Blood Res.
2013 Jun;48(2):107-114. 10.5045/br.2013.48.2.107.
Human bone marrow-derived mesenchymal stem cell gene expression patterns vary with culture conditions
- Affiliations
-
- 1Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. hhkoo@skku.edu, hema2170@skku.edu
- KMID: 2172908
- DOI: http://doi.org/10.5045/br.2013.48.2.107
Abstract
- BACKGROUND
Because of the heterogeneity of human mesenchymal stem cells (MSCs), methods for cell expansion in culture and the effects on gene expression are critical factors that need to be standardized for preparing MSCs. We investigated gene expression patterns of MSCs with different seeding densities and culture times.
METHODS
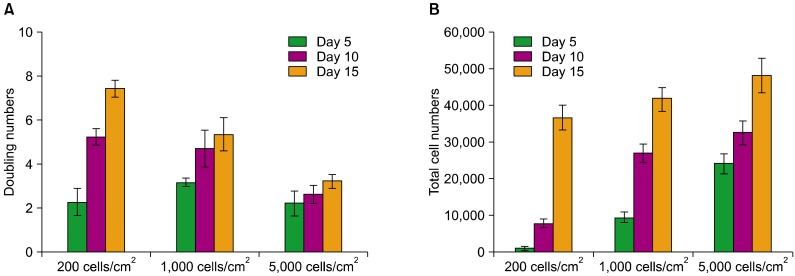
Bone marrow-derived MSCs were plated at densities from 200 cells/cm2 to 5,000 cells/cm2, and the gene expression patterns were evaluated over time using a reverse-transcription polymerase chain reaction assay.
RESULTS
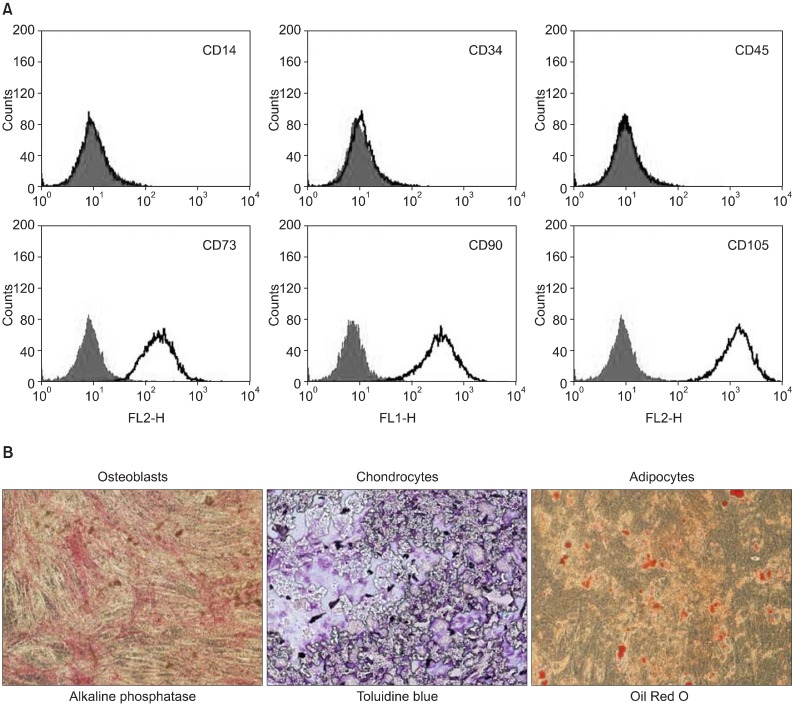
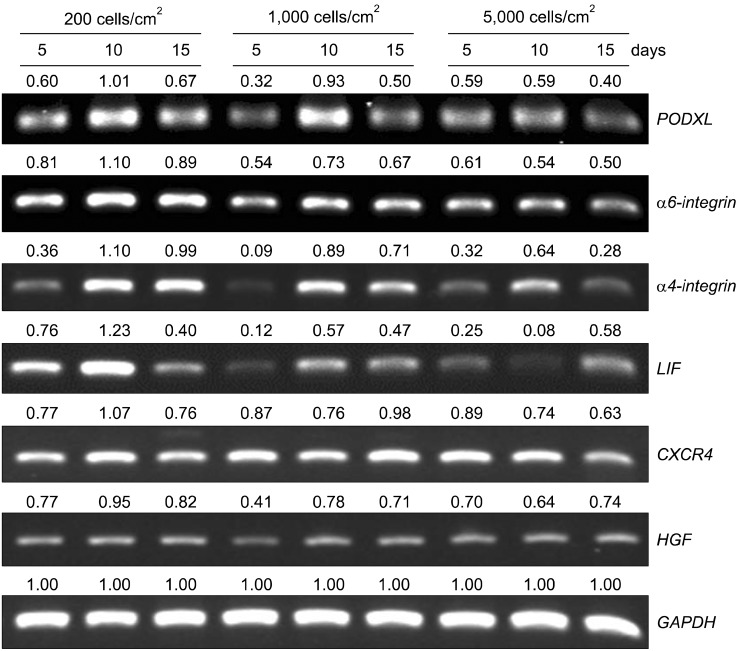
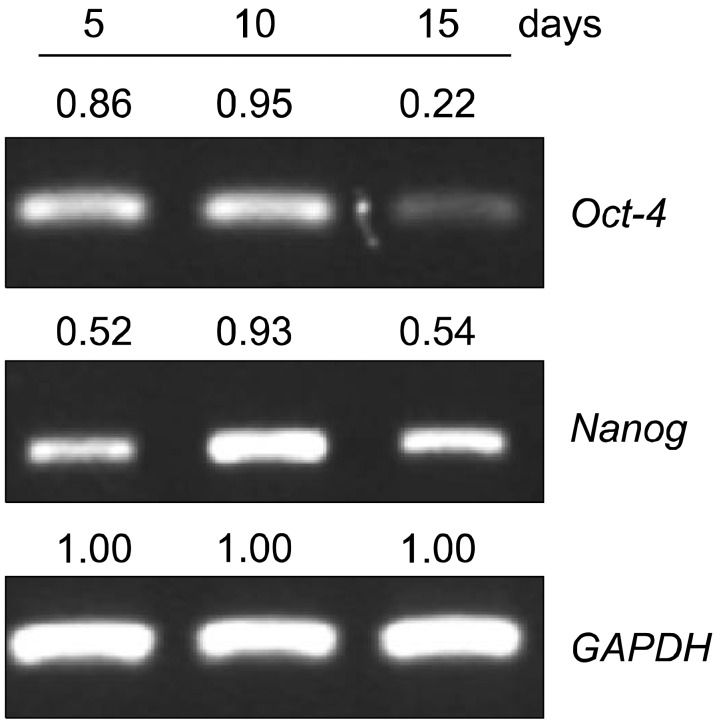
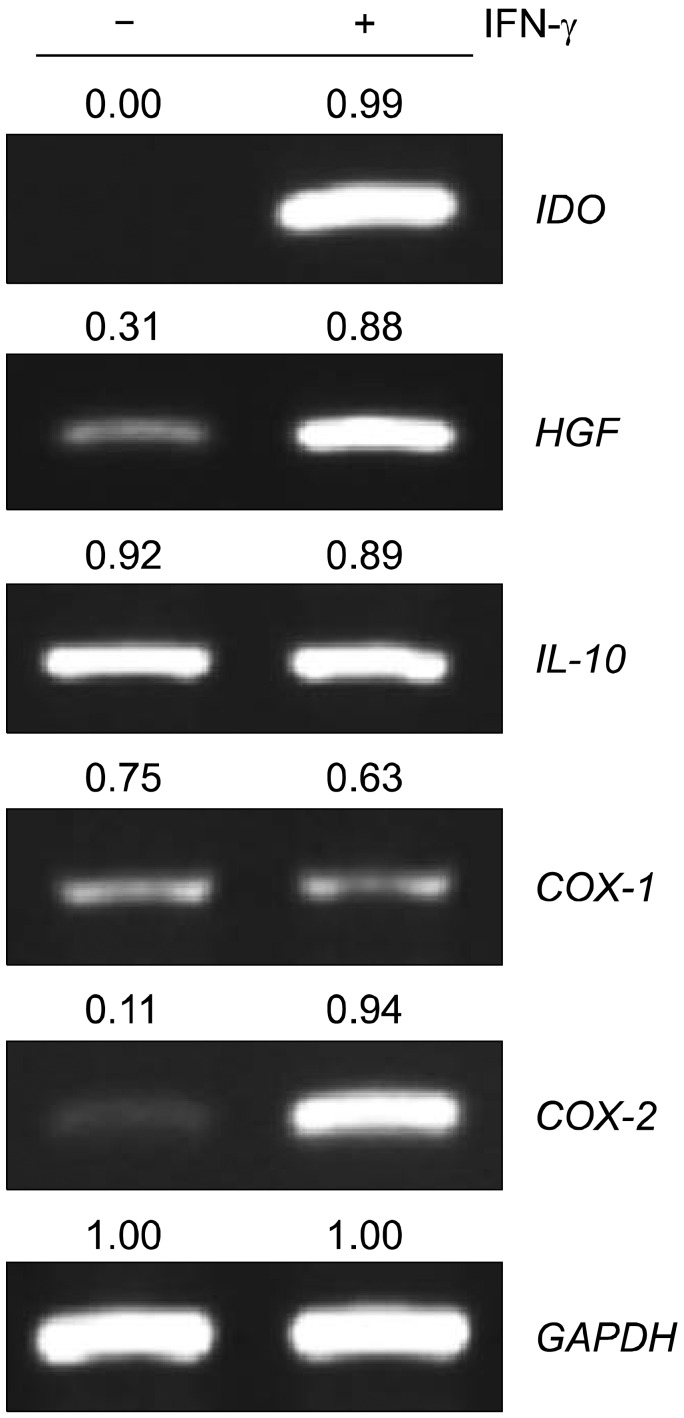
The mRNA levels of factors that play a critical role in cell migration and tissue regeneration, such as podocalyxin-like protein (PODXL), alpha4-integrin, alpha6-integrin, and leukemia inhibitory factor (LIF), were higher in MSCs plated at 200 cells/cm2 than in MSCs plated at 5,000 cells/cm2. The mRNA levels of these factors gradually increased for 10 days and then decreased by day 15 in culture. MSCs seeded at 200 cells/cm2 that were cultured for 10 days expressed high levels of Oct-4 and Nanog. Indoleamine 2,3-dioxygenase, cyclooxygenase-1, and hepatocyte growth factor expression were upregulated in the presence of the proinflammatory cytokine interferon-gamma in these cells.
CONCLUSION
We found differences in the gene expression patterns of MSCs under different culture conditions. MSCs from 10-day cultures seeded at a low density were efficiently expanded, expressed PODXL, alpha6-integrin, alpha4-integrin, and LIF, and maintained properties like stemness and immunomodulation. Therefore, ex vivo expansion of MSCs maintained for an adequate culture time after plating at low cell density can provide an effective regenerative medicinal strategy for cell therapies using MSCs.
MeSH Terms
-
Cell Count
Cell Movement
Cyclooxygenase 1
Gene Expression
Hepatocyte Growth Factor
Humans
Immunomodulation
Indoleamine-Pyrrole 2,3,-Dioxygenase
Interferon-gamma
Leukemia Inhibitory Factor
Mesenchymal Stromal Cells
Polymerase Chain Reaction
Population Characteristics
Regeneration
RNA, Messenger
Seeds
Tissue Therapy
Cyclooxygenase 1
Hepatocyte Growth Factor
Indoleamine-Pyrrole 2,3,-Dioxygenase
Interferon-gamma
Leukemia Inhibitory Factor
RNA, Messenger
Figure
Reference
-
1. Sasaki M, Abe R, Fujita Y, Ando S, Inokuma D, Shimizu H. Mesenchymal stem cells are recruited into wounded skin and contribute to wound repair by transdifferentiation into multiple skin cell type. J Immunol. 2008; 180:2581–2587. PMID: 18250469.
Article2. Toma C, Pittenger MF, Cahill KS, Byrne BJ, Kessler PD. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation. 2002; 105:93–98. PMID: 11772882.
Article3. Keyser KA, Beagles KE, Kiem HP. Comparison of mesenchymal stem cells from different tissues to suppress T-cell activation. Cell Transplant. 2007; 16:555–562. PMID: 17708345.
Article4. Nauta AJ, Fibbe WE. Immunomodulatory properties of mesenchymal stromal cells. Blood. 2007; 110:3499–3506. PMID: 17664353.
Article5. Lee RH, Seo MJ, Pulin AA, Gregory CA, Ylostalo J, Prockop DJ. The CD34-like protein PODXL and alpha6-integrin (CD49f) identify early progenitor MSCs with increased clonogenicity and migration to infarcted heart in mice. Blood. 2009; 113:816–826. PMID: 18818395.6. Lee RH, Hsu SC, Munoz J, et al. A subset of human rapidly self-renewing marrow stromal cells preferentially engraft in mice. Blood. 2006; 107:2153–2161. PMID: 16278305.
Article7. Colter DC, Sekiya I, Prockop DJ. Identification of a subpopulation of rapidly self-renewing and multipotential adult stem cells in colonies of human marrow stromal cells. Proc Natl Acad Sci U S A. 2001; 98:7841–7845. PMID: 11427725.
Article8. Digirolamo CM, Stokes D, Colter D, Phinney DG, Class R, Prockop DJ. Propagation and senescence of human marrow stromal cells in culture: a simple colony-forming assay identifies samples with the greatest potential to propagate and differentiate. Br J Haematol. 1999; 107:275–281. PMID: 10583212.
Article9. Smith JR, Pochampally R, Perry A, Hsu SC, Prockop DJ. Isolation of a highly clonogenic and multipotential subfraction of adult stem cells from bone marrow stroma. Stem Cells. 2004; 22:823–831. PMID: 15342946.
Article10. Ho AD, Wagner W, Franke W. Heterogeneity of mesenchymal stromal cell preparations. Cytotherapy. 2008; 10:320–330. PMID: 18574765.
Article11. Reyes M, Lund T, Lenvik T, Aguiar D, Koodie L, Verfaillie CM. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood. 2001; 98:2615–2625. PMID: 11675329.12. Sotiropoulou PA, Perez SA, Salagianni M, Baxevanis CN, Papamichail M. Characterization of the optimal culture conditions for clinical scale production of human mesenchymal stem cells. Stem Cells. 2006; 24:462–471. PMID: 16109759.
Article13. Qian H, Tryggvason K, Jacobsen SE, Ekblom M. Contribution of alpha6 integrins to hematopoietic stem and progenitor cell homing to bone marrow and collaboration with alpha4 integrins. Blood. 2006; 107:3503–3510. PMID: 16439681.14. Patterson BK, Tjernlund A, Andersson J. Endogenous inhibitors of HIV: potent anti-HIV activity of leukemia inhibitory factor. Curr Mol Med. 2002; 2:713–722. PMID: 12462392.
Article15. Pruijt JF, Lindley IJ, Heemskerk DP, Willemze R, Fibbe WE. Leukemia inhibitory factor induces in vivo expansion of bone marrow progenitor cells that accelerate hematopoietic reconstitution but do not enhance radioprotection in lethally irradiated mice. Stem Cells. 1997; 15:50–55. PMID: 9007222.
Article16. Almeida-Porada G, Flake AW, Glimp HA, Zanjani ED. Cotransplantation of stroma results in enhancement of engraftment and early expression of donor hematopoietic stem cells in utero. Exp Hematol. 1999; 27:1569–1575. PMID: 10517499.
Article17. Furness SG, McNagny K. Beyond mere markers: functions for CD34 family of sialomucins in hematopoiesis. Immunol Res. 2006; 34:13–32. PMID: 16720896.
Article18. Nasef A, Mazurier C, Bouchet S, et al. Leukemia inhibitory factor: Role in human mesenchymal stem cells mediated immunosuppression. Cell Immunol. 2008; 253:16–22. PMID: 18639869.
Article19. O'Shea JJ, Ma A, Lipsky P. Cytokines and autoimmunity. Nat Rev Immunol. 2002; 2:37–45. PMID: 11905836.20. Yoo KH, Jang IK, Lee MW, et al. Comparison of immunomodulatory properties of mesenchymal stem cells derived from adult human tissues. Cell Immunol. 2009; 259:150–156. PMID: 19608159.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Difference in Cell Characteristics among the Monoclonal Cell Populations Obtained from the Human Umbilical Cord Blood Derived Mesenchymal Stem Cell Population
- Concise Review: Differentiation of Human Adult Stem Cells Into Hepatocyte-like Cells In vitro
- Clinical Safety and Efficacy of Autologous Bone Marrow-Derived Mesenchymal Stem Cell Transplantation in Sensorineural Hearing Loss Patients
- Gene expression profile in mesenchymal stem cells derived from dental tissues and bone marrow
- Temporal Patterns of Expression of Type II Procollagen Splice Forms During the Chondrogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stem Cells