Korean J Physiol Pharmacol.
2009 Dec;13(6):475-482. 10.4196/kjpp.2009.13.6.475.
Rifampicin Inhibits the LPS-induced Expression of Toll-like Receptor 2 via the Suppression of NF-kappaB DNA-binding Activity in RAW 264.7 Cells
- Affiliations
-
- 1Institute of Hansen's Disease, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea. sblee@catholic.ac.kr
- 2Department of Pathology, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea.
- KMID: 2285384
- DOI: http://doi.org/10.4196/kjpp.2009.13.6.475
Abstract
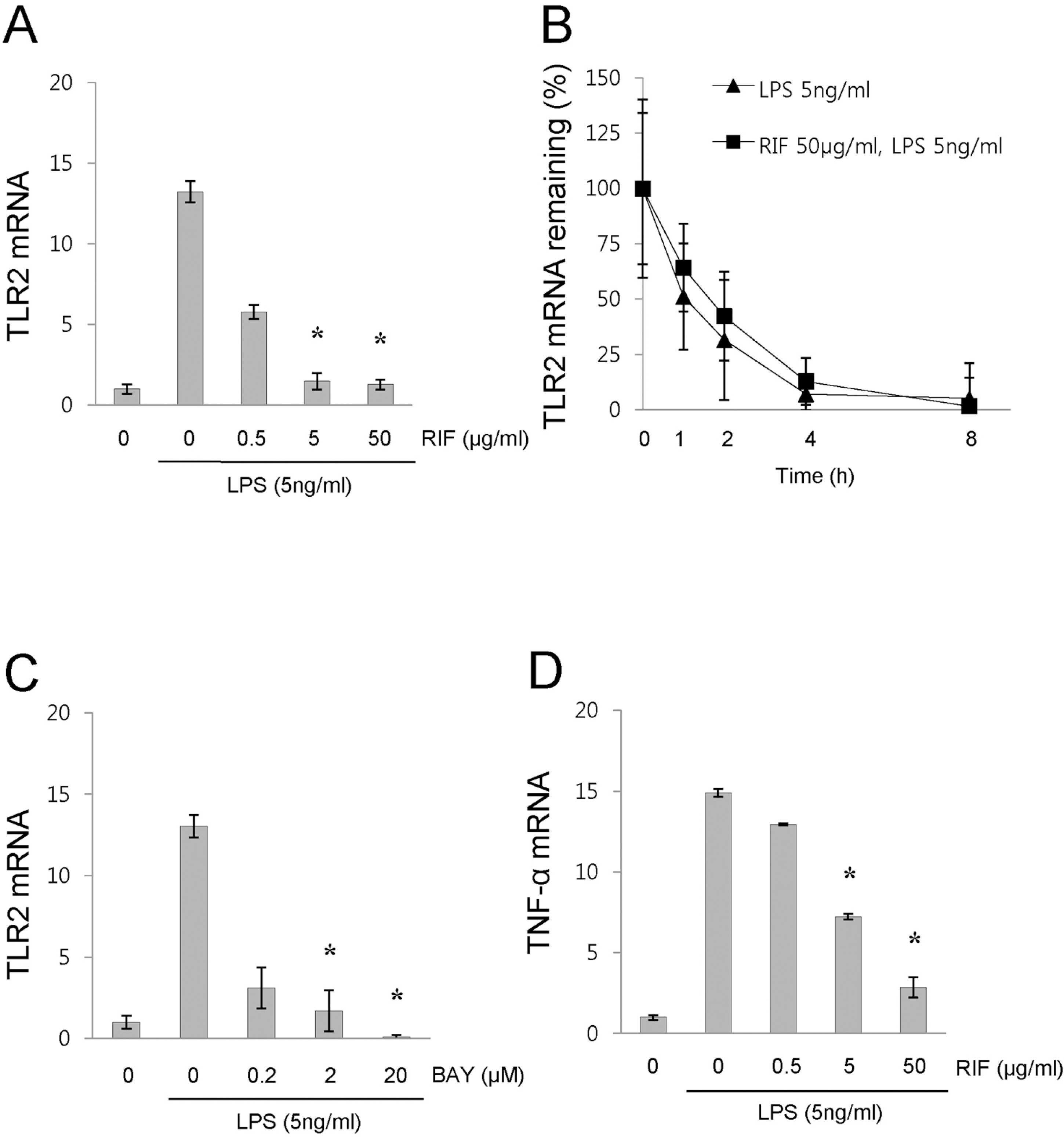
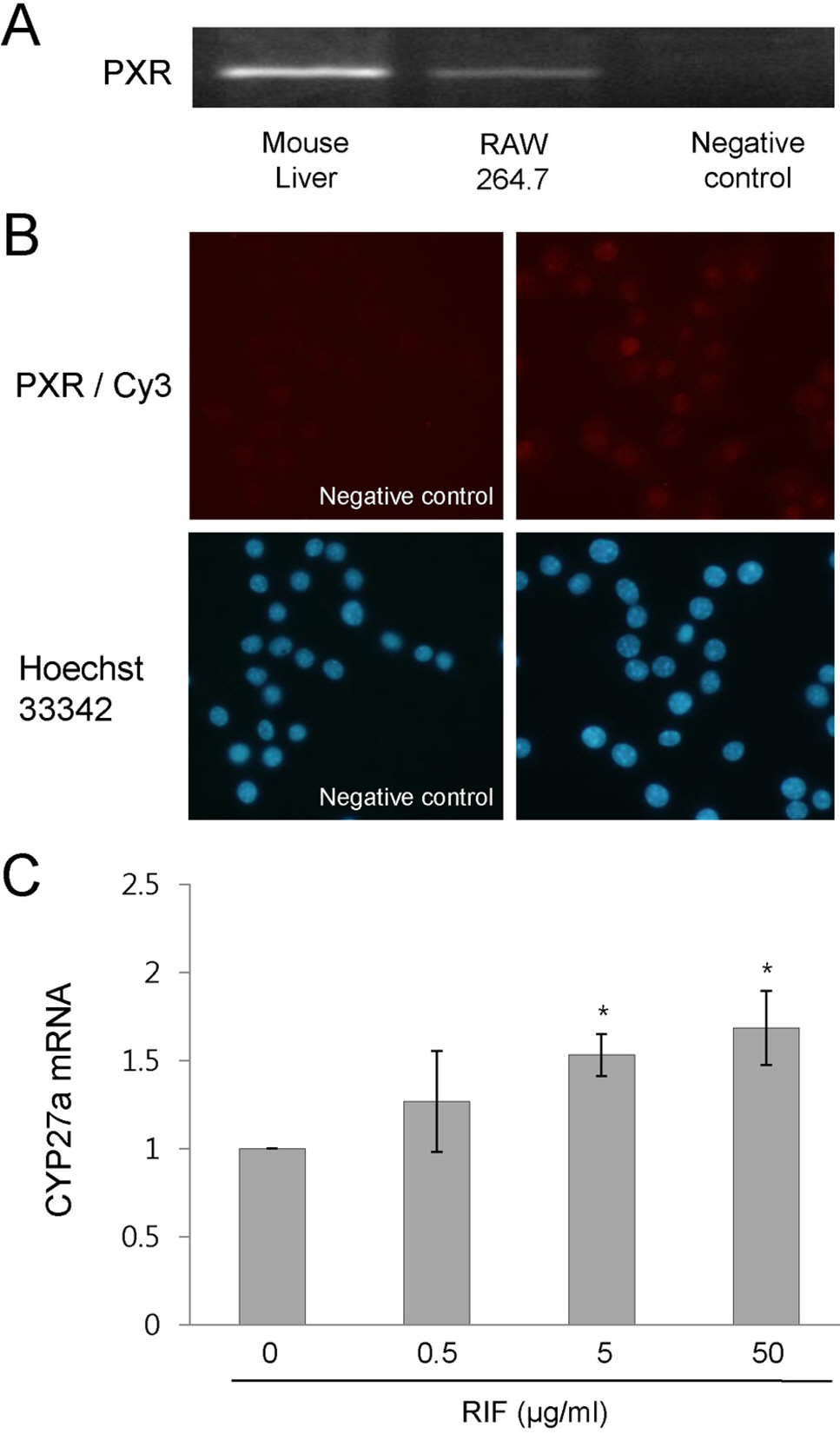
- Rifampicin is a macrocyclic antibiotic which is used extensively for treatment against Mycobacterium tuberculosis and other mycobacterial infections. Recently, a number of studies have focused on the immune-regulatory effects of rifampicin. Therefore, we hypothesized that rifampicin may influence the TLR2 expression in LPS-activated RAW 264.7 cells. In this study, we determined that rifampicin suppresses LPS-induced TLR2 mRNA expression. The down-regulation of TLR2 expression coincided with decreased production of TNF-alpha. Since NF-kappaB is a major transcription factor that regulates genes for TLR2 and TNF-alpha, we examined the effect of rifampicin on the LPS-induced NF-kappaB activation. Rifampicin inhibited NF-kappaB DNA-binding activity in LPS-activated RAW 264.7 cells, while it did not affect IKKalpha/beta activity. However, rifampicin slightly inhibited the nuclear translocation of NF-kappaB p65. In addition, rifampicin increased physical interaction between pregnane X receptor, a receptor for rifampicin, and NF-kappaB p65, suggesting pregnane X receptor interferes with NF-kappaB binding to DNA. Taken together, our results demonstrate that rifampicin inhibits LPS-induced TLR2 expression, at least in part, via the suppression of NF-kappaB DNA-binding activity in RAW 264.7 cells. Thus, the present results suggest that the rifampicin-mediated inhibition of TLR2 via the suppression of NF-kappaB DNA-binding activity may be a novel mechanism of the immune-suppressive effects of rifampicin.
Keyword
MeSH Terms
-
DNA
Down-Regulation
Mycobacterium tuberculosis
NF-kappa B
Receptors, Steroid
Rifampin
RNA, Messenger
Toll-Like Receptor 2
Toll-Like Receptors
Transcription Factors
Tumor Necrosis Factor-alpha
DNA
NF-kappa B
RNA, Messenger
Receptors, Steroid
Rifampin
Toll-Like Receptor 2
Toll-Like Receptors
Transcription Factors
Tumor Necrosis Factor-alpha
Figure
Reference
-
References
Aravalli RN., Hu S., Rowen TN., Palmquist JM., Lokensgard JR. Cutting edge: TLR2-mediated proinflammatory cytokine and chemokine production by microglial cells in response to herpes simplex virus. J Immunol. 175:4189–4193. 2005.
ArticleBaldwin AS Jr. The NF-kappa B and I kappa B proteins: new discoveries and insights. Annu Rev Immunol. 14:649–683. 1996.Bellahsene A., Forsgren A. Effect of rifampin on the immune response in mice. Infect Immunl. 27:15–20. 1980.Ghosh S., May MJ., Kopp EB. NF-kappa B and Rel proteins: evolutionarily conserved mediators of immune responses. Annu Rev Immunol. 16:225–260. 1998.Gu X., Ke S., Liu D., Sheng T., Thomas PE., Rabson AB., Gallo MA., Xie W., Tian Y. Role of NF-kappaB in regulation of PXR-mediated gene expression: a mechanism for the suppression of cytochrome P-450 3A4 by proinflammatory agents. J Biol Chem. 281:17882–17889. 2006.Haehnel V., Schwarzfischer L., Fenton MJ., Rehli M. Transcriptional regulation of the human toll-like receptor 2 gene in monocytes and macrophages. J Immunol. 168:5629–5637. 2002.Jeon YJ., Han SH., Lee YW., Lee M., Yang KH., Kim HM. Dexamethasone inhibits IL-1 beta gene expression in LPS-stimulated RAW 264.7 cells by blocking NF-kappa B/Rel and AP-1 activation. Immunopharmacology. 48:173–183. 2000.Kalkhoven E., Wissink S., van der Saag PT., van der Burg B. Negative interaction between the RelA(p65) subunit of NF-kappaB and the progesterone receptor. J Biol Chem. 271:6217–6224. 1996.Lehmann JM., McKee DD., Watson MA., Willson TM., Moore JT., Kliewer SA. The human orphan nuclear receptor PXR is activated by compounds that regulate CYP3A4 gene expression and cause drug interactions. J Clin Invest. 102:1016–1023. 1998.
ArticleLi T., Chen W., Chiang JY. PXR induces CYP27A1 and regulates cholesterol metabolism in the intestine. J Lipid Res. 48:373–384. 2007.
ArticleLi T., Chiang JY. Mechanism of rifampicin and pregnane X receptor inhibition of human cholesterol 7 alpha-hydroxylase gene transcription. Am J Physiol Gastrointest Liver Physiol. 288:G74–G84. 2005.Mlambo G., Sigola LB. Rifampicin and dexamethasone have similar effects on macrophage phagocytosis of zymosan, but differ in their effects on nitrite and TNF-alpha production. Int Immunopharmacol. 3:513–522. 2003.Moreau A., Vilarem MJ., Maurel P., Pascussi JM. Xenoreceptors CAR and PXR activation and consequences on lipid metabolism, glucose homeostasis, and inflammatory response. Mol Pharm. 5:35–41. 2008.
ArticleMun HS., Aosai F., Norose K., Chen M., Piao LX., Takeuchi O., Akira S., Ishikura H., Yano A. TLR2 as an essential molecule for protective immunity against Toxoplasma gondii infection. Int Immunol. 15:1081–1087. 2003.
ArticleNakamura K., Moore R., Negishi M., Sueyoshi T. Nuclear pregnane X receptor cross-talk with FoxA2 to mediate drug-induced regulation of lipid metabolism in fasting mouse liver. J Biol Chem. 282:9768–9776. 2007.
ArticlePahlevan AA., Wright DJ., Bradley L., Smith C., Foxwell BM. Potential of rifamides to inhibit TNF-induced NF-kappaB activation. J Antimicrob Chemother. 49:531–534. 2002.
ArticlePalvimo JJ., Reinikainen P., Ikonen T., Kallio PJ., Moilanen A., Janne OA. Mutual transcriptional interference between RelA and androgen receptor. J Biol Chem. 271:24151–24156. 1996.
ArticleRay A., Prefontaine KE. Physical association and functional antagonism between the p65 subunit of transcription factor NF-kappa B and the glucocorticoid receptor. Proc Natl Acad Sci U S A. 91:752–756. 1994.
ArticleRay A., Prefontaine KE., Ray P. Down-modulation of interleukin-6 gene expression by 17 beta-estradiol in the absence of high affinity DNA binding by the estrogen receptor. J Biol Chem. 269:12940–12946. 1994.
ArticleShah YM., Ma X., Morimura K., Kim I., Gonzalez FJ. Pregnane X receptor activation ameliorates DSS-induced inflammatory bowel disease via inhibition of NF-kappaB target gene expression. Am J Physiol Gastrointest Liver Physiol. 292:G1114–G1122. 2007.Stein B., Yang MX. Repression of the interleukin-6 promoter by estrogen receptor is mediated by NF-kappa B and C/EBP beta. Mol Cell Biol. 15:4971–4979. 1995.
ArticleTsankov N., Angelova I. Rifampin in dermatology. Clin Dermatol. 21:50–55. 2003.
ArticleXu DX., Wei W., Sun MF., Wu CY., Wang JP., Wei LZ., Zhou CF. Kupffer cells and reactive oxygen species partially mediate lipopolysaccharide-induced downregulation of nuclear receptor pregnane × receptor and its target gene CYP3a in mouse liver. Free Radic Biol Med. 37:10–22. 2004.Yuhas Y., Berent E., Ovadiah H., Azoulay I., Ashkenazi S. Rifampin augments cytokine-induced nitric oxide production in human alveolar epithelial cells. Antimicrob Agents Chemother. 50:396–398. 2006.
ArticleZhang H., LeCulyse E., Liu L., Hu M., Matoney L., Zhu W., Yan B. Rat pregnane X receptor: molecular cloning, tissue distribution, and xenobiotic regulation. Arch Biochem Biophys. 368:14–22. 1999.
ArticleZhou C., Tabb MM., Nelson EL., Grun F., Verma S., Sadatrafiei A., Lin M., Mallick S., Forman BM., Thummel KE., Blumberg B. Mutual repression between steroid and xenobiotic receptor and NF-kappaB signaling pathways links xenobiotic metabolism and inflammation. J Clin Invest. 116:2280–2289. 2006.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diclofenac inhibits IFN-gamma plus lipopolysaccharide-induced iNOS gene expression via suppression of NF-kappaB activation in RAW 264.7 macrophages
- Aromadendrin Inhibits Lipopolysaccharide-Induced Nuclear Translocation of NF-kappaB and Phosphorylation of JNK in RAW 264.7 Macrophage Cells
- Curcumin suppresses the production of interleukin-6 in Prevotella intermedia lipopolysaccharide-activated RAW 264.7 cells
- Role of NF- kappaB binding sites in the regulation of inducible nitric oxide synthase by tyrosine kinase
- Differential regulation of inducible nitric oxide synthase and cyclooxygenase-2 expression by superoxide dismutase in lipopolysaccharide stimulated RAW 264.7 cells