J Korean Surg Soc.
2013 Dec;85(6):296-301. 10.4174/jkss.2013.85.6.296.
Inhibition of intimal hyperplasia by local perivascular application of rapamycin and imatinib mesilate after carotid balloon injury
- Affiliations
-
- 1Department of Surgery, Seoul National University College of Medicine, Seoul, Korea. skminmd@snuh.org
- 2Department of Biochemistry and Molecular Biology, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 2212555
- DOI: http://doi.org/10.4174/jkss.2013.85.6.296
Abstract
- PURPOSE
Inhibition of the intimal hyperplasia after vascular surgery is an important issue. The purpose of this study is to define whether perivascular application of rapamycin, imatinib mesylate or cysteamine can reduce intimal hyperplasia in a carotid balloon injury model.
METHODS
Each drug was mixed with 40% pluronic gel solution and was topically applied over the injured carotid artery evenly. Two or four weeks after injury, the arteries were harvested and morphometric analysis was done.
RESULTS
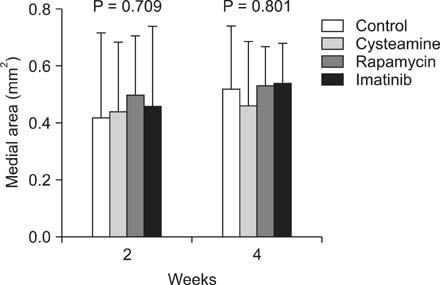
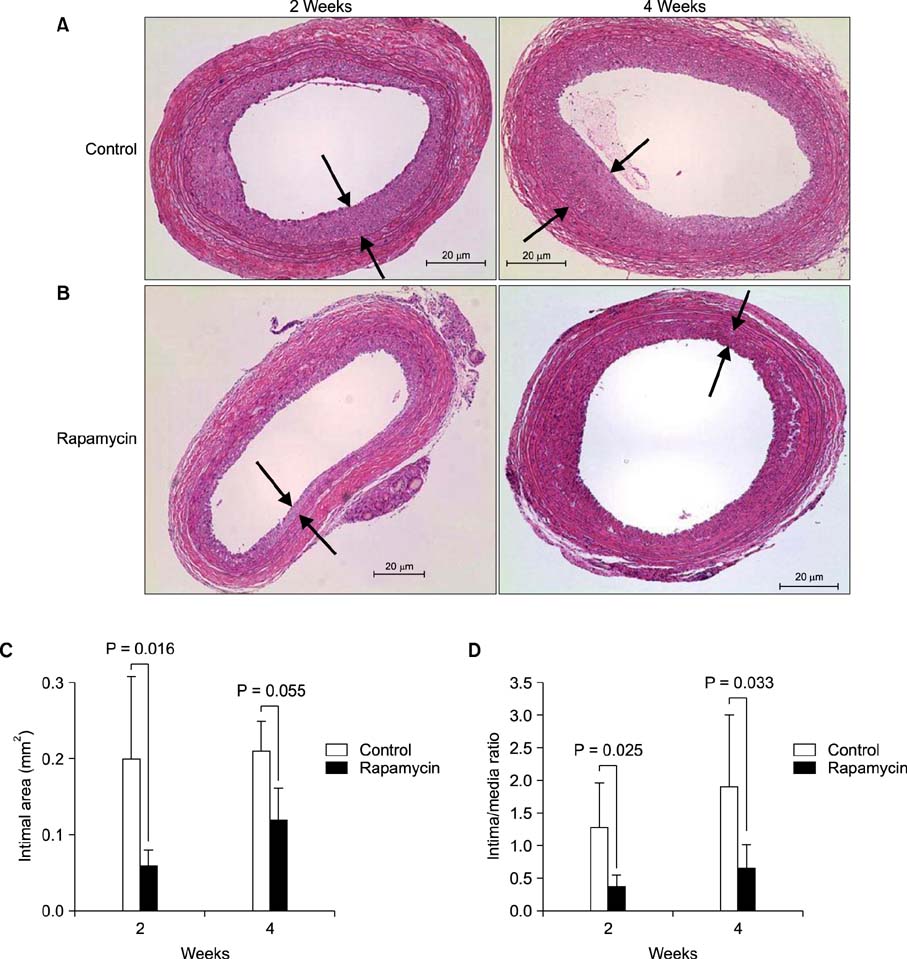
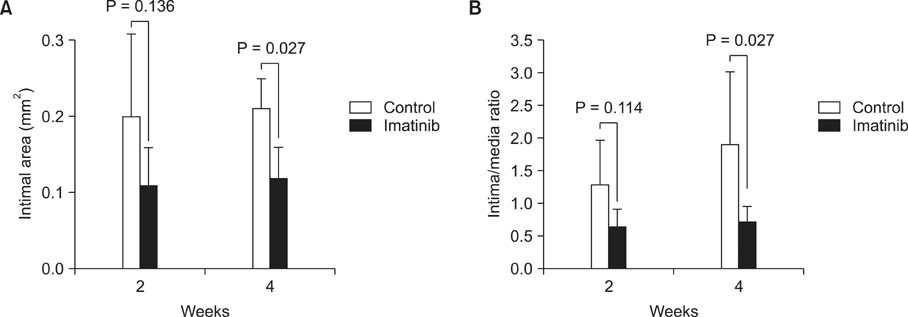
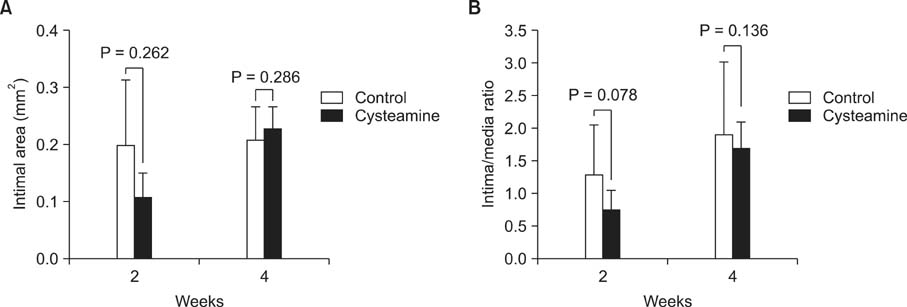
The medial areas were not significantly different in each group and a thinning of the media as a toxic drug effect was not observed in any treatment group. The intimal area and intima-to-media (I/M) ratio were significantly reduced in rapamycin-treated group and imatinib-treated group (P < 0.05). But cysteamine-treated group showed a trend of decrease in I/M ratio in 2 weeks, but no difference in 4 weeks.
CONCLUSION
Perivascular delivery of imatinib or rapamycin with pluronic gel attenuated the development of intimal hyperplasia. But cysteamine did not. Further studies are needed to refine the optimal drug dosages in large animal models.
MeSH Terms
Figure
Reference
-
1. Bauters C, Isner JM. The biology of restenosis. Prog Cardiovasc Dis. 1997; 40:107–116.2. Kester M, Waybill P, Kozak M. New strategies to prevent restenosis. Am J Cardiovasc Drugs. 2001; 1:77–83.3. Clowes AW, Reidy MA, Clowes MM. Kinetics of cellular proliferation after arterial injury. I. Smooth muscle growth in the absence of endothelium. Lab Invest. 1983; 49:327–333.4. Min SK, Kenagy RD, Clowes AW. Induction of vascular atrophy as a novel approach to treating restenosis: a review. J Vasc Surg. 2008; 47:662–670.5. Moses JW, Leon MB, Popma JJ, Fitzgerald PJ, Holmes DR, O'Shaughnessy C, et al. Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med. 2003; 349:1315–1323.6. Park SJ, Shim WH, Ho DS, Raizner AE, Park SW, Hong MK, et al. A paclitaxel-eluting stent for the prevention of coronary restenosis. N Engl J Med. 2003; 348:1537–1545.7. Navarese EP, Austin D, Gurbel PA, Andreotti F, Tantry U, James S, et al. Drug-coated balloons in treatment of in-stent restenosis: a meta-analysis of randomised controlled trials. Clin Res Cardiol. 2013; 102:279–287.8. Kohler TR, Toleikis PM, Gravett DM, Avelar RL. Inhibition of neointimal hyperplasia in a sheep model of dialysis access failure with the bioabsorbable Vascular Wrap paclitaxel-eluting mesh. J Vasc Surg. 2007; 45:1029–1037.9. Kwon JS, Park SS, Kim YG, Son JH, Lee YS, Kim KS, et al. Perivascular delivery of paclitaxel with F-127 pluronic gel inhibits neointimal hyperplasia in a rat carotid artery injury model. Korean Circ J. 2005; 35:221–227.10. Miyazaki S, Takeuchi S, Yokouchi C, Takada M. Pluronic F-127 gels as a vehicle for topical administration of anticancer agents. Chem Pharm Bull (Tokyo). 1984; 32:4205–4208.11. Myllärniemi M, Frosen J, Calderon Ramirez LG, Buchdunger E, Lemstrom K, Hayry P. Selective tyrosine kinase inhibitor for the platelet-derived growth factor receptor in vitro inhibits smooth muscle cell proliferation after reinjury of arterial intima in vivo. Cardiovasc Drugs Ther. 1999; 13:159–168.12. Min SK, Huh S, Ahn MS, Ha J, Chung JK, Kim SJ. Expression of MMPs and TIMPs in balloon-injured rat artery. Asian J Surg. 2001; 24:270–277.13. Gregory CR, Huie P, Billingham ME, Morris RE. Rapamycin inhibits arterial intimal thickening caused by both alloimmune and mechanical injury. Its effect on cellular, growth factor, and cytokine response in injured vessels. Transplantation. 1993; 55:1409–1418.14. Buchdunger E, Cioffi CL, Law N, Stover D, Ohno-Jones S, Druker BJ, et al. Abl protein-tyrosine kinase inhibitor STI571 inhibits in vitro signal transduction mediated by c-kit and platelet-derived growth factor receptors. J Pharmacol Exp Ther. 2000; 295:139–145.15. Wang CH, Anderson N, Li SH, Szmitko PE, Cherng WJ, Fedak PW, et al. Stem cell factor deficiency is vasculoprotective: unraveling a new therapeutic potential of imatinib mesylate. Circ Res. 2006; 99:617–625.16. Sihvola R, Koskinen P, Myllarniemi M, Loubtchenkov M, Hayry P, Buchdunger E, et al. Prevention of cardiac allograft arteriosclerosis by protein tyrosine kinase inhibitor selective for platelet-derived growth factor receptor. Circulation. 1999; 99:2295–2301.17. Lassila M, Allen TJ, Cao Z, Thallas V, Jandeleit-Dahm KA, Candido R, et al. Imatinib attenuates diabetes-associated atherosclerosis. Arterioscler Thromb Vasc Biol. 2004; 24:935–942.18. Vamvakopoulos JE, Petrov L, Aavik S, Lehti S, Aavik E, Hayry P. Synergistic suppression of rat neointimal hyperplasia by rapamycin and imatinib mesylate: implications for the prevention of accelerated arteriosclerosis. J Vasc Res. 2006; 43:184–192.19. Iismaa SE, Mearns BM, Lorand L, Graham RM. Transglutaminases and disease: lessons from genetically engineered mouse models and inherited disorders. Physiol Rev. 2009; 89:991–1023.20. Zemskov EA, Janiak A, Hang J, Waghray A, Belkin AM. The role of tissue transglutaminase in cell-matrix interactions. Front Biosci. 2006; 11:1057–1076.21. Signore PE, Machan LS, Jackson JK, Burt H, Bromley P, Wilson JE, et al. Complete inhibition of intimal hyperplasia by perivascular delivery of paclitaxel in balloon-injured rat carotid arteries. J Vasc Interv Radiol. 2001; 12:79–88.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Different Responses of Neointimal Cells to Imatinib Mesylate and Rapamycin Compared with Normal Vascular Smooth Muscle Cells
- Effect of imatinib mesylate and rapamycin on the preformed intimal hyperplasia in rat carotid injury model
- Clinical Study for Inhibition of Intimal Hyperplasia : Past & Present
- Perivascular Delivery of Rapamycin in Pluronic Gel Inhibits Neointimal Hyperplasia in a Rat Carotid Artery Injury Model, and the Complementary Role of Carotid Arteriography
- Intimal Hyperplasia