Diabetes Metab J.
2012 Aug;36(4):293-299. 10.4093/dmj.2012.36.4.293.
Intracerebroventricular Injection of Metformin Induces Anorexia in Rats
- Affiliations
-
- 1Department of Physiology, Yeungnam University College of Medicine, Daegu, Korea. ywkim@med.yu.ac.kr
- 2Department of Internal Medicine, Yeungnam University College of Medicine, Daegu, Korea.
- KMID: 2174427
- DOI: http://doi.org/10.4093/dmj.2012.36.4.293
Abstract
- BACKGROUND
Metformin, an oral biguanide insulin-sensitizing agent, is well known to decrease appetite. Although there is evidence that metformin could affect the brain directly, the exact mechanism is not yet known.
METHODS
To evaluate whether metformin induces anorexia via the hypothalamus, various concentrations of metformin were injected into the lateral ventricle of rats through a chronically implanted catheter and food intake was measured for 24 hours. The hypothalamic neuropeptides associated with regulation of food intake were also analyzed following 1 hour of intracerebroventricular (ICV) injections of metformin.
RESULTS
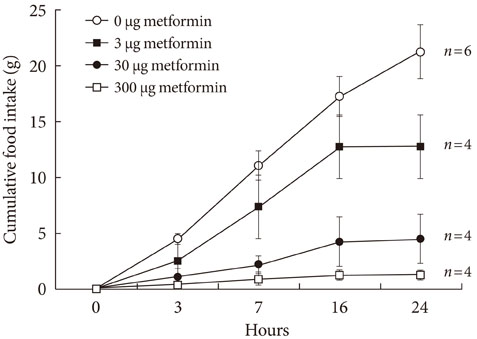
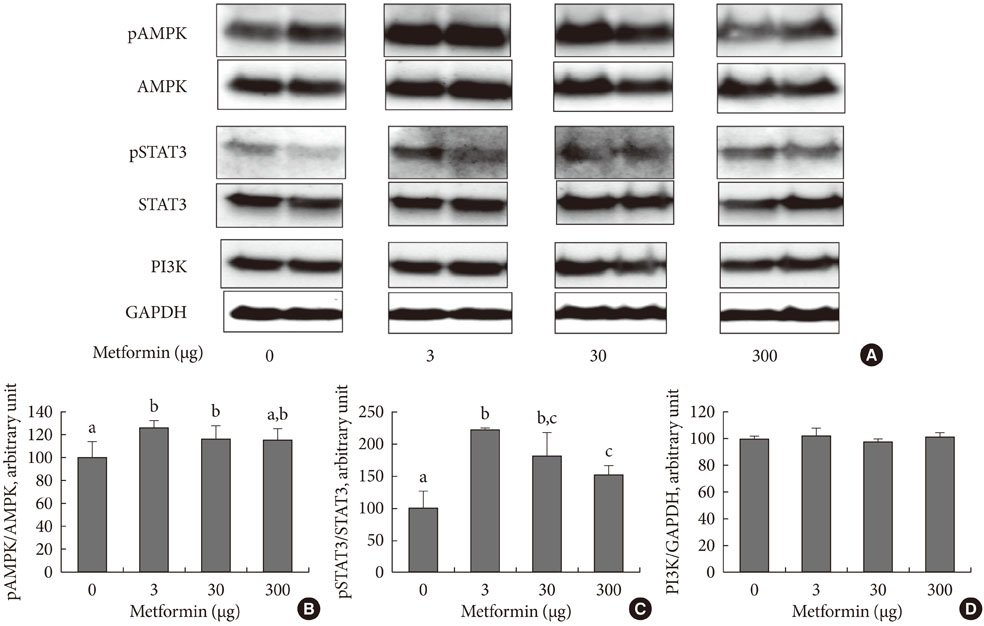
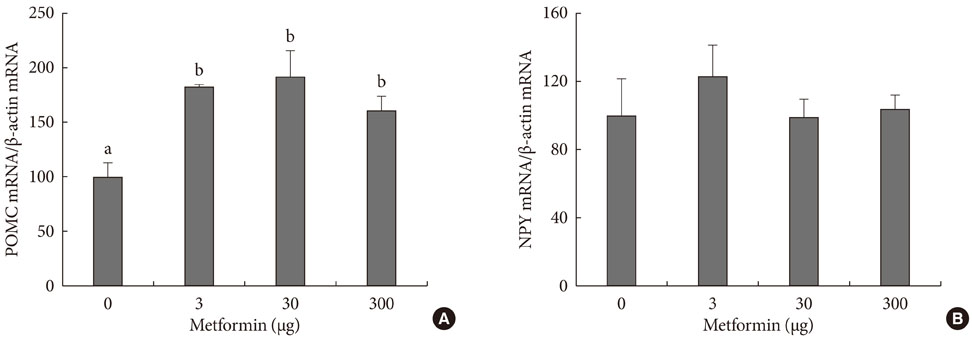
An ICV injection of metformin decreased food intake in a dose-dependent manner in unrestrained conscious rats. Hypothalamic phosphorylated AMP-activated protein kinase (pAMPK) increased by 3 microg with metformin treatment, but there was no further increase in pAMPK with increases in metformin dosage. The hypothalamic phosphorylated signal transducer and activator of transcription 3 (pSTAT3) increased by 3 microg with metformin treatment, but, there was no further increase in pSTAT3 level following increases of metformin dosage. Hypothalamic proopiomelanocortin was elevated with metformin treatment, while neuropeptide Y was not significantly changed.
CONCLUSION
Our results suggest that metformin induces anorexia via direct action in the hypothalamus and the increase in pSTAT3, at least in part, is involved in the process. However, hypothalamic pAMPK appears not to contribute to metformin-induced appetite reduction in normal rats. Further studies exploring new pathways connecting metformin and feeding regulation are needed.
Keyword
MeSH Terms
Figure
Reference
-
1. Morin-Papunen LC, Koivunen RM, Tomas C, Ruokonen A, Martikainen HK. Decreased serum leptin concentrations during metformin therapy in obese women with polycystic ovary syndrome. J Clin Endocrinol Metab. 1998. 83:2566–2568.2. Paolisso G, Amato L, Eccellente R, Gambardella A, Tagliamonte MR, Varricchio G, Carella C, Giugliano D, D'Onofrio F. Effect of metformin on food intake in obese subjects. Eur J Clin Invest. 1998. 28:441–446.3. Kay JP, Alemzadeh R, Langley G, D'Angelo L, Smith P, Holshouser S. Beneficial effects of metformin in normoglycemic morbidly obese adolescents. Metabolism. 2001. 50:1457–1461.4. Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N, Hirshman MF, Goodyear LJ, Moller DE. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest. 2001. 108:1167–1174.5. Hawley SA, Gadalla AE, Olsen GS, Hardie DG. The antidiabetic drug metformin activates the AMP-activated protein kinase cascade via an adenine nucleotide-independent mechanism. Diabetes. 2002. 51:2420–2425.6. Musi N, Hirshman MF, Nygren J, Svanfeldt M, Bavenholm P, Rooyackers O, Zhou G, Williamson JM, Ljunqvist O, Efendic S, Moller DE, Thorell A, Goodyear LJ. Metformin increases AMP-activated protein kinase activity in skeletal muscle of subjects with type 2 diabetes. Diabetes. 2002. 51:2074–2081.7. Xie Z, Dong Y, Scholz R, Neumann D, Zou MH. Phosphorylation of LKB1 at serine 428 by protein kinase C-zeta is required for metformin-enhanced activation of the AMP-activated protein kinase in endothelial cells. Circulation. 2008. 117:952–962.8. Kim YW, Kim JY, Park YH, Park SY, Won KC, Choi KH, Huh JY, Moon KH. Metformin restores leptin sensitivity in high-fat-fed obese rats with leptin resistance. Diabetes. 2006. 55:716–724.9. Billa E, Kapolla N, Nicopoulou SC, Koukkou E, Venaki E, Milingos S, Antsaklis A, Adamopoulos DA. Metformin administration was associated with a modification of LH, prolactin and insulin secretion dynamics in women with polycystic ovarian syndrome. Gynecol Endocrinol. 2009. 25:427–434.10. Vigersky RA, Filmore-Nassar A, Glass AR. Thyrotropin suppression by metformin. J Clin Endocrinol Metab. 2006. 91:225–227.11. Minokoshi Y, Alquier T, Furukawa N, Kim YB, Lee A, Xue B, Mu J, Foufelle F, Ferre P, Birnbaum MJ, Stuck BJ, Kahn BB. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature. 2004. 428:569–574.12. Chau-Van C, Gamba M, Salvi R, Gaillard RC, Pralong FP. Metformin inhibits adenosine 5'-monophosphate-activated kinase activation and prevents increases in neuropeptide Y expression in cultured hypothalamic neurons. Endocrinology. 2007. 148:507–511.13. Ropelle ER, Pauli JR, Zecchin KG, Ueno M, de Souza CT, Morari J, Faria MC, Velloso LA, Saad MJ, Carvalheira JB. A central role for neuronal adenosine 5'-monophosphate-activated protein kinase in cancer-induced anorexia. Endocrinology. 2007. 148:5220–5229.14. Borst SE, Snellen HG, Ross H, Scarpace PJ, Kim YW. Metformin restores responses to insulin but not to growth hormone in Sprague-Dawley rats. Biochem Biophys Res Commun. 2002. 291:722–726.15. Kim YW, Choi DW, Park YH, Huh JY, Won KC, Choi KH, Park SY, Kim JY, Lee SK. Leptin-like effects of MTII are augmented in MSG-obese rats. Regul Pept. 2005. 127:63–70.16. Andersson U, Filipsson K, Abbott CR, Woods A, Smith K, Bloom SR, Carling D, Small CJ. AMP-activated protein kinase plays a role in the control of food intake. J Biol Chem. 2004. 279:12005–12008.17. Lee K, Li B, Xi X, Suh Y, Martin RJ. Role of neuronal energy status in the regulation of adenosine 5'-monophosphate-activated protein kinase, orexigenic neuropeptides expression, and feeding behavior. Endocrinology. 2005. 146:3–10.18. Kim EK, Miller I, Aja S, Landree LE, Pinn M, McFadden J, Kuhajda FP, Moran TH, Ronnett GV. C75, a fatty acid synthase inhibitor, reduces food intake via hypothalamic AMP-activated protein kinase. J Biol Chem. 2004. 279:19970–19976.19. Kim MS, Park JY, Namkoong C, Jang PG, Ryu JW, Song HS, Yun JY, Namgoong IS, Ha J, Park IS, Lee IK, Viollet B, Youn JH, Lee HK, Lee KU. Anti-obesity effects of alpha-lipoic acid mediated by suppression of hypothalamic AMP-activated protein kinase. Nat Med. 2004. 10:727–733.20. Shek EW, Scarpace PJ. Resistance to the anorexic and thermogenic effects of centrally administrated leptin in obese aged rats. Regul Pept. 2000. 92:65–71.21. Scarpace PJ, Matheny M, Tumer N. Hypothalamic leptin resistance is associated with impaired leptin signal transduction in aged obese rats. Neuroscience. 2001. 104:1111–1117.22. Hill JW, Williams KW, Ye C, Luo J, Balthasar N, Coppari R, Cowley MA, Cantley LC, Lowell BB, Elmquist JK. Acute effects of leptin require PI3K signaling in hypothalamic proopiomelanocortin neurons in mice. J Clin Invest. 2008. 118:1796–1805.23. Hill JW, Xu Y, Preitner F, Fukuda M, Cho YR, Luo J, Balthasar N, Coppari R, Cantley LC, Kahn BB, Zhao JJ, Elmquist JK. Phosphatidyl inositol 3-kinase signaling in hypothalamic proopiomelanocortin neurons contributes to the regulation of glucose homeostasis. Endocrinology. 2009. 150:4874–4882.24. Scarpace PJ, Matheny M, Tumer N, Cheng KY, Zhang Y. Leptin resistance exacerbates diet-induced obesity and is associated with diminished maximal leptin signalling capacity in rats. Diabetologia. 2005. 48:1075–1083.25. Rouru J, Pesonen U, Koulu M, Huupponen R, Santti E, Virtanen K, Rouvari T, Jhanwar-Uniyal M. Anorectic effect of metformin in obese Zucker rats: lack of evidence for the involvement of neuropeptide Y. Eur J Pharmacol. 1995. 273:99–106.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of pretreatment of serotonin synthesis inhibitor p-chlorophenylalanine on lipopolysaccharide-induced anorexia in rats
- Metformin Enhances Leptin Sensitivity in Aged Rats
- Successful Treatment of Anorexia Nervosa in a 10-year-old Boy with Risperidone Long-acting Injection
- The Effect of Clozapine on Central Insulin Response in Rats
- The Effect of Intracerebroventricular Gabapentin on the Formalin Test in Rats