J Korean Acad Prosthodont.
2015 Jul;53(3):198-206. 10.4047/jkap.2015.53.3.198.
Effect of titanium surface microgrooves and thermal oxidation on in vitro osteoblast responses
- Affiliations
-
- 1Department of Biomaterials & Prosthodontics, Kyung Hee University Hospital at Gangdong, School of Dentistry, Kyung Hee University, Seoul, Republic of Korea. ysprosth@hanmail.net
- 2Green Ceramics Division, Korea Institute of Ceramic Engineering and Technology, Seoul, Republic of Korea.
- KMID: 2195249
- DOI: http://doi.org/10.4047/jkap.2015.53.3.198
Abstract
- PURPOSE
We aimed to investigate the effect of combined various microgrooves and thermal oxidation on the titanium (Ti) and to evaluate various in vitro responses of human periodontal ligament cells (PLCs).
MATERIALS AND METHODS
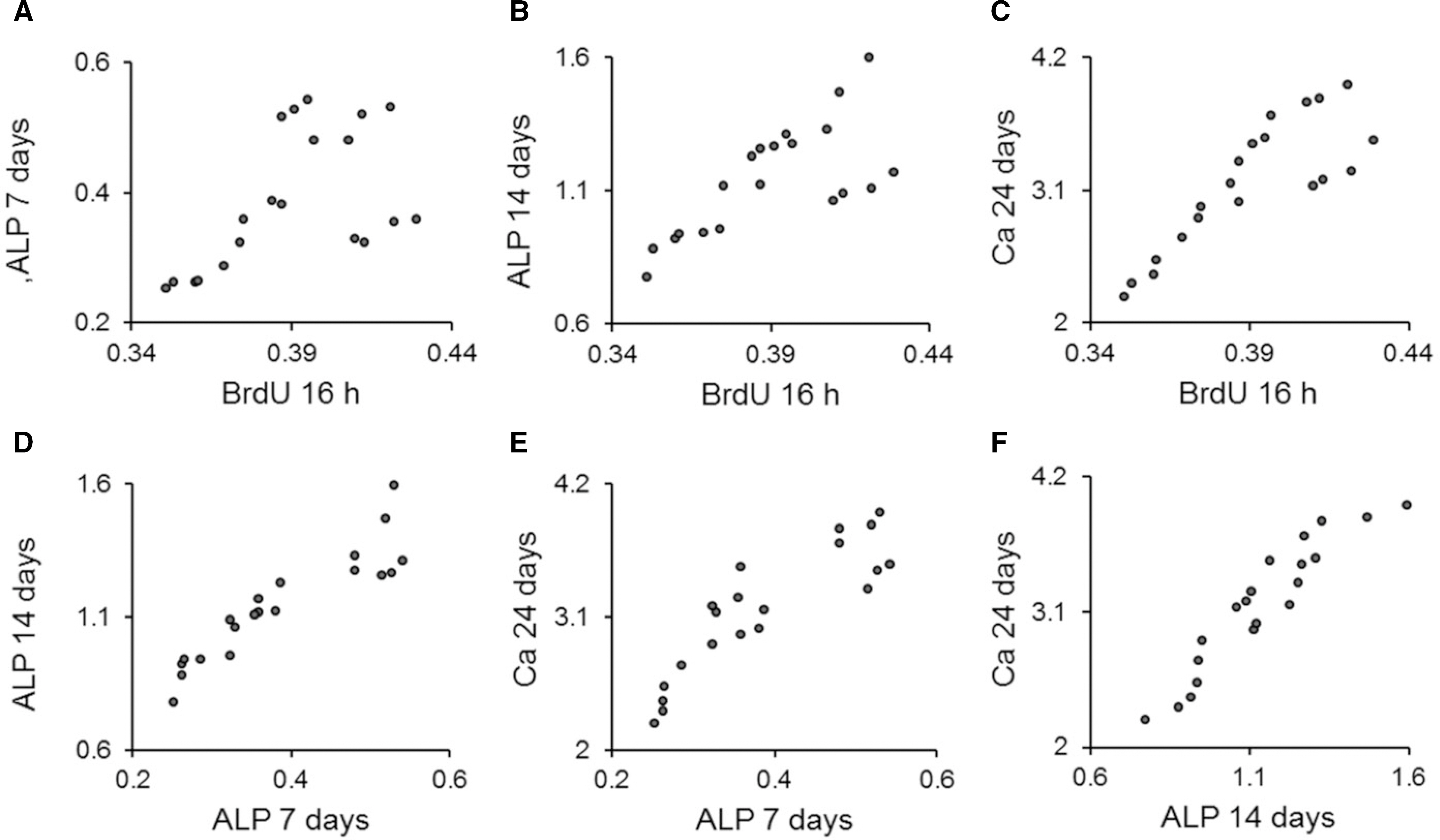
Grade II titanium disks were fabricated. Microgrooves were applied on titanium discs to have 0/0 microm, 15/3.5 microm, 30/10 microm, and 60/10 microm of respective width/depth by photolithography. Thermal oxidation was performed on the microgrooves of Ti substrata for 3 h at 700degrees C in air. The experiments were divided into 3 groups: control group (ST), thermal oxidation group (ST/TO), and combined microgrooves and thermal oxidation group (Gr15-TO, Gr30-TO, Gr60-TO). Surface characterization was performed by field-emission scanning microscopy. Cell adhesion, osteoblastic differentiation, and mineralization were analyzed using the bromodeoxyurdine (BrdU), Alkaline phosphatase (ALP) activity, and extracellular calcium deposition assays, respectively. Statistical analysis was performed using the oneway analysis of variance and Pearson's bivariate correlation analysis (SPSS Version 17.0).
RESULTS
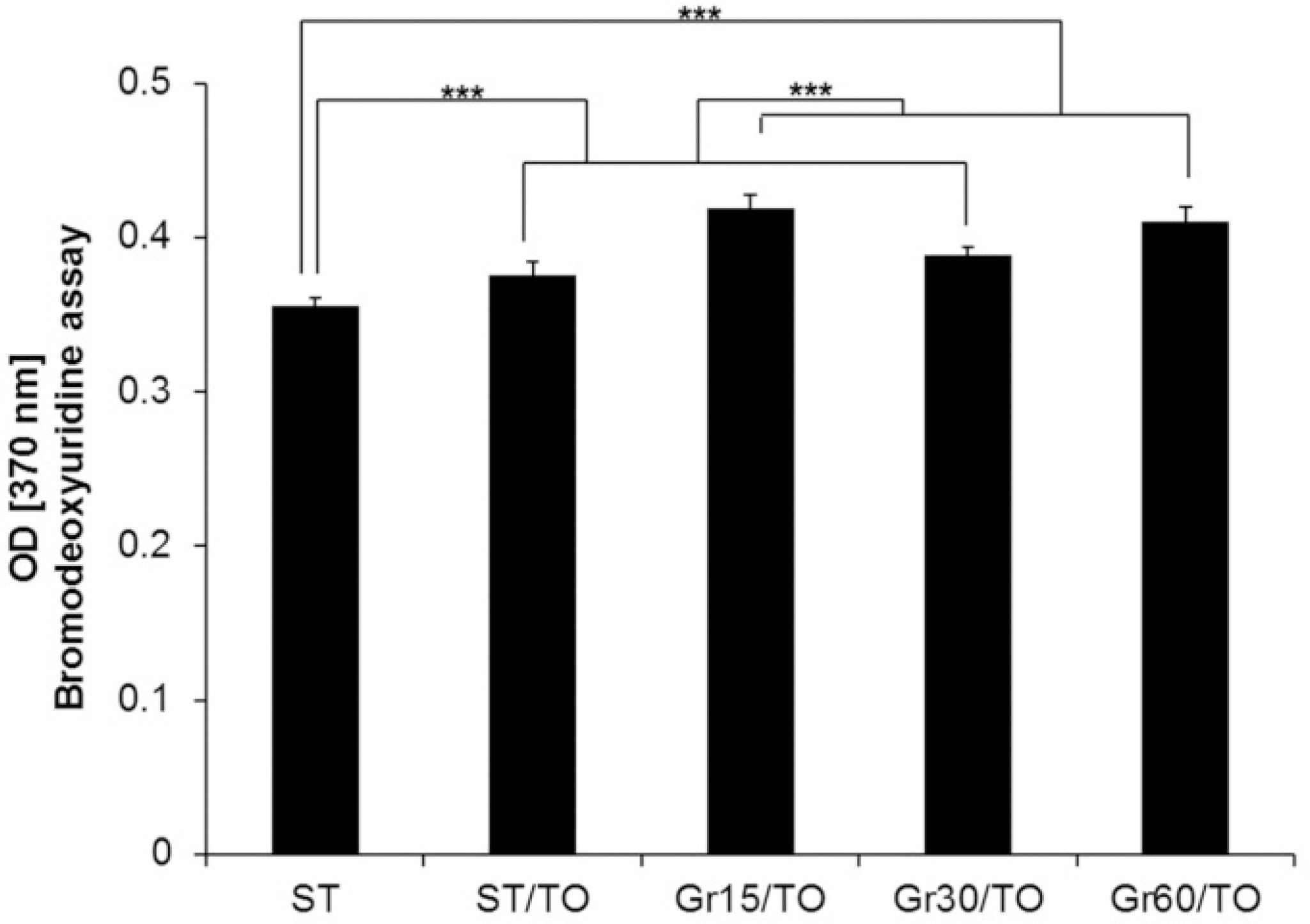
In general, the combined microgrooves and thermal oxidation group (Gr15-TO, Gr30-TO, Gr60-TO) showed significantly higher levels compared with the control (ST) or thermal oxidation (ST-TO) groups in the BrdU expression, ALP activity, and extracellular calcium deposition. Gr60-TO group induced highest levels of cell adhesion and osteoblastic differentiation.
CONCLUSION
Within the limitation of this study, we conclude that the Ti surface treatment using combined microgrooves and thermal oxidation is highly effective in inducing the cell adhesion andosteoblastic differentiation. The propose surface is also expected to be effective in inducing rapid and strong osseointegration of Ti oral implants.
MeSH Terms
Figure
Reference
-
1. Albrektsson T, Wennerberg A. Oral implant surfaces: Part 1–review focusing on topographic and chemical properties of different surfaces and in vivo responses to them. Int J Prosthodont. 2004; 17:536–43.2. Long M, Rack HJ. Titanium alloys in total joint replacement–a materials science perspective. Biomaterials. 1998; 19:1621–39.
Article3. Jansen JA, van de Waerden JP, Wolke JG, de Groot K. Histologic evaluation of the osseous adaptation to titanium and hydroxya-patite-coated titanium implants. J Biomed Mater Res. 1991; 25:973–89.
Article4. Gaviria L, Salcido JP, Guda T, Ong JL. Current trends in dental implants. J Korean Assoc Oral Maxillofac Surg. 2014; 40:50–60.
Article5. Wong M, Eulenberger J, Schenk R, Hunziker E. Effect of surface topology on the osseointegration of implant materials in trabecular bone. J Biomed Mater Res. 1995; 29:1567–75.
Article6. Junker R, Dimakis A, Thoneick M, Jansen JA. Effects of implant surface coatings and composition on bone integration: a systematic review. Clin Oral Implants Res. 2009; 20:185–206.
Article7. Balasundaram G, Yao C, Webster TJ. TiO2 nanotubes functionalized with regions of bone morphogenetic protein-2 increases osteoblast adhesion. J Biomed Mater Res A. 2008; 84:447–53.8. Buch F, Albrektsson T, Herbst E. Direct current influence on bone formation in titanium implants. Biomaterials. 1984; 5:341–6.
Article9. Eisenbarth E, Velten D, Schenk-Meuser K, Linez P, Biehl V, Duschner H, Breme J, Hildebrand H. Interactions between cells and titanium surfaces. Biomol Eng. 2002; 19:243–9.
Article10. Kataoka Y, Tamaki Y, Miyazaki T. Synergistic responses of su-perficial chemistry and micro topography of titanium created by wire-type electric discharge machining. Biomed Mater Eng. 2011; 21:113–21.
Article11. Feng B, Chen JY, Qi SK, He L, Zhao JZ, Zhang XD. Characterization of surface oxide films on titanium and bioactivity. J Mater Sci. 2002; 13:457–64.12. Raikar GN, Gregory JC, Ong JL, Lucas LC, Lemons JE, Kawahara D, Nakamura M. Surface characterization of titanium implants. J Vac Sci Technol. 1995; 13:2633–7.
Article13. Boyan BD, Hummert TW, Dean DD, Schwartz Z. Role of material surfaces in regulating bone and cartilage cell response. Biomaterials. 1996; 17:137–46.
Article14. Garc l′a-Alonso MC, Saldañ a L, Vallé s G, Gonza′lez-Carrasco JL, Gonza′lez-Cabrero J, Mart l′nez ME, Gil-Garay E, Munuera L. In vitro corrosion behaviour and osteoblast response of thermally oxidised Ti6Al4V alloy. Biomaterials. 2003; 24:19–26.
Article15. Saldañ a L, Vilaboa N, Vallé s G, Gonza′lez-Cabrero J, Munuera L. Osteoblast response to thermally oxidized Ti6Al4V alloy. J Biomed Mater Res A. 2005; 73:97–107.16. den Braber ET, de Ruijter JE, Smits HT, Ginsel LA, von Recum AF, Jansen JA. Quantitative analysis of cell proliferation and ori-entation on substrata with uniform parallel surface microgrooves. Biomaterials. 1996; 17:1093–9.
Article17. Lee SW, Kim SY, Rhyu IC, Chung WY, Leesungbok R, Lee KW. Influence of microgroove dimension on cell behavior of human gingival fibroblasts cultured on titanium substrata. Clin Oral Implants Res. 2009; 20:56–66.
Article18. Zhao G, Zinger O, Schwartz Z, Wieland M, Landolt D, Boyan BD. Osteoblast-like cells are sensitive to submicron-scale surface structure. Clin Oral Implants Res. 2006; 17:258–64.
Article19. Zhao L, Mei S, Chu PK, Zhang Y, Wu Z. The influence of hierarchical hybrid micro/nano-textured titanium surface with titania nanotubes on osteoblast functions. Biomaterials. 2010; 31:5072–82.
Article20. Gittens RA, McLachlan T, Olivares-Navarrete R, Cai Y, Berner S, Tannenbaum R, Schwartz Z, Sandhage KH, Boyan BD. The effects of combined micron-/submicron-scale surface roughness and nanoscale features on cell proliferation and differentiation. Biomaterials. 2011; 32:3395–403.
Article21. Gratzner HG. Monoclonal antibody to 5-bromo- and 5-iodo-deoxyuridine: A new reagent for detection of DNA replication. Science. 1982; 218:474–5.
Article22. Owen TA, Aronow M, Shalhoub V, Barone LM, Wilming L, Tassinari MS, Kennedy MB, Pockwinse S, Lian JB, Stein GS. Progressive development of the rat osteoblast phenotype in vitro: reciprocal relationships in expression of genes associated with osteoblast proliferation and differentiation during formation of the bone extracellular matrix. J Cell Physiol. 1990; 143:420–30.
Article23. Feng B, Weng J, Yang BC, Qu SX, Zhang XD. Characterization of surface oxide films on titanium and adhesion of osteoblast. Biomaterials. 2003; 24:4663–70.
Article24. Lee MH, Oh N, Lee SW, Leesungbok R, Kim SE, Yun YP, Kang JH. Factors influencing osteoblast maturation on microgrooved titanium substrata. Biomaterials. 2010; 31:3804–15.
Article25. Lee MH, Kang JH, Lee SW. The effect of surface microgrooves and anodic oxidation on the surface characteristics of titanium and the osteogenic activity of human periodontal ligament cells. Arch Oral Biol. 2013; 58:59–66.
Article26. Im BJ, Lee SW, Oh N, Lee MH, Kang JH, Leesungbok R, Lee SC, Ahn SJ, Park JS. Texture direction of combined microgrooves and submicroscale topographies of titanium substrata influence adhesion, proliferation, and differentiation in human primary cells. Arch Oral Biol. 2012; 57:898–905.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Response of osteoblast-like cells on titanium surface treatment
- Effect of etched microgrooves on hydrophilicity of titanium and osteoblast responses: A pilot study
- Pre-Treatment of Titanium Alloy with Platelet-Rich Plasma Enhances Human Osteoblast Responses
- Biological responses of osteoblast-like cells to different Titanium surface by anodizing modification
- Osteoblast adhesion and differentiation on magnesium titanate surface