Allergy Asthma Immunol Res.
2016 Jul;8(4):362-374. 10.4168/aair.2016.8.4.362.
House Dust Mite-Derived Chitin Enhances Th2 Cell Response to Inhaled Allergens, Mainly via a TNF-α-Dependent Pathway
- Affiliations
-
- 1Institute of Convergence Medicine, Ewha Womans University School of Medicine and Medical Center, Seoul, Korea. juinea@ewha.ac.kr
- 2Department of Internal Medicine, Gacheon University College of Medicine, Incheon, Korea.
- 3Department of Life Sciences, Pohang University of Science and Technology (POSTECH), Pohang, Korea.
- 4Academy of Immunology and Microbiology (AIM), Institute for Basic Science (IBS), Pohang, Korea.
- 5Department of Internal Medicine, Dankook University College of Medicine, Cheonan, Korea.
- KMID: 2165920
- DOI: http://doi.org/10.4168/aair.2016.8.4.362
Abstract
- PURPOSE
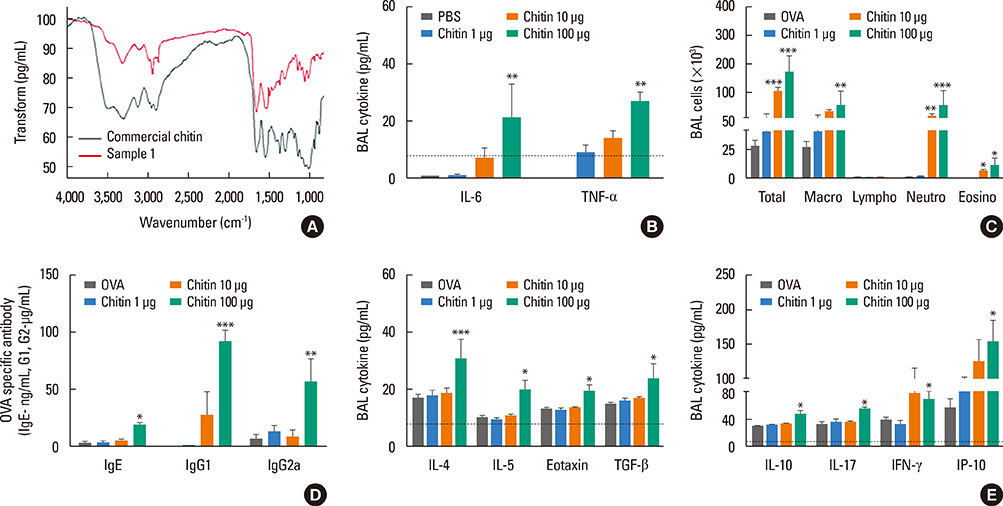
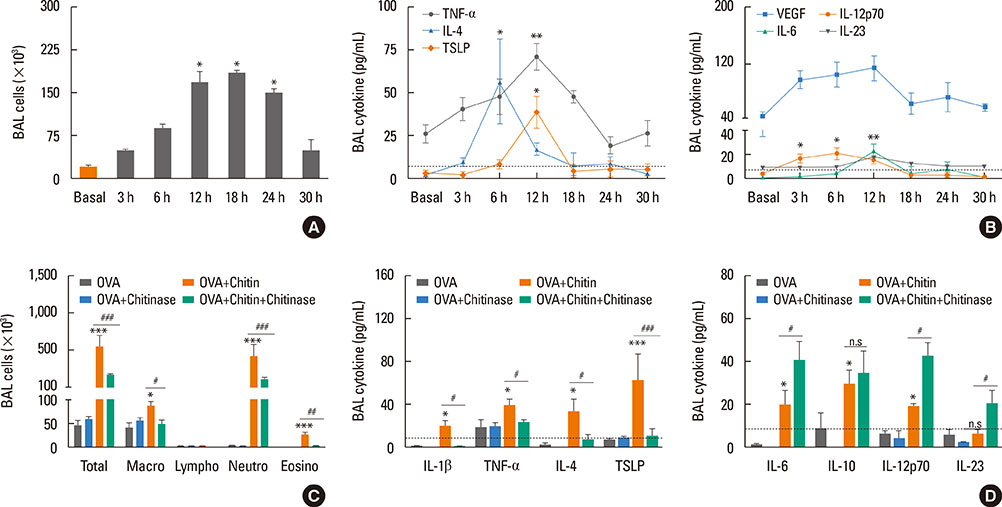
Chitin is a potent adjuvant in the development of immune response to inhaled allergens in the airways. According to other studies, chitin is known as multi-faced adjuvants which can induce Th2 responses. Recently, we found that TNF-α is a key mediator in the development of Th2 cell response to inhaled allergens. Here, we evaluated the immunologic mechanisms in the development of airway hypersensitivity to inhaled allergens, enhanced by house dust mite (HDM)-derived chitin.
METHODS
The role of TNF-α and TLRs was evaluated in an airway hypersensitivity mouse model induced by a sensitization with an allergen (ovalbumin, OVA) and HDM-derived chitin using mice with the null mutation of target genes.
RESULTS
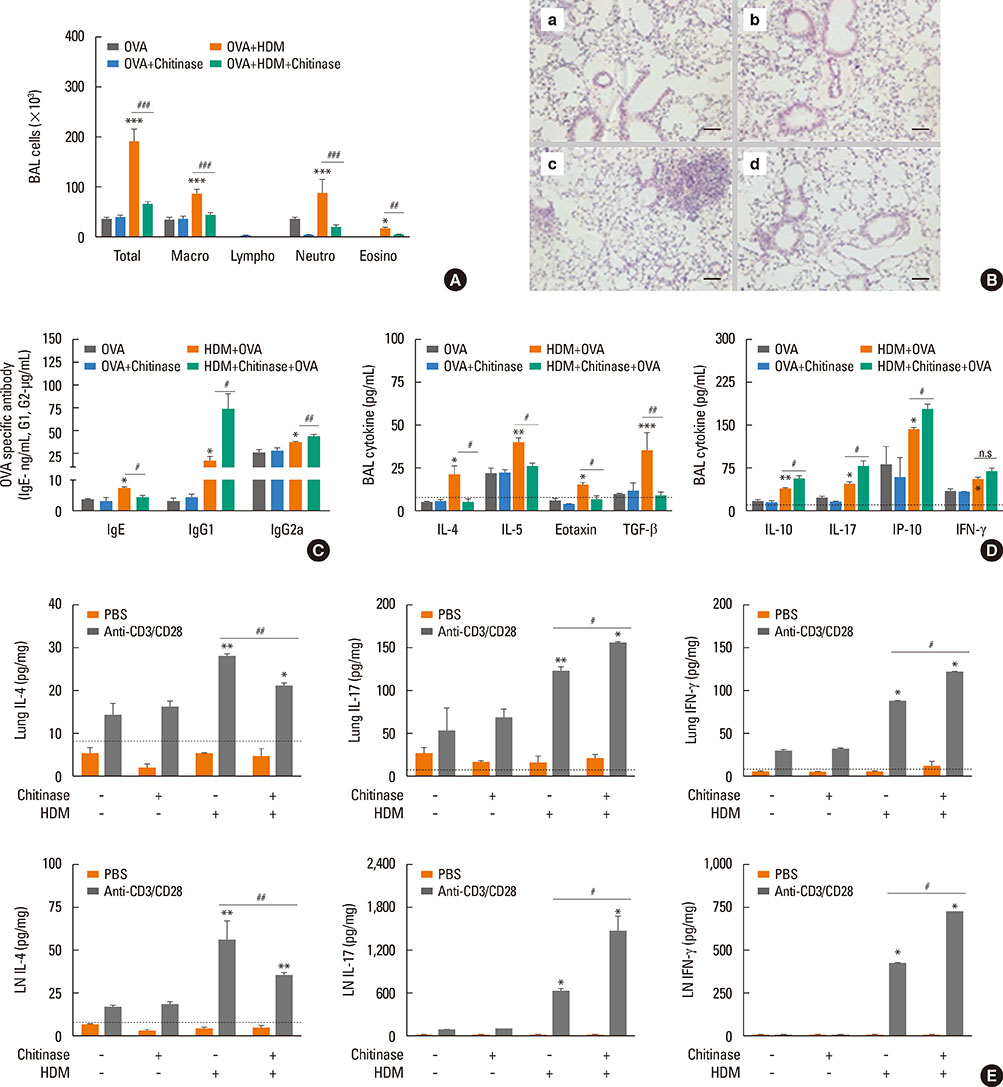
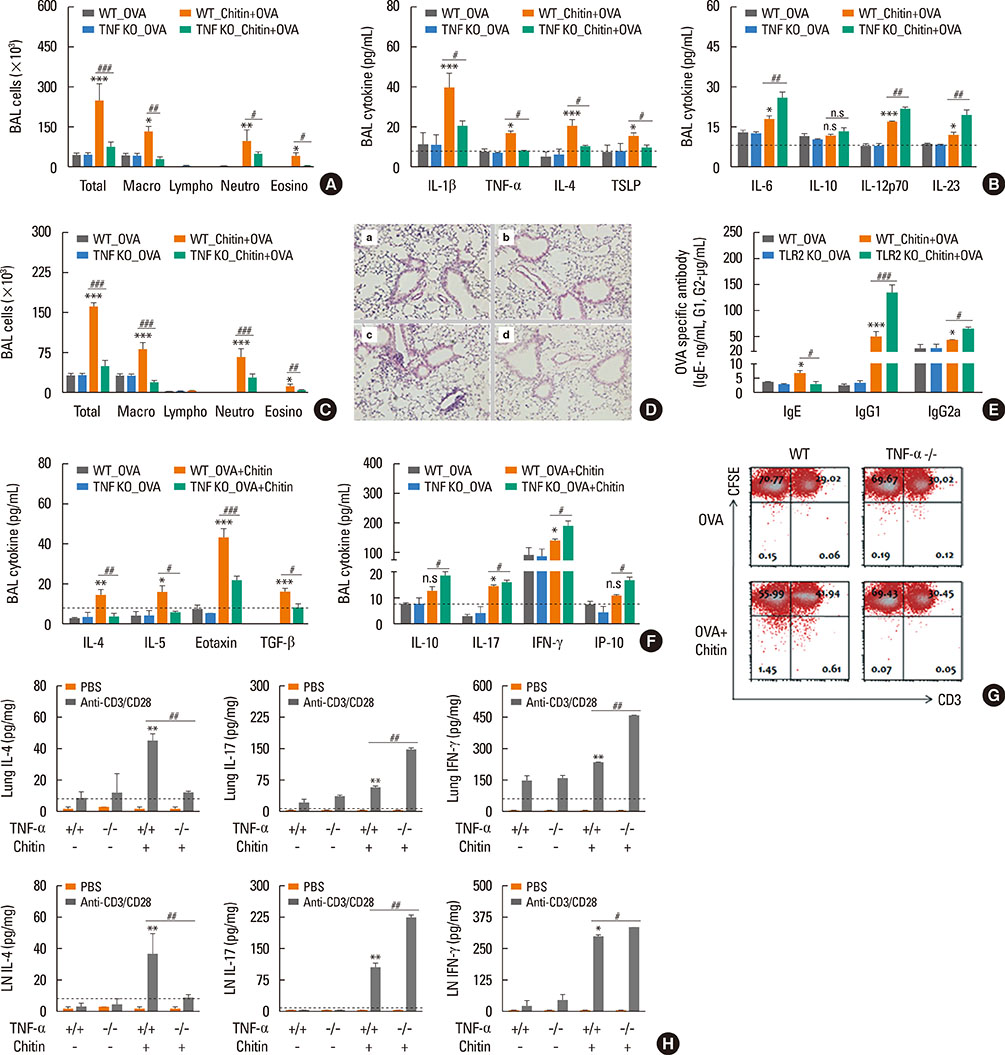
The present study showed that airway sensitization with HDM-derived chitin plus OVA enhanced OVA-induced airway inflammation v. OVA alone. This phenotype was associated with the increased expression of Th1, Th2, and Th17 cytokines and also with the enhanced production of OVA-specific IgE, IgG1, and IgG2a. As for T cell responses, OVA-specific Th2 cell response, enhanced by chitin, was abolished by the treatment of chitinase, whereas Th1 and Th17 cell responses enhanced by this treatment. Moreover, the null mutation of the TNF-α gene revealed similar effects as the chitinase treatment. In contrast, all the OVA-specific T cell responses, enhanced by chitin, were blocked by the absence of TLR2, but not of TLR1, TLR4, or TLR6.
CONCLUSIONS
In conclusion, these data suggest that HDM-derived chitin may enhance airway hypersensitivity to inhaled allergens, via the TLR2-dependent pathway, and that chitin-induced TNF-α can be a key mediator in the development of Th2 cell response to inhaled allergens.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Bcl11b Regulates IL-17 Through the TGF-β/Smad Pathway in HDM-Induced Asthma
Si Chen, Yuting Han, Hao Chen, Jing Wu, Min Zhang
Allergy Asthma Immunol Res. 2018;10(5):543-554. doi: 10.4168/aair.2018.10.5.543.
Reference
-
1. Dowse GK, Turner KJ, Stewart GA, Alpers MP, Woolcock AJ. The association between Dermatophagoides mites and the increasing prevalence of asthma in village communities within the Papua New Guinea highlands. J Allergy Clin Immunol. 1985; 75:75–83.2. Charpin D, Birnbaum J, Haddi E, Genard G, Lanteaume A, Toumi M, et al. Altitude and allergy to house-dust mites. A paradigm of the influence of environmental exposure on allergic sensitization. Am Rev Respir Dis. 1991; 143:983–986.3. Platts-Mills TA, Vervloet D, Thomas WR, Aalberse RC, Chapman MD. Indoor allergens and asthma: report of the Third International Workshop. J Allergy Clin Immunol. 1997; 100:S2–S24.4. Neville AC, Parry DA, Woodhead-Galloway J. The chitin crystallite in arthropod cuticle. J Cell Sci. 1976; 21:73–82.5. Fuhrman JA, Piessens WF. Chitin synthesis and sheath morphogenesis in Brugia malayi microfilariae. Mol Biochem Parasitol. 1985; 17:93–104.6. Araujo AC, Souto-Padrón T, de Souza W. Cytochemical localization of carbohydrate residues in microfilariae of Wuchereria bancrofti and Brugia malayi. J Histochem Cytochem. 1993; 41:571–578.7. Debono M, Gordee RS. Antibiotics that inhibit fungal cell wall development. Annu Rev Microbiol. 1994; 48:471–497.8. Jeong KY, Park JW, Hong CS. House dust mite allergy in Korea: the most important inhalant allergen in current and future. Allergy Asthma Immunol Res. 2012; 4:313–325.9. Gao YF, Wang de Y, Ong TC, Tay SL, Yap KH, Chew FT. Identification and characterization of a novel allergen from Blomia tropicalis: Blo t 21. J Allergy Clin Immunol. 2007; 120:105–112.10. Da Silva CA, Pochard P, Lee CG, Elias JA. Chitin particles are multifaceted immune adjuvants. Am J Respir Crit Care Med. 2010; 182:1482–1491.11. Shibata Y, Foster LA, Bradfield JF, Myrvik QN. Oral administration of chitin down-regulates serum IgE levels and lung eosinophilia in the allergic mouse. J Immunol. 2000; 164:1314–1321.12. Strong P, Clark H, Reid K. Intranasal application of chitin microparticles down-regulates symptoms of allergic hypersensitivity to Dermatophagoides pteronyssinus and Aspergillus fumigatus in murine models of allergy. Clin Exp Allergy. 2002; 32:1794–1800.13. Medzhitov R, Janeway C Jr. Innate immunity. N Engl J Med. 2000; 343:338–344.14. Duez C, Gosset P, Tonnel AB. Dendritic cells and toll-like receptors in allergy and asthma. Eur J Dermatol. 2006; 16:12–16.15. Kim YK, Oh SY, Jeon SG, Park HW, Lee SY, Chun EY, et al. Airway exposure levels of lipopolysaccharide determine type 1 versus type 2 experimental asthma. J Immunol. 2007; 178:5375–5382.16. Choi JP, Kim YS, Kim OY, Kim YM, Jeon SG, Roh TY, et al. TNF-alpha is a key mediator in the development of Th2 cell response to inhaled allergens induced by a viral PAMP double-stranded RNA. Allergy. 2012; 67:1138–1148.17. Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010; 11:373–384.18. Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006; 124:783–801.19. Choi JP, Kim YS, Tae YM, Choi EJ, Hong BS, Jeon SG, et al. A viral PAMP double-stranded RNA induces allergen-specific Th17 cell response in the airways which is dependent on VEGF and IL-6. Allergy. 2010; 65:1322–1330.20. Jeon SG, Oh SY, Park HK, Kim YS, Shim EJ, Lee HS, et al. TH2 and TH1 lung inflammation induced by airway allergen sensitization with low and high doses of double-stranded RNA. J Allergy Clin Immunol. 2007; 120:803–812.21. Fuchs B, Braun A. Improved mouse models of allergy and allergic asthma--chances beyond ovalbumin. Curr Drug Targets. 2008; 9:495–502.22. Kumar RK, Herbert C, Foster PS. The "classical" ovalbumin challenge model of asthma in mice. Curr Drug Targets. 2008; 9:485–494.23. Yamashita M, Nakayama T. Progress in allergy signal research on mast cells: regulation of allergic airway inflammation through toll-like receptor 4-mediated modification of mast cell function. J Pharmacol Sci. 2008; 106:332–335.24. Fattouh R, Pouladi MA, Alvarez D, Johnson JR, Walker TD, Goncharova S, et al. House dust mite facilitates ovalbumin-specific allergic sensitization and airway inflammation. Am J Respir Crit Care Med. 2005; 172:314–321.25. Shuhui L, Mok YK, Wong WS. Role of mammalian chitinases in asthma. Int Arch Allergy Immunol. 2009; 149:369–377.26. Reese TA, Liang HE, Tager AM, Luster AD, Van Rooijen N, Voehringer D, et al. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature. 2007; 447:92–96.27. Shibata Y, Foster LA, Metzger WJ, Myrvik QN. Alveolar macrophage priming by intravenous administration of chitin particles, polymers of N-acetyl-D-glucosamine, in mice. Infect Immun. 1997; 65:1734–1741.28. Da Silva CA, Chalouni C, Williams A, Hartl D, Lee CG, Elias JA. Chitin is a size-dependent regulator of macrophage TNF and IL-10 production. J Immunol. 2009; 182:3573–3582.29. Da Silva CA, Hartl D, Liu W, Lee CG, Elias JA. TLR-2 and IL-17A in chitin-induced macrophage activation and acute inflammation. J Immunol. 2008; 181:4279–4286.30. Chen Q, Davidson TS, Huter EN, Shevach EM. Engagement of TLR2 does not reverse the suppressor function of mouse regulatory T cells, but promotes their survival. J Immunol. 2009; 183:4458–4466.31. Choi JP, Kim YM, Choi HI, Choi SJ, Park HT, Lee WH, et al. An important role of tumor necrosis factor receptor-2 on natural killer T cells on the development of dsRNA-enhanced Th2 cell response to inhaled allergens. Allergy. 2014; 69:186–198.32. Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010; 11:889–896.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Innate Immune Response in House Dust Mite-Induced Allergic Inflammation
- House Dust Mite Allergy in Korea: The Most Important Inhalant Allergen in Current and Future
- House dust mite dose not directly activate the human peripheral blood eosinophils in house dust mite-sensitized asthmatics
- Food and house dust mite allergens in children with atopic dermatitis
- Newly sensitization to house dust mite in an isocyanate-induced asthmatic patient