Anat Cell Biol.
2010 Sep;43(3):179-184. 10.5115/acb.2010.43.3.179.
PINK1 and Parkin to control mitochondria remodeling
- Affiliations
-
- 1Department of Pharmacology, Mitochondria Hub Regulation Center (MHRC), Dong-A University College of Medicine, Busan, Korea. hjkoh@dau.ac.kr
- 2School of Biological Sciences, Seoul National University, Seoul, Korea.
- KMID: 2168872
- DOI: http://doi.org/10.5115/acb.2010.43.3.179
Abstract
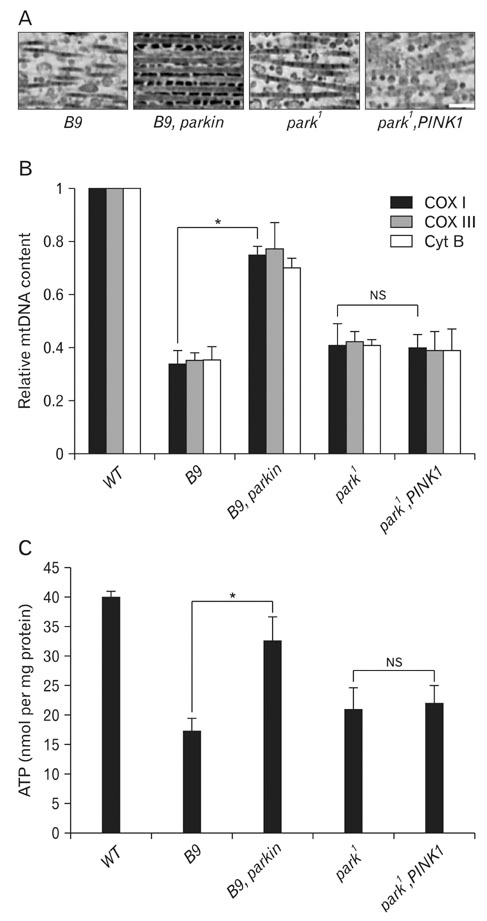
- Parkinson's disease (PD), one of the most common neurodegenerative diseases, is characterized by movement disorders and a loss of dopaminergic (DA) neurons. PD mainly occurs sporadically, but may also result from genetic mutations in several PD-linked genes. Recently, genetic studies with Drosophila mutants, parkin and PINK1, two common PD-associated genes, demonstrated that Parkin acts downstream of PINK1 in maintaining mitochondrial function and integrity. Further studies revealed that PINK1 translocates Parkin to mitochondria and regulates critical mitochondrial remodeling processes. These findings, which suggest that mitochondrial dysfunction is a prominent cause of PD pathogenesis, provide valuable insights which may aid in the development of effective treatments for PD.
Keyword
MeSH Terms
Figure
Reference
-
1. Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT.. Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat Neurosci. 2000. 3:1301–1306.2. Bonifati V, Rizzu P, van Baren MJ, et al. Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science. 2003. 299:256–259.3. Cha GH, Kim S, Park J, et al. Parkin negatively regulates JNK pathway in the dopaminergic neurons of Drosophila. Proc Natl Acad Sci U S A. 2005. 102:10345–10350.4. Chan DC. Mitochondrial fusion and fission in mammals. Annu Rev Cell Dev Biol. 2006. 22:79–99.5. Cicchetti F, Lapointe N, Roberge-Tremblay A, et al. Systemic exposure to paraquat and maneb models early Parkinson's disease in young adult rats. Neurobiol Dis. 2005. 20:360–371.6. Clark IE, Dodson MW, Jiang C, et al. Drosophila pink1 is required for mitochondrial function and interacts genetically with parkin. Nature. 2006. 441:1162–1166.7. Coulom H, Birman S. Chronic exposure to rotenone models sporadic Parkinson's disease in Drosophila melanogaster. J Neurosci. 2004. 24:10993–10998.8. Deng H, Dodson MW, Huang H, Guo M. The Parkinson's disease genes pink1 and parkin promote mitochondrial fission and/or inhibit fusion in Drosophila. Proc Natl Acad Sci U S A. 2008. 105:14503–14508.9. Exner N, Treske B, Paquet D, et al. Loss-of-function of human PINK1 results in mitochondrial pathology and can be rescued by parkin. J Neurosci. 2007. 27:12413–12418.10. Geisler S, Holmström KM, Skujat D, et al. PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat Cell Biol. 2010. 12:119–231.11. Greene JC, Whitworth AJ, Kuo I, Andrews LA, Feany MB, Pallanck LJ.. Mitochondrial pathology and apoptotic muscle degeneration in Drosophila parkin mutants. Proc Natl Acad Sci U S A. 2003. 100:4078–4083.12. Henchcliffe C, Beal MF. Mitochondrial biology and oxidative stress in Parkinson disease pathogenesis. Nat Clin Pract Neurol. 2008. 4:600–609.13. Imai Y, Soda M, Inoue H, Hattori N, Mizuno Y, Takahashi R. An unfolded putative transmembrane polypeptide, which can lead to endoplasmic reticulum stress, is a substrate of Parkin. Cell. 2001. 105:891–902.14. Keeney PM, Xie J, Capaldi RA, Bennett JP Jr. Parkinson's disease brain mitochondrial complex I has oxidatively damaged subunits and is functionally impaired and misassembled. J Neurosci. 2006. 26:5256–5264.15. Kim Y, Park J, Kim S, et al. PINK1 controls mitochondrial localization of Parkin through direct phosphorylation. Biochem Biophys Res Commun. 2008. 377:975–980.16. Kitada T, Asakawa S, Hattori N, et al. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature. 1998. 392:605–608.17. Klein C, Lohmann-Hedrich K. Impact of recent genetic findings in Parkinson's disease. Curr Opin Neurol. 2007. 20:453–464.18. Lang AE, Lozano AM. Parkinson's disease. First of two parts. N Engl J Med. 1998. 339:1044–1053.19. Langston JW, Ballard P, Tetrud JW, Irwin I. Chronic Parkinsonism in humans due to a product of meperidine-analog synthesis. Science. 1983. 219:979–980.20. Lee JY, Nagano Y, Taylor JP, Lim KL, Yao TP. Disease-causing mutations in parkin impair mitochondrial ubiquitination, aggregation, and HDAC6-dependent mitophagy. J Cell Biol. 2010. 189:671–679.21. Matsuda N, Sato S, Shiba K, et al. PINK1 stabilized by mitochondrial depolarization recruits Parkin to damaged mitochondria and activates latent Parkin for mitophagy. J Cell Biol. 2010. 189:211–221.22. Narendra D, Tanaka A, Suen DF, Youle RJ. Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J Cell Biol. 2008. 183:795–803.23. Narendra DP, Jin SM, Tanaka A, et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol. 2010. 8:e1000298.24. Paisán-Ruíz C, Jain S, Evans EW, et al. Cloning of the gene containing mutations that cause PARK8-linked Parkinson's disease. Neuron. 2004. 44:595–600.25. Park J, Lee G, Chung J. The PINK1-Parkin pathway is involved in the regulation of mitochondrial remodeling process. Biochem Biophys Res Commun. 2009. 378:518–523.26. Park J, Lee SB, Lee S, et al. Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by parkin. Nature. 2006. 441:1157–1161.27. Parker WD Jr, Parks JK, Swerdlow RH.. Complex I deficiency in Parkinson's disease frontal cortex. Brain Res. 2008. 1189:215–218.28. Pesah Y, Pham T, Burgess H, et al. Drosophila parkin mutants have decreased mass and cell size and increased sensitivity to oxygen radical stress. Development. 2004. 131:2183–2194.29. Polymeropoulos MH, Lavedan C, Leroy E, et al. Mutation in the alpha-synuclein gene identified in families with Parkinson's disease. Science. 1997. 276:2045–2047.30. Poole AC, Thomas RE, Andrews LA, McBride HM, Whitworth AJ, Pallanck LJ. The PINK1/Parkin pathway regulates mitochondrial morphology. Proc Natl Acad Sci U S A. 2008. 105:1638–1643.31. Ramirez A, Heimbach A, Grndemann J, et al. Hereditary parkinsonism with dementia is caused by mutations in ATP13A2, encoding a lysosomal type 5 P-type ATPase. Nat Genet. 2006. 38:1184–1191.32. Shimura H, Hattori N, Kubo S, et al. Familial Parkinson disease gene product, parkin, is a ubiquitin-protein ligase. Nat Genet. 2000. 25:302–305.33. Staropoli JF, McDermott C, Martinat C, Schulman B, Demireva E, Abeliovich A. Parkin is a component of an SCF-like ubiquitin ligase complex and protects postmitotic neurons from kainate excitotoxicity. Neuron. 2003. 37:735–749.34. Valente EM, Abou-Sleiman PM, Caputo V, et al. Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science. 2004. 304:1158–1160.35. Vives-Bauza C, Zhou C, Huang Y, et al. PINK1-dependent recruitment of Parkin to mitochondria in mitophagy. Proc Natl Acad Sci U S A. 2010. 107:378–383.36. Whitworth AJ, Pallanck LJ. The PINK1/Parkin pathway: a mitochondrial quality control system? J Bioenerg Biomembr. 2009. 41:499–503.37. Wood-Kaczmar A, Gandhi S, Yao Z, et al. PINK1 is necessary for long term survival and mitochondrial function in human dopaminergic neurons. PLoS One. 2008. 3:e2455.38. Yang Y, Gehrke S, Imai Y, et al. Mitochondrial pathology and muscle and dopaminergic neuron degeneration caused by inactivation of Drosophila Pink1 is rescued by Parkin. Proc Natl Acad Sci U S A. 2006. 103:10793–10798.39. Yang Y, Ouyang Y, Yang L, et al. Pink1 regulates mitochondrial dynamics through interaction with the fission/fusion machinery. Proc Natl Acad Sci U S A. 2008. 105:7070–7075.40. Zhang Y, Gao J, Chung KK, Huang H, Dawson VL, Dawson TM. Parkin functions as an E2-dependent ubiquitin- protein ligase and promotes the degradation of the synaptic vesicle-associated protein, CDCrel-1. Proc Natl Acad Sci U S A. 2000. 97:13354–13359.41. Ziviani E, Tao RN, Whitworth AJ. Drosophila parkin requires PINK1 for mitochondrial translocation and ubiquitinates mitofusin. Proc Natl Acad Sci U S A. 2010. 107:5018–5023.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Mitochondrial Homeostasis Molecules: Regulation by a Trio of Recessive Parkinson's Disease Genes
- Dysfunctional Mitochondria Clearance in Situ: Mitophagy in Obesity and Diabetes-Associated Cardiometabolic Diseases
- The Emerging Importance of Mitochondria in White Adipocytes: Neither Last nor Least
- Clinico-Genetic Profiles of Seven Patients With PINK1-Related Parkinson’s Disease: A Case Series From a Tertiary Care Centre in India and a Review of the Literature
- Role of PTEN-Induced Protein Kinase 1 as a Mitochondrial Dysfunction Regulator in Cardiovascular Disease Pathogenesis