J Pathol Transl Med.
2016 Jan;50(1):45-51. 10.4132/jptm.2015.10.16.
Eosinophils in Colorectal Neoplasms Associated with Expression of CCL11 and CCL24
- Affiliations
-
- 1Department of Pathology, Kyung Hee University Medical Center, Graduate School of Medicine, Kyung Hee University, Seoul, Korea.
- 2Department of Pathology, Kyung Hee University Hospital of Gangdong, Seoul, Korea. sungjig@khu.ac.kr
- 3Department of Pathogy, Kyung Hee University Hospital of Gangdong, Graduate School of Medicine, Kyung Hee University, Seoul, Korea.
- 4Department of Pathology, Kyung Hee University Medical Center, Seoul, Korea.
- KMID: 2211400
- DOI: http://doi.org/10.4132/jptm.2015.10.16
Abstract
- BACKGROUND
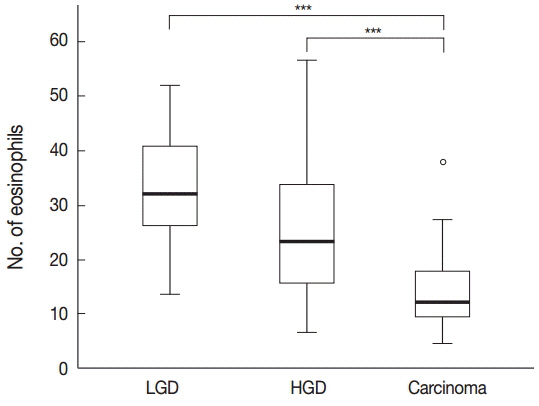
A decrease in the number of tissue eosinophils is known to reflect the malignancy potential of neoplastic lesions and even prognosis. Increased levels of the chemokines CCL11 and CCL24 in serum and tissue are also known to have diagnostic value as serum tumor markers or prognostic factors. The aim of this study was to evaluate the correlation between the degree of tissue eosinophilia and the expression of these chemokines in the glandular and stromal cells of colorectal neoplastic lesions ranging from benign to malignant tumors.
METHODS
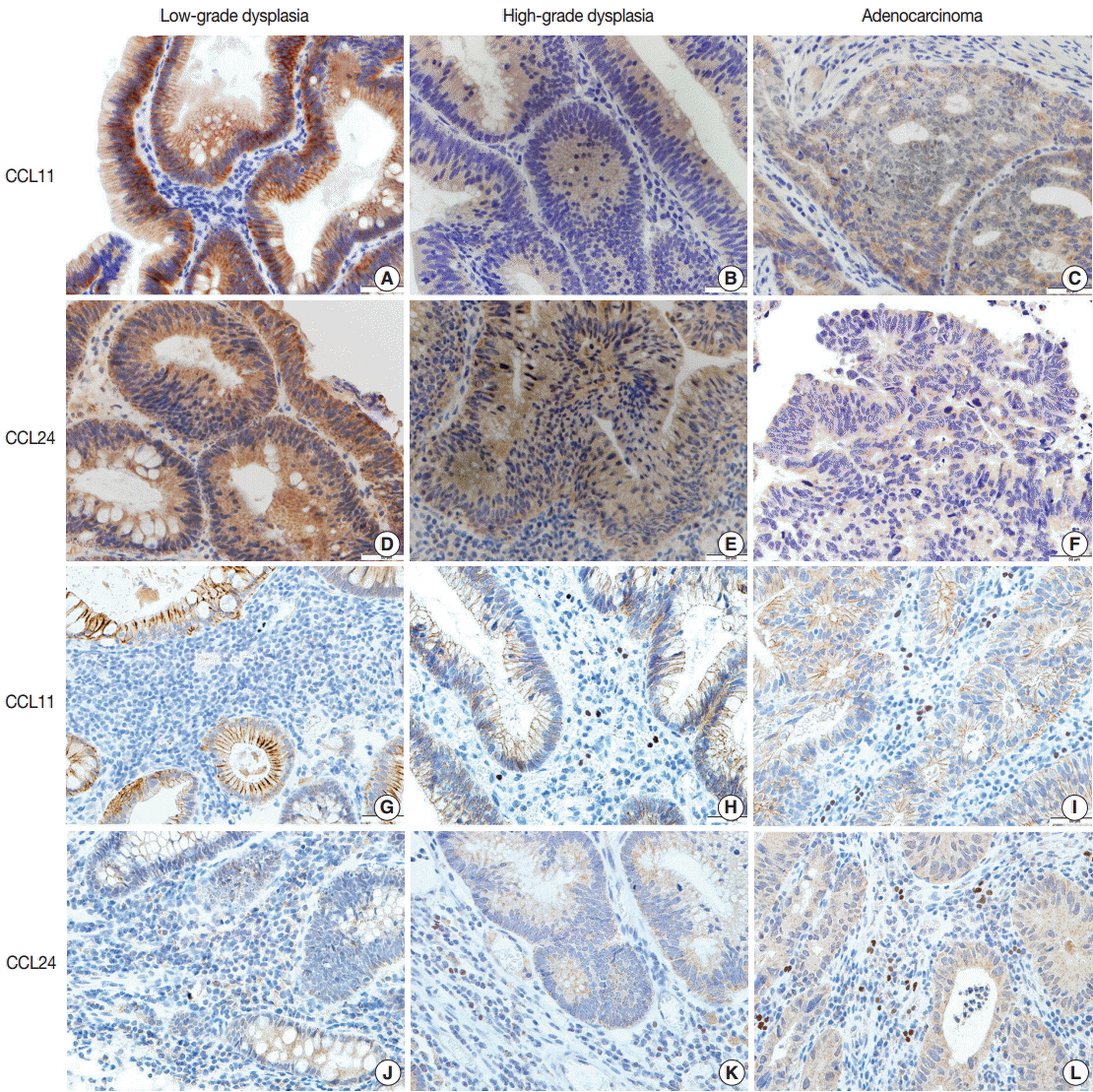
We counted the number of infiltrating eosinophils in neoplastic lesion tissue and we evaluated the expression of CCL11 and CCL24 in glandular cells and stromal cells by immunohistochemical staining.
RESULTS
The results showed that the number of eosinophils decreased significantly and the expression of CCL11 and CCL24 in glandular cells decreased with tumor progression, whereas the stromal expression of CCL11 and CCL24 appeared to increase.
CONCLUSIONS
The discrepancy in CCL11 and CCL24 expression between glandular cells and stromal cells might shed light on how colorectal cancer evades the immune system, which would enable further development of immunotherapies that target these chemokines. Further research on eosinophil biology and the expression pattern of chemokines in tumor cells is needed.
Keyword
MeSH Terms
Figure
Reference
-
1. Rothenberg ME, Hogan SP. The eosinophil. Annu Rev Immunol. 2006; 24:147–74.
Article2. Adkinson NF, Middleton E Jr. Middleton’s allergy: principles and practice. Philadelphia: Mosby/Elsevier;2009.3. Legrand F, Driss V, Delbeke M, et al. Human eosinophils exert TNF-alpha and granzyme A-mediated tumoricidal activity toward colon carcinoma cells. J Immunol. 2010; 185:7443–51.4. Capron M, Legrand F. Functions of eosinophil granulocytes: from anti-parasite immunity to anti-tumoral potential. Bull Acad Natl Med. 2009; 193:339–46.5. Pretlow TP, Keith EF, Cryar AK, et al. Eosinophil infiltration of human colonic carcinomas as a prognostic indicator. Cancer Res. 1983; 43:2997–3000.6. Teo PZ, Utz PJ, Mollick JA. Using the allergic immune system to target cancer: activity of IgE antibodies specific for human CD20 and MUC1. Cancer Immunol Immunother. 2012; 61:2295–309.
Article7. Woerly G, Roger N, Loiseau S, Dombrowicz D, Capron A, Capron M. Expression of CD28 and CD86 by human eosinophils and role in the secretion of type 1 cytokines (interleukin 2 and interferon gamma): inhibition by immunoglobulin a complexes. J Exp Med. 1999; 190:487–95.8. Dorta RG, Landman G, Kowalski LP, Lauris JR, Latorre MR, Oliveira DT. Tumour-associated tissue eosinophilia as a prognostic factor in oral squamous cell carcinomas. Histopathology. 2002; 41:152–7.
Article9. Fujii M, Yamashita T, Ishiguro R, Tashiro M, Kameyama K. Significance of epidermal growth factor receptor and tumor associated tissue eosinophilia in the prognosis of patients with nasopharyngeal carcinoma. Auris Nasus Larynx. 2002; 29:175–81.
Article10. Isaacson NH, Rapoport P. Eosinophilia in malignant tumors: its significance. Ann Intern Med. 1946; 25:893–902.
Article11. Goldsmith MM, Belchis DA, Cresson DH, Merritt WD 3rd, Askin FB. The importance of the eosinophil in head and neck cancer. Otolaryngol Head Neck Surg. 1992; 106:27–33.
Article12. Thompson AC, Bradley PJ, Griffin NR. Tumor-associated tissue eosinophilia and long-term prognosis for carcinoma of the larynx. Am J Surg. 1994; 168:469–71.
Article13. Nielsen HJ, Hansen U, Christensen IJ, Reimert CM, Brünner N, Moesgaard F. Independent prognostic value of eosinophil and mast cell infiltration in colorectal cancer tissue. J Pathol. 1999; 189:487–95.
Article14. Fernández-Acenero MJ, Galindo-Gallego M, Sanz J, Aljama A. Prognostic influence of tumor-associated eosinophilic infiltrate in colorectal carcinoma. Cancer. 2000; 88:1544–8.
Article15. Fisher ER, Paik SM, Rockette H, Jones J, Caplan R, Fisher B. Prognostic significance of eosinophils and mast cells in rectal cancer: findings from the National Surgical Adjuvant Breast and Bowel Project (protocol R-01). Hum Pathol. 1989; 20:159–63.
Article16. Oliveira DT, Biassi TP, Faustino SE, Carvalho AL, Landman G, Kowalski LP. Eosinophils may predict occult lymph node metastasis in early oral cancer. Clin Oral Investig. 2012; 16:1523–8.
Article17. Kiziltaş S, Sezgin Ramadan S, Topuzoğlu A, Küllü S. Does the severity of tissue eosinophilia of colonic neoplasms reflect their malignancy potential? Turk J Gastroenterol. 2008; 19:239–44.18. Polydorides AD, Mukherjee B, Gruber SB, McKenna BJ, Appelman HD, Greenson JK. Adenoma-infiltrating lymphocytes (AILs) are a potential marker of hereditary nonpolyposis colorectal cancer. Am J Surg Pathol. 2008; 32:1661–6.
Article19. Moezzi J, Gopalswamy N, Haas RJ Jr, Markert RJ, Suryaprasad S, Bhutani MS. Stromal eosinophilia in colonic epithelial neoplasms. Am J Gastroenterol. 2000; 95:520–3.
Article20. Provost V, Larose MC, Langlois A, Rola-Pleszczynski M, Flamand N, Laviolette M. CCL26/eotaxin-3 is more effective to induce the migration of eosinophils of asthmatics than CCL11/eotaxin-1 and CCL24/eotaxin-2. J Leukoc Biol. 2013; 94:213–22.
Article21. Schaefer D, Meyer JE, Pods R, et al. Endothelial and epithelial expression of eotaxin-2 (CCL24) in nasal polyps. Int Arch Allergy Immunol. 2006; 140:205–14.
Article22. Menzies-Gow A, Ying S, Sabroe I, et al. Eotaxin (CCL11) and eotaxin-2 (CCL24) induce recruitment of eosinophils, basophils, neutrophils, and macrophages as well as features of early- and late-phase allergic reactions following cutaneous injection in human atopic and nonatopic volunteers. J Immunol. 2002; 169:2712–8.
Article23. Cheadle EJ, Riyad K, Subar D, et al. Eotaxin-2 and colorectal cancer: a potential target for immune therapy. Clin Cancer Res. 2007; 13:5719–28.
Article24. Erreni M, Bianchi P, Laghi L, et al. Expression of chemokines and chemokine receptors in human colon cancer. Methods Enzymol. 2009; 460:105–21.25. Cui G, Shi Y, Cui J, Tang F, Florholmen J. Immune microenvironmental shift along human colorectal adenoma-carcinoma sequence: is it relevant to tumor development, biomarkers and biotherapeutic targets? Scand J Gastroenterol. 2012; 47:367–77.
Article26. Wågsäter D, Löfgren S, Hugander A, Dienus O, Dimberg J. Analysis of single nucleotide polymorphism in the promoter and protein expression of the chemokine eotaxin-1 in colorectal cancer patients. World J Surg Oncol. 2007; 5:84.
Article27. Watanabe H, Miki C, Okugawa Y, Toiyama Y, Inoue Y, Kusunoki M. Decreased expression of monocyte chemoattractant protein-1 predicts poor prognosis following curative resection of colorectal cancer. Dis Colon Rectum. 2008; 51:1800–5.
Article28. Agarwal M, He C, Siddiqui J, Wei JT, Macoska JA. CCL11 (eotaxin-1): a new diagnostic serum marker for prostate cancer. Prostate. 2013; 73:573–81.
Article29. Monson JR, Ramsden C, Guillou PJ. Decreased interleukin-2 production in patients with gastrointestinal cancer. Br J Surg. 1986; 73:483–6.
Article30. King J, Caplehorn JR, Ross WB, Morris DL. High serum carcinoembryonic antigen concentration in patients with colorectal liver metastases is associated with poor cell-mediated immunity, which is predictive of survival. Br J Surg. 1997; 84:1382–5.
Article31. Nielsen HJ, Moesgaard F, Hammer JH. Effect of ranitidine and low-dose interleukin-2 in vitro on NK-cell activity in peripheral blood from patients with liver metastases from colorectal cancer. Eur J Surg Oncol. 1995; 21:526–30.
Article32. Bovo G, Brivio F, Brenna A, et al. Pre-operative interleukin-2 immunotherapy induces eosinophilic infiltration in colorectal neoplastic stroma. Pathologica. 1995; 87:135–8.33. Yoshida N, Aizu-Yokota E, Sonoda Y, Moriwaki Y, Kishi K, Kasahara T. Production and regulation of eotaxin-2/CCL24 in a differentiated human leukemic cell line, HT93. Biol Pharm Bull. 2007; 30:1826–32.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Plasma Eotaxin Family Level in Aspirin-Intolerant and Aspirin-Tolerant Asthma Patients
- Serum CCL11 Levels in Benign Prostatic Hyperplasia and Prostate Cancer
- Multiple Signaling Pathways Contribute to the Thrombin-induced Secretory Phenotype in Vascular Smooth Muscle Cells
- Immunohistochemieal Study of Expression of nm23 and CD44 Protein in
- Correlation between expression of matrix metalloproteinase-2 (MMP-2), and matrix metalloproteinase-9 (MMP-9) and angiogenesis in colorectal adenocarcinoma