Immune Netw.
2015 Oct;15(5):232-240. 10.4110/in.2015.15.5.232.
A New Triggering Receptor Expressed on Myeloid Cells (TREM) Family Member, TLT-6, is Involved in Activation and Proliferation of Macrophages
- Affiliations
-
- 1Department of Physiology, School of Medicine, Konkuk University, Chungju 27478, Korea.
- 2Departmet of Biomedical Science, College of Life Science, CHA University, Seongnam 13496, Korea.
- 3National Institute of Animal Science, RDA, Wanju 55365, Korea.
- 4Department of Animal Sciences, Division of Applied Life Science, Gyeongsang National University, Jinju 52828, Korea.
- 5Department of Oral Pharmacology, School of Dentistry and Institute of Dental Bioscience, BK21 plus, Chonbuk National University, Jeonju 54907, Korea.
- 6CHA Biotech. Co., Ltd., Seoul 05053, Korea.
- 7Nuri Science Co., Ltd., Seoul 05053, Korea. ceo@nurisci.com
- KMID: 2150852
- DOI: http://doi.org/10.4110/in.2015.15.5.232
Abstract
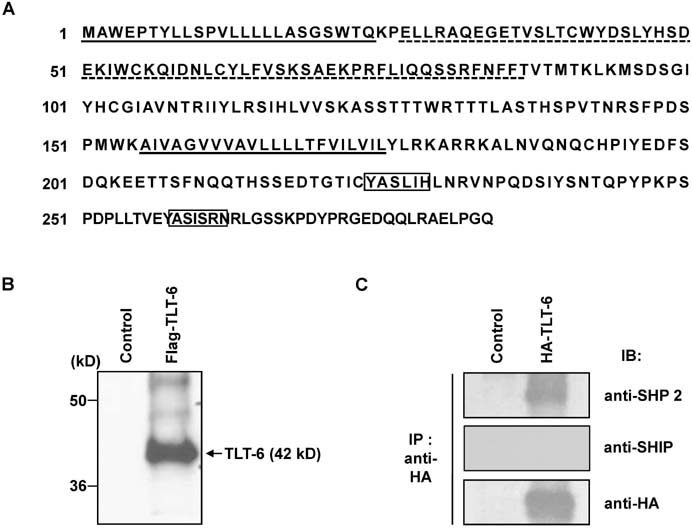
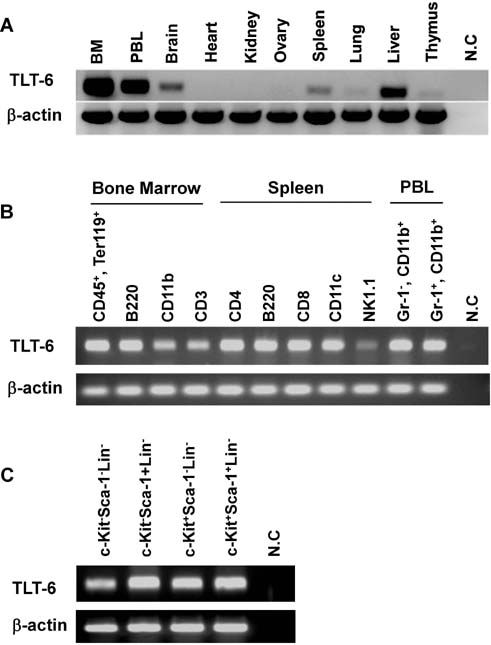
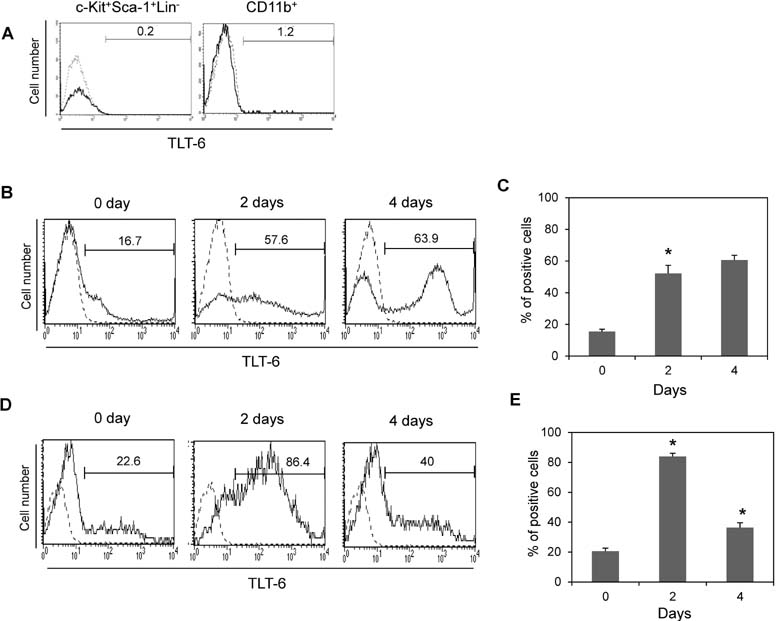
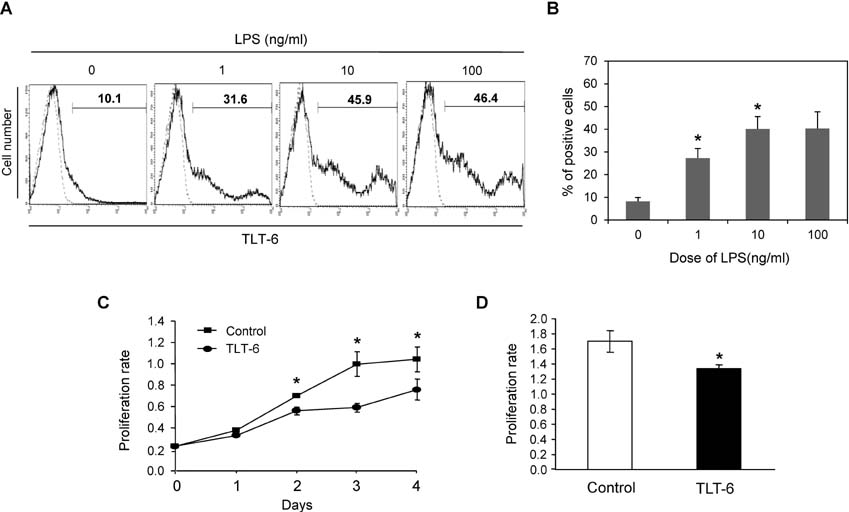
- The triggering receptor expressed on myeloid cells (TREM) family, which is abundantly expressed in myeloid lineage cells, plays a pivotal role in innate and adaptive immune response. In this study, we aimed to identify a novel receptor expressed on hematopoietic stem cells (HSCs) by using in silico bioinformatics and to characterize the identified receptor. We thus found the TREM-like transcript (TLT)-6, a new member of TREM family. TLT-6 has a single immunoglobulin domain in the extracellular region and a long cytoplasmic region containing 2 immunoreceptor tyrosine-based inhibitory motif-like domains. TLT-6 transcript was expressed in HSCs, monocytes and macrophages. TLT-6 protein was up-regulated on the surface of bone marrow-derived and peritoneal macrophages by lipopolysaccharide stimulation. TLT-6 exerted anti-proliferative effects in macrophages. Our results demonstrate that TLT-6 may regulate the activation and proliferation of macrophages.
Keyword
MeSH Terms
Figure
Reference
-
1. Ford JW, McVicar DW. TREM and TREM-like receptors in inflammation and disease. Curr Opin Immunol. 2009; 21:38–46.
Article2. Klesney-Tait J, Turnbull IR, Colonna M. The TREM receptor family and signal integration. Nat Immunol. 2006; 7:1266–1273.
Article3. Zanzinger K, Schellack C, Nausch N, Cerwenka A. Regulation of triggering receptor expressed on myeloid cells 1 expression on mouse inflammatory monocytes. Immunology. 2009; 128:185–195.
Article4. Kusanovic JP, Romero R, Chaiworapongsa T, Mittal P, Mazaki-Tovi S, Vaisbuch E, Erez O, Gotsch F, Than NG, Edwin SS, Pacora P, Jodicke C, Yeo L, Hassan SS. Amniotic fluid sTREM-1 in normal pregnancy, spontaneous parturition at term and preterm, and intra-amniotic infection/inflammation. J Matern Fetal Neonatal Med. 2010; 23:34–47.
Article5. Turnbull IR, Gilfillan S, Cella M, Aoshi T, Miller M, Piccio L, Hernandez M, Colonna M. Cutting edge: TREM-2 attenuates macrophage activation. J Immunol. 2006; 177:3520–3524.
Article6. Washington AV, Schubert RL, Quigley L, Disipio T, Feltz R, Cho EH, McVicar DW. A TREM family member, TLT-1, is found exclusively in the alpha-granules of megakaryocytes and platelets. Blood. 2004; 104:1042–1047.
Article7. Washington AV, Gibot S, Acevedo I, Gattis J, Quigley L, Feltz R, De La MA, Schubert RL, Gomez-Rodriguez J, Cheng J, Dutra A, Pak E, Chertov O, Rivera L, Morales J, Lubkowski J, Hunter R, Schwartzberg PL, McVicar DW. TREM-like transcript-1 protects against inflammation-associated hemorrhage by facilitating platelet aggregation in mice and humans. J Clin Invest. 2009; 119:1489–1501.
Article8. King RG, Herrin BR, Justement LB. Trem-like transcript 2 is expressed on cells of the myeloid/granuloid and B lymphoid lineage and is up-regulated in response to inflammation. J Immunol. 2006; 176:6012–6021.
Article9. Hashiguchi M, Kobori H, Ritprajak P, Kamimura Y, Kozono H, Azuma M. Triggering receptor expressed on myeloid cell-like transcript 2 (TLT-2) is a counter-receptor for B7-H3 and enhances T cell responses. Proc Natl Acad Sci U S A. 2008; 105:10495–10500.
Article10. Hemmi H, Idoyaga J, Suda K, Suda N, Kennedy K, Noda M, Aderem A, Steinman RM. A new triggering receptor expressed on myeloid cells (Trem) family member, Trem-like 4, binds to dead cells and is a DNAX activation protein 12-linked marker for subsets of mouse macrophages and dendritic cells. J Immunol. 2009; 182:1278–1286.
Article11. Sharif O, Knapp S. From expression to signaling: roles of TREM-1 and TREM-2 in innate immunity and bacterial infection. Immunobiology. 2008; 213:701–713.
Article12. Washington AV, Quigley L, McVicar DW. Initial characterization of TREM-like transcript (TLT)-1: a putative inhibitory receptor within the TREM cluster. Blood. 2002; 100:3822–3824.
Article13. Barrow AD, Astoul E, Floto A, Brooke G, Relou IA, Jennings NS, Smith KG, Ouwehand W, Farndale RW, Alexander DR, Trowsdale J. Cutting edge: TREM-like transcript-1, a platelet immunoreceptor tyrosine-based inhibition motif encoding costimulatory immunoreceptor that enhances, rather than inhibits, calcium signaling via SHP-2. J Immunol. 2004; 172:5838–5842.
Article14. Davies JQ, Gordon S. Isolation and culture of murine macrophages. Methods Mol Biol. 2005; 290:91–103.
Article15. Levesque SA, Kukulski F, Enjyoji K, Robson SC, Sevigny J. NTPDase1 governs P2X7-dependent functions in murine macrophages. Eur J Immunol. 2010; 40:1473–1485.16. Sordet O, Rébé C, Plenchette S, Zermati Y, Hermine O, Vainchenker W, Garrido C, Solary E, Dubrez-Daloz L. Specific involvement of caspases in the differentiation of monocytes into macrophages. Blood. 2002; 100(13):4446–4453.
Article17. Kim MS, Kim YK, Kim YS, Seong M, Choi JK, Baek KH. Deubiquitinating enzyme USP36 contains the PEST motif and is polyubiquitinated. Biochem Biophys Res Commun. 2005; 30:797–804.
Article18. Gu BC, Kwon MS, Kim TW. Control of expression for GFP gene using tetracycline inducible retrovirus vector system. Reprod Dev Biol. 2005; 29:57–62.19. Deneault E, Cellot S, Faubert A, Laverdure JP, Frechette M, Chagraoui J, Mayotte N, Sauvageau M, Ting SB, Sauvageau G. A functional screen to identify novel effectors of hematopoietic stem cell activity. Cell. 2009; 137:369–379.
Article20. Seita J, Weissman IL. Hematopoietic stem cell: self-renewal versus differentiation. Wiley Interdiscip Rev Syst Biol Med. 2010; 2:640–653.
Article21. Sharma S, Gurudutta GU, Satija NK, Pati S, Afrin F, Gupta P, Verma YK, Singh VK, Tripathi RP. Stem cell c-KIT and HOXB4 genes: critical roles and mechanisms in self-renewal, proliferation, and differentiation. Stem Cells Dev. 2006; 15:755–778.
Article22. Nonas SA, Finigan JH, Gao L, Garcia JG. Functional genomic insights into acute lung injury: role of ventilators and mechanical stress. Proc Am Thorac Soc. 2005; 2:188–194.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnostic Utility of Pleural Fluid Soluble Triggering Receptor Expressed on Myeloid Cells 1 Protein in Patients with Exudative Pleural Effusion
- Cigarette Smoke Extract-Treated Mouse Airway Epithelial Cells-Derived Exosomal LncRNA MEG3 Promotes M1 Macrophage Polarization and Pyroptosis in Chronic Obstructive Pulmonary Disease by Upregulating TREM-1 via m6A Methylation
- Regulation of Immune Responses by the Activating and Inhibitory Myeloid-Associate Immunoglobuline-Like Receptors (MAIR) (CD300)
- Prognostic Utility of the Soluble Triggering Receptor Expressed on Myeloid Cells-1 in Patients with Acute Respiratory Distress Syndrome
- Protease-activated Receptor 2 is Associated with Activation of Human Macrophage Cell Line THP-1