Allergy Asthma Immunol Res.
2014 Sep;6(5):449-457. 10.4168/aair.2014.6.5.449.
Prostaglandin E2 Induces IL-6 and IL-8 Production by the EP Receptors/Akt/NF-kappaB Pathways in Nasal Polyp-Derived Fibroblasts
- Affiliations
-
- 1Brain Korea 21 Plus for Biomedical Science, Korea University College of Medicine, Seoul, Korea. lhman@korea.ac.kr
- 2Institute for Medical Devices Clinical Trial Center, Guro Hospital, Korea University, Seoul, Korea.
- 3Department of Otorhinolaryngology-Head and Neck Surgery, College of Medicine, Korea University, Seoul, Korea.
- KMID: 2181082
- DOI: http://doi.org/10.4168/aair.2014.6.5.449
Abstract
- PURPOSE
Interleukin 6 (IL-6) and IL-8 participate in the pathogenesis of chronic rhinosinusitis with nasal polyps, and their levels are increased by prostaglandin E2 (PGE2) in different cell types. The purposes of this study were to determine whether PGE2 has any effect on the increase in the levels of IL-6 and IL-8 in nasal polyp-derived fibroblasts (NPDFs) and subsequently investigate the possible mechanism of this effect.
METHODS
Different concentrations of PGE2 were used to stimulate NPDFs at different time intervals. NPDFs were treated with agonists and antagonists of E prostanoid (EP) receptors. To determine the signaling pathway for the expression of PGE2-induced IL-6 and IL-8, PGE2 was treated with Akt and NF-kappaB inhibitors in NPDFs. Reverse transcription-polymerase chain reaction for IL-6 and IL-8 mRNAs was performed. IL-6 and IL-8 levels were measured byenzyme-linked immunosorbent assay (ELISA). The activation of Akt and NF-kappaB was evaluated by western blot analysis.
RESULTS
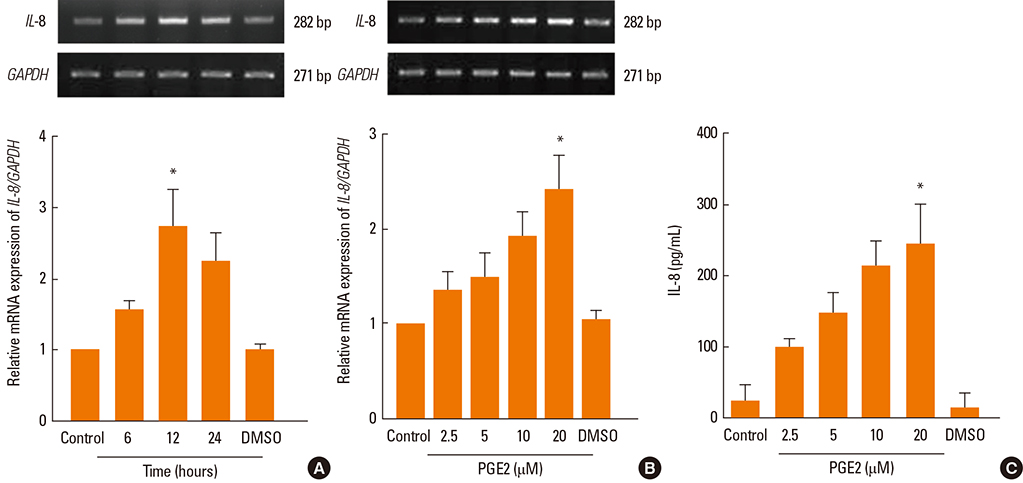
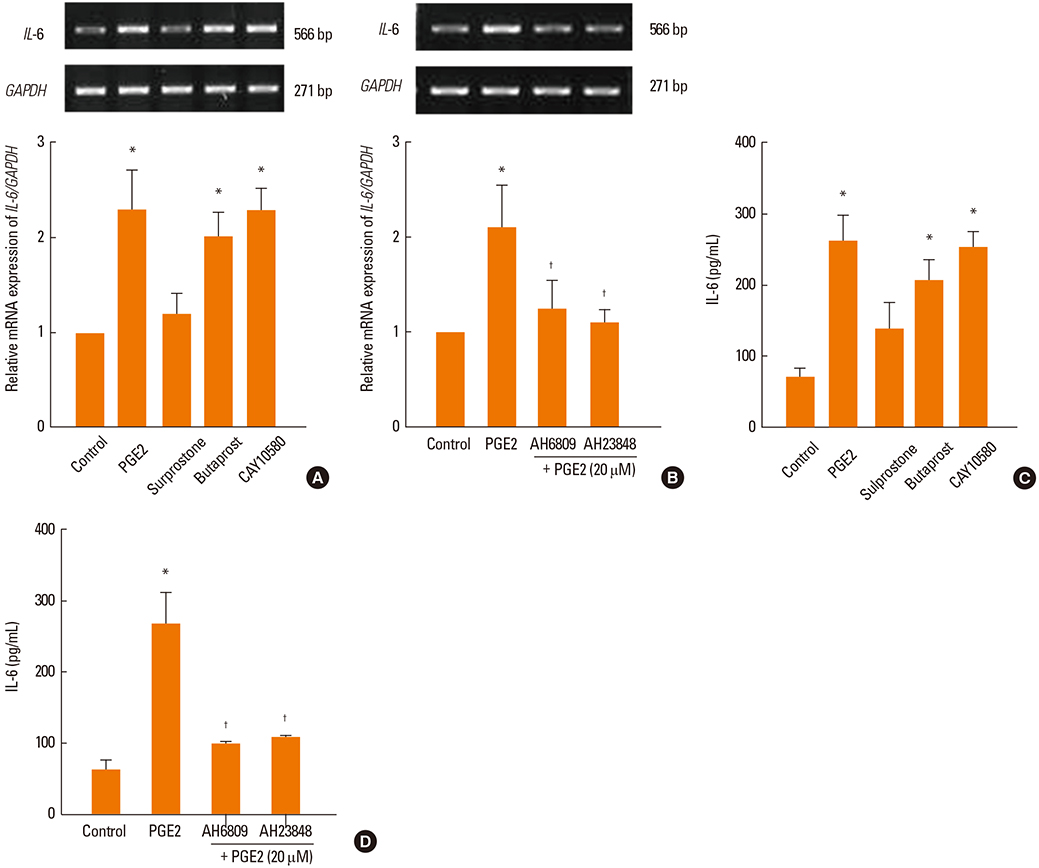
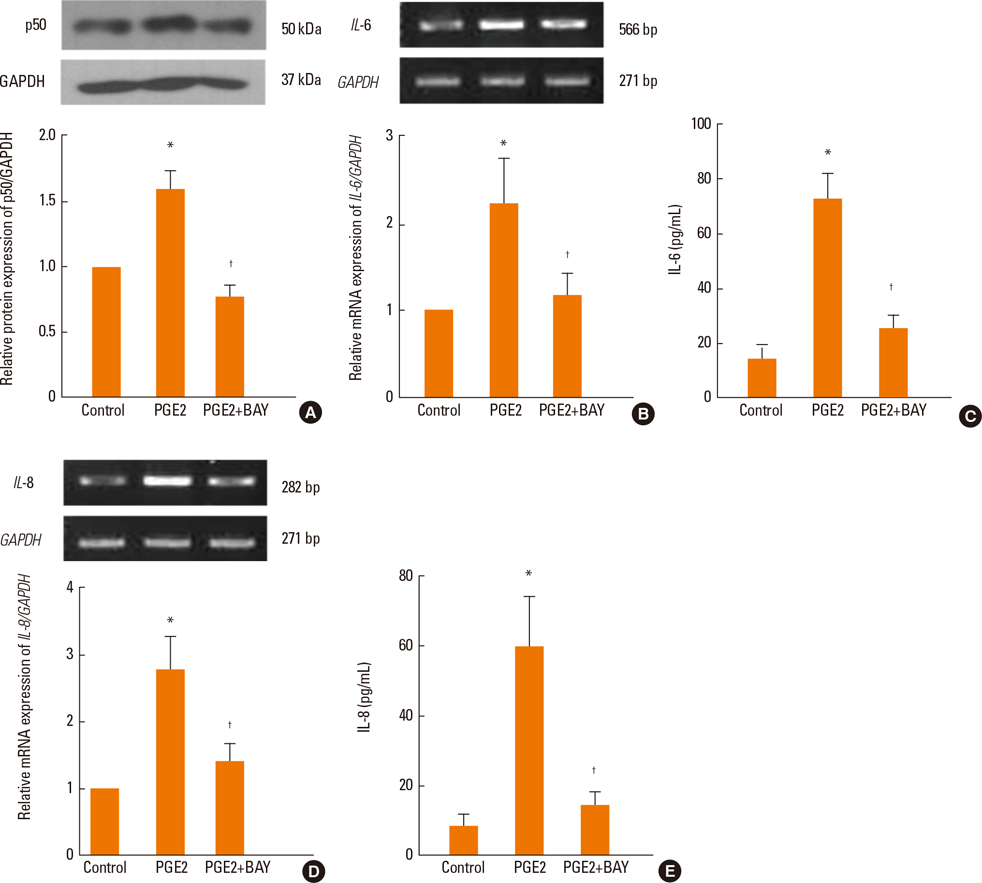
PGE2 significantly increased the mRNA and protein expression levels of IL-6 and IL-8 in NPDFs. The EP2 and EP4 agonists and antagonists induced and inhibited IL-6 expression. However, the EP4 agonist and antagonist were only observed to induce and inhibit IL-8 expression level. The Akt and NF-kappaB inhibitors significantly blocked PGE2-induced expression of IL-6 and IL-8.
CONCLUSIONS
PGE2 increases IL-6 expression via EP2 and EP4 receptors, and IL-8 expression via the EP4 receptor in NPDFs. It also activates the Akt and NF-kappaB signal pathways for the production of IL-6 and IL-8 in NPDFs. These results suggest that signaling pathway for IL-6 and IL-8 expression induced by PGE2 might be a useful therapeutic target for the treatment of nasal polyposis.
MeSH Terms
Figure
Reference
-
1. Pawankar R. Nasal polyposis: an update: editorial review. Curr Opin Allergy Clin Immunol. 2003; 3:1–6.2. Meltzer EO, Hamilos DL. Rhinosinusitis diagnosis and management for the clinician: a synopsis of recent consensus guidelines. Mayo Clin Proc. 2011; 86:427–443.3. Park JY, Pillinger MH, Abramson SB. Prostaglandin E2 synthesis and secretion: the role of PGE2 synthases. Clin Immunol. 2006; 119:229–240.4. Breyer RM, Bagdassarian CK, Myers SA, Breyer MD. Prostanoid receptors: subtypes and signaling. Annu Rev Pharmacol Toxicol. 2001; 41:661–690.5. Roca-Ferrer J, Pérez-Gonzalez M, Garcia-Garcia FJ, Pereda J, Pujols L, Alobid I, Mullol J, Picado C. Low prostaglandin E2 and cyclooxygenase expression in nasal mucosa fibroblasts of aspirin-intolerant asthmatics. Respirology. 2013; 18:711–717.6. Okano M, Fujiwara T, Haruna T, Kariya S, Makihara S, Higaki T, Nishizaki K. Prostaglandin E(2) suppresses staphylococcal enterotoxin-induced eosinophilia-associated cellular responses dominantly through an E-prostanoid 2-mediated pathway in nasal polyps. J Allergy Clin Immunol. 2009; 123:868–874.e13.7. Rinia AB, Kostamo K, Ebbens FA, van Drunen CM, Fokkens WJ. Nasal polyposis: a cellular-based approach to answering questions. Allergy. 2007; 62:348–358.8. Xi X, McMillan DH, Lehmann GM, Sime PJ, Libby RT, Huxlin KR, Feldon SE, Phipps RP. Ocular fibroblast diversity: implications for inflammation and ocular wound healing. Invest Ophthalmol Vis Sci. 2011; 52:4859–4865.9. Kim EC, Zhu Y, Andersen V, Sciaky D, Cao HJ, Meekins H, Smith TJ, Lance P. Cytokine-mediated PGE2 expression in human colonic fibroblasts. Am J Physiol. 1998; 275:C988–C994.10. Fray TR, Watson AL, Croft JM, Baker CD, Bailey J, Sirel N, Tobias A, Markwell PJ. A combination of aloe vera, curcumin, vitamin C, and taurine increases canine fibroblast migration and decreases tritiated water diffusion across canine keratinocytes in vitro. J Nutr. 2004; 134:2117S–2119S.11. Ritchlin C. Fibroblast biology. Effector signals released by the synovial fibroblast in arthritis. Arthritis Res. 2000; 2:356–360.12. Aso H, Ito S, Mori A, Morioka M, Suganuma N, Kondo M, Imaizumi K, Hasegawa Y. Prostaglandin E2 enhances interleukin-8 production via EP4 receptor in human pulmonary microvascular endothelial cells. Am J Physiol Lung Cell Mol Physiol. 2012; 302:L266–L273.13. Fiebich BL, Schleicher S, Spleiss O, Czygan M, Hüll M. Mechanisms of prostaglandin E2-induced interleukin-6 release in astrocytes: possible involvement of EP4-like receptors, p38 mitogen-activated protein kinase and protein kinase C. J Neurochem. 2001; 79:950–958.14. Cho JS, Moon YM, Park IH, Um JY, Moon JH, Park SJ, Lee SH, Kang HJ, Lee HM. Epigenetic regulation of myofibroblast differentiation and extracellular matrix production in nasal polyp-derived fibroblasts. Clin Exp Allergy. 2012; 42:872–882.15. Malerba M, Ricciardolo F, Radaeli A, Torregiani C, Ceriani L, Mori E, Bontempelli M, Tantucci C, Grassi V. Neutrophilic inflammation and IL-8 levels in induced sputum of alpha-1-antitrypsin PiMZ subjects. Thorax. 2006; 61:129–133.16. Ghaffar O, Lavigne F, Kamil A, Renzi P, Hamid Q. Interleukin-6 expression in chronic sinusitis: colocalization of gene transcripts to eosinophils, macrophages, T lymphocytes, and mast cells. Otolaryngol Head Neck Surg. 1998; 118:504–511.17. Danielsen A, Tynning T, Brokstad KA, Olofsson J, Davidsson A. Interleukin 5, IL6, IL12, IFN-gamma, RANTES and Fractalkine in human nasal polyps, turbinate mucosa and serum. Eur Arch Otorhinolaryngol. 2006; 263:282–289.18. Ural A, Tezer MS, Yücel A, Atilla H, Ileri F. Interleukin-4, interleukin-8 and E-selectin levels in intranasal polyposis patients with and without allergy: a comparative study. J Int Med Res. 2006; 34:520–524.19. Rhyoo C, Sanders SP, Leopold DA, Proud D. Sinus mucosal IL-8 gene expression in chronic rhinosinusitis. J Allergy Clin Immunol. 1999; 103:395–400.20. Shun CT, Lin SK, Hong CY, Huang HM, Liu CM. Hypoxia induces cysteine-rich 61, vascular endothelial growth factor, and interleukin-8 expressions in human nasal polyp fibroblasts: An implication of neutrophils in the pathogenesis of nasal polyposis. Am J Rhinol Allergy. 2011; 25:15–18.21. Takeuchi K, Yuta A, Sakakura Y. Interleukin-8 gene expression in chronic sinusitis. Am J Otolaryngol. 1995; 16:98–102.22. Allen JS, Eisma R, Leonard G, Lafreniere D, Kreutzer D. Interleukin-8 expression in human nasal polyps. Otolaryngol Head Neck Surg. 1997; 117:535–541.23. Dey I, Giembycz MA, Chadee K. Prostaglandin E(2) couples through EP(4) prostanoid receptors to induce IL-8 production in human colonic epithelial cell lines. Br J Pharmacol. 2009; 156:475–485.24. Mohan S, Ahmad AS, Glushakov AV, Chambers C, Doré S. Putative role of prostaglandin receptor in intracerebral hemorrhage. Front Neurol. 2012; 3:145.25. Benyahia C, Gomez I, Kanyinda L, Boukais K, Danel C, Leséche G, Longrois D, Norel X. PGE(2) receptor (EP(4)) agonists: potent dilators of human bronchi and future asthma therapy? Pulm Pharmacol Ther. 2012; 25:115–118.26. Sastre B, del Pozo V. Role of PGE2 in asthma and nonasthmatic eosinophilic bronchitis. Mediators Inflamm. 2012; 2012:645383.27. Bos CL, Richel DJ, Ritsema T, Peppelenbosch MP, Versteeg HH. Prostanoids and prostanoid receptors in signal transduction. Int J Biochem Cell Biol. 2004; 36:1187–1205.28. Wang P, Zhu F, Konstantopoulos K. Prostaglandin E2 induces interleukin-6 expression in human chondrocytes via cAMP/protein kinase A- and phosphatidylinositol 3-kinase-dependent NF-kappaB activation. Am J Physiol Cell Physiol. 2010; 298:C1445–C1456.29. Han SS, Yun H, Son DJ, Tompkins VS, Peng L, Chung ST, Kim JS, Park ES, Janz S. NF-kappaB/STAT3/PI3K signaling crosstalk in iMyc E mu B lymphoma. Mol Cancer. 2010; 9:97.30. Hanada T, Yoshimura A. Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev. 2002; 13:413–421.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Histamine Promotes the Release of Interleukin-6 via the H1R/p38 and NF-kappaB Pathways in Nasal Fibroblasts
- Effect of Prostaglandin E2 on Vascular Endothelial Growth Factor Production in Nasal Polyp Fibroblasts
- Prostaglandin E2 receptors as therapeutic targets in renal fibrosis
- Regulation of Interleukin-17 Production in Patients with Rheumatoid Arthritis by Phosphoinositide 3-kinase (PI3K)/ Akt and Nuclear Factor KappaB (NF-kappaB) Dependent Signal Transduction Pathway
- Comparison of Cytokine and Chemokine Production and Activation of NF-kappaB in RSV- and Adenovirus-infected Bronchial Epithelial Cells