcineurin-dependent gene expression is altered by loss of the endogenous inhibitor calcipressin. Nat Immunol. 4:874–881. 2003.
ArticleSanna B., Brandt EB., Kaiser RA., Pfluger P., Witt SA., Kimball TR., van Rooij E., De Windt LJ., Rothenberg ME., Tschop MH., Benoit SC., Molkentin JD. Modulatory calcineurin-interacting proteins 1 and 2 function as calcineurin facilitators in vivo. Proc Natl Acad Sci U S A. 103:7327–7332. 2006.
ArticleVousden KH., Lu X. Live or let die: the cell's response to p53. Nat Rev Cancer. 2:594–604. 2002.
ArticleWolter KG., Hsu YT., Smith CL., Nechushtan A., Xi XG., Youle RJ. Movement of Bax from the cytosol to mitochondria during apoptosis. J Cell Biol. 139:1281–1292. 1997.
ArticleWu X., Bayle JH., Olson D., Levine AJ. The p53-mdm-2 autoregulatory feedback loop. Genes Dev. 7:1126–1132. 1993.
ArticleZhao P., Xiao X., Kim AS., Leite MF., Xu J., Zhu X., Ren J., Li J. c-Jun inhibits thapsigargin-induced ER stress through up-regulation of DSCR1/Adapt78. Exp Biol Med (Maywood). 233:1289–1300. 2008.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Fas and Fas ligand on Ultraviolet B-irradiated Skin
- p53 and DNA-dependent protein kinase catalytic subunit independently function in regulating actin damage-induced tetraploid G1 arrest
- The effect of cyclophosphamide on Fas-mediated apoptosis
- p53 Suppressor Gene Ove rex p ression in the Cornea after Mechanical-and Chemical-induced Epithelial Injury
- Upregulation of Reactive Oxygen Species and Resistance to Fas-mediated Apoptosis in Cervical Cancer Cell Lines Transfected with the IL-18 Receptor