Immune Netw.
2012 Dec;12(6):277-283. 10.4110/in.2012.12.6.277.
Vitamin C Up-regulates Expression of CD80, CD86 and MHC Class II on Dendritic Cell Line, DC-1 Via the Activation of p38 MAPK
- Affiliations
-
- 1School of Korean Medicine, Pusan National University, Pusan 626-870, Korea.
- 2Laboratory of Immunology and Vitamin C, Department of Anatomy, Seoul National University College of Medicine, Seoul 110-799, Korea. genius29@snu.ac.kr
- KMID: 2150760
- DOI: http://doi.org/10.4110/in.2012.12.6.277
Abstract
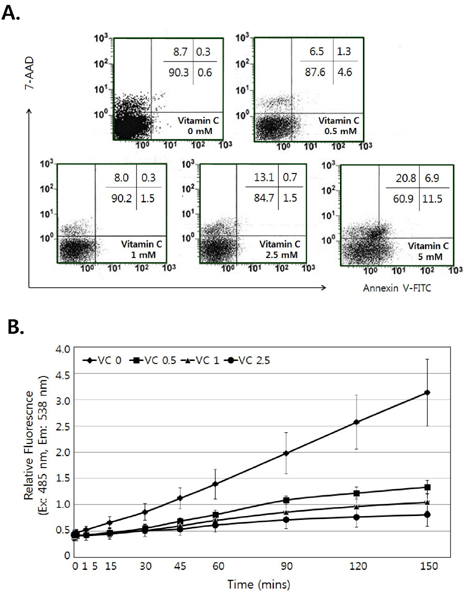
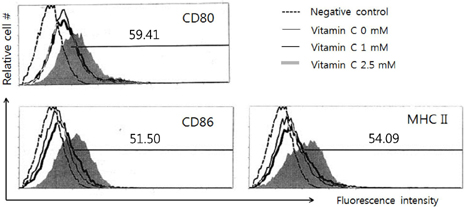
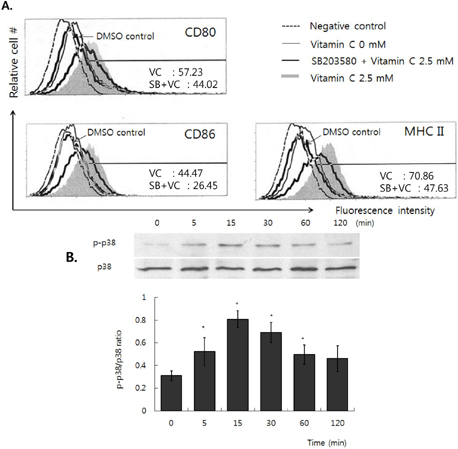
- Vitamin C is an essential water-soluble nutrient which primarily exerts its effect on host defense mechanisms and immune homeostasis, but the mechanism related to immune-potentiation is poorly understood. Since dendritic cells (DCs) are known as a potent antigen presenting cell (APC) that could enhance the antigen specific immune responses, we investigate the effects of vitamin C on activation of DCs and its related mechanism by using dendritic cell lines, DC-1. First, we found that there was no damage on DC-1 by 2.5 mM of vitamin C. In the presence of vitamin C, the expression of CD80, CD86, and MHC molecules was increased, but it was decreased by the pre-treatment of SB203580, p38 MAPK-specific inhibitor. We confirmed the phosphorylation of p38 MAPK was increased by the treatment of vitamin C. Taken together, these results suggest that vitamin C could enhance the activity of dendritic cells via the up-regulation of the expression of CD80, CD86, and MHC molecules and the activation of p38 MAPK is related to this process.
Keyword
MeSH Terms
Figure
Reference
-
1. Härtel C, Strunk T, Bucsky P, Schultz C. Effects of vitamin C on intracytoplasmic cytokine production in human whole blood monocytes and lymphocytes. Cytokine. 2004. 27:101–106.
Article2. Hartel C, Puzik A, Gopel W, Temming P, Bucsky P, Schultz C. Immunomodulatory effect of vitamin C on intracytoplasmic cytokine production in neonatal cord blood cells. Neonatology. 2007. 91:54–60.
Article3. Bergsten P, Amitai G, Kehrl J, Dhariwal KR, Klein HG, Levine M. Millimolar concentrations of ascorbic acid in purified human mononuclear leukocytes. Depletion and reaccumulation. J Biol Chem. 1990. 265:2584–2587.
Article4. May JM, Huang J, Qu ZC. Macrophage uptake and recycling of ascorbic acid: response to activation by lipopolysaccharide. Free Radic Biol Med. 2005. 39:1449–1459.
Article5. Washko P, Rotrosen D, Levine M. Ascorbic acid in human neutrophils. Am J Clin Nutr. 1991. 54(6 Suppl):1221S–1227S.
Article6. Levine M, Conry-Cantilena C, Wang Y, Welch RW, Washko PW, Dhariwal KR, Park JB, Lazarev A, Graumlich JF, King J, Cantilena LR. Vitamin C pharmacokinetics in healthy volunteers: evidence for a recommended dietary allowance. Proc Natl Acad Sci U S A. 1996. 93:3704–3709.
Article7. Anderson R, Oosthuizen R, Maritz R, Theron A, Van Rensburg AJ. The effects of increasing weekly doses of ascorbate on certain cellular and humoral immune functions in normal volunteers. Am J Clin Nutr. 1980. 33:71–76.
Article8. Anderson R, Smit MJ, Joone GK, Van Staden AM. Vitamin C and cellular immune functions. Protection against hypochlorous acid-mediated inactivation of glyceraldehyde-3-phosphate dehydrogenase and ATP generation in human leukocytes as a possible mechanism of ascorbatemediated immunostimulation. Ann N Y Acad Sci. 1990. 587:34–48.9. Levy R, Shriker O, Porath A, Riesenberg K, Schlaeffer F. Vitamin C for the treatment of recurrent furunculosis in patients with imparied neutrophil functions. J Infect Dis. 1996. 173:1502–1505.
Article10. Jacob RA, Kelley DS, Pianalto FS, Swendseid ME, Henning SM, Zhang JZ, Ames BN, Fraga CG, Peters JH. Immunocompetence and oxidant defense during ascorbate depletion of healthy men. Am J Clin Nutr. 1991. 54:6 Suppl. 1302S–1309S.
Article11. Heuser G, Vojdani A. Enhancement of natural killer cell activity and T and B cell function by buffered vitamin C in patients exposed to toxic chemicals: the role of protein kinase-C. Immunopharmacol Immunotoxicol. 1997. 19:291–312.
Article12. Noh K, Lim H, Moon SK, Kang JS, Lee WJ, Lee D, Hwang YI. Mega-dose Vitamin C modulates T cell functions in Balb/c mice only when administered during T cell activation. Immunol Lett. 2005. 98:63–72.
Article13. Kay NE, Holloway DE, Hutton SW, Bone ND, Duane WC. Human T-cell function in experimental ascorbic acid deficiency and spontaneous scurvy. Am J Clin Nutr. 1982. 36:127–130.
Article14. Lee CW, Wang XD, Chien KL, Ge Z, Rickman BH, Rogers AB, Varro A, Whary MT, Wang TC, Fox JG. Vitamin C supplementation does not protect L-gulono-gamma-lactone oxidase-deficient mice from Helicobacter pylori-induced gastritis and gastric premalignancy. Int J Cancer. 2008. 122:1068–1076.
Article15. Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, Pulendran B, Palucka K. Immunobiology of dendritic cells. Annu Rev Immunol. 2000. 18:767–811.
Article16. Kumaraguru U, Pack CD, Rouse BT. Toll-like receptor ligand links innate and adaptive immune responses by the production of heat-shock proteins. J Leukoc Biol. 2003. 73:574–583.
Article17. Asai Y, Makimura Y, Ogawa T. Toll-like receptor 2-mediated dendritic cell activation by a Porphyromonas gingivalis synthetic lipopeptide. J Med Microbiol. 2007. 56:459–465.
Article18. Gaddis DE, Michalek SM, Katz J. Requirement of TLR4 and CD14 in dendritic cell activation by Hemagglutinin B from Porphyromonas gingivalis. Mol Immunol. 2009. 46:2493–2504.
Article19. Sinha A, Singh A, Satchidanandam V, Natarajan K. Impaired generation of reactive oxygen species during differentiation of dendritic cells (DCs) by Mycobacterium tuberculosis secretory antigen (MTSA) and subsequent activation of MTSA-DCs by mycobacteria results in increased intracellular survival. J Immunol. 2006. 177:468–478.
Article20. Wang D, Malo D, Hekimi S. Elevated mitochondrial reactive oxygen species generation affects the immune response via hypoxia-inducible factor-1alpha in long-lived Mclk1+/- mouse mutants. J Immunol. 2010. 184:582–590.
Article21. Cho D, Hahm E, Kang JS, Kim YI, Yang Y, Park JH, Kim D, Kim S, Kim YS, Hur D, Park H, Pang S, Hwang YI, Lee WJ. Vitamin C downregulates interleukin-18 production by increasing reactive oxygen intermediate and mitogen-activated protein kinase signalling in B16F10 murine melanoma cells. Melanoma Res. 2003. 13:549–554.
Article22. Levine M, Conry-Cantilena C, Wang Y, Welch RW, Washko PW, Dhariwal KR, Park JB, Lazarev A, Graumlich JF, King J, Cantilena LR. Vitamin C pharmacokinetics in healthy volunteers: evidence for a recommended dietary allowance. Proc Natl Acad Sci U S A. 1996. 93:3704–3709.
Article23. Levine M, Wang Y, Padayatty SJ, Morrow J. A new recommended dietary allowance of vitamin C for healthy young women. Proc Natl Acad Sci U S A. 2001. 98:9842–9846.
Article24. Padayatty SJ, Sun H, Wang Y, Riordan HD, Hewitt SM, Katz A, Wesley RA, Levine M. Vitamin C pharmacokinetics: implications for oral and intravenous use. Ann Intern Med. 2004. 140:533–537.
Article25. Kang JS, Cho D, Kim YI, Hahm E, Yang Y, Kim D, Hur D, Park H, Bang S, Hwang YI, Lee WJ. L-ascorbic acid (vitamin C) induces the apoptosis of B16 murine melanoma cells via a caspase-8-independent pathway. Cancer Immunol Immunother. 2003. 52:693–698.
Article26. Kang JS, Cho D, Kim YI, Hahm E, Kim YS, Jin SN, Kim HN, Kim D, Hur D, Park H, Hwang YI, Lee WJ. Sodium ascorbate (vitamin C) induces apoptosis in melanoma cells via the down-regulation of transferrin receptor dependent iron uptake. J Cell Physiol. 2005. 204:192–197.
Article27. Hahm E, Jin DH, Kang JS, Kim YI, Hong SW, Lee SK, Kim HN, Jung da J, Kim JE, Shin DH, Hwang YI, Kim YS, Hur DY, Yang Y, Cho D, Lee MS, Lee WJ. The molecular mechanisms of vitamin C on cell cycle regulation in B16F10 murine melanoma. J Cell Biochem. 2007. 102:1002–1010.
Article28. Lee SK, Kang JS, Jung da J, Hur DY, Kim JE, Hahm E, Bae S, Kim HW, Kim D, Cho BJ, Cho D, Shin DH, Hwang YI, Lee WJ. Vitamin C suppresses proliferation of the human melanoma cell SK-MEL-2 through the inhibition of cyclooxygenase-2 (COX-2) expression and the modulation of insulin-like growth factor II (IGF-II) production. J Cell Physiol. 2008. 216:180–188.
Article29. Kim HN, Kim H, Kong JM, Bae S, Kim YS, Lee N, Cho BJ, Lee SK, Kim HR, Hwang YI, Kang JS, Lee WJ. Vitamin C down-regulates VEGF production in B16F10 murine melanoma cells via the suppression of p42/44 MAPK activation. J Cell Biochem. 2011. 112:894–901.
Article30. Yu Y, Bae S, Kim H, Kim Y, Chu NB, Chu NK, Kang JS, Lee WJ. The Anti-tumor Activity of Vitamin C via the Increase of Fas (CD95) and MHC I Expression on Human Stomach Cancer Cell Line, SNU1. Immune Netw. 2011. 11:210–215.
Article31. Iijima N, Yanagawa Y, Iwabuchi K, Onoé K. Selective regulation of CD40 expression in murine dendritic cells by thiol antioxidants. Immunology. 2003. 110:197–205.
Article32. Tomiyama C, Watanabe H, Izutsu Y, Watanabe M, Abo T. Suppressive role of hepatic dendritic cells in concanavalin A-induced hepatitis. Clin Exp Immunol. 2011. 166:258–268.
Article33. Chen KD, Hsu LW, Goto S, Yeh CW, Nakano T, Lai CY, Chang YC, Hou CH, Wang CC, Cheng YF, Chiu KW, Lin CC, Chen CL. Adaptor protein Shc acts as an immune-regulator for the LPS-stimulated maturation of bone marrow-derived dendritic cells. BMC Immunol. 2011. 12:32.
Article34. Linster CL, Van Schaftingen E. Vitamin C. Biosynthesis, recycling and degradation in mammals. FEBS J. 2007. 274:1–22.35. Tsukaguchi H, Tokui T, Mackenzie B, Berger UV, Chen XZ, Wang Y, Brubaker RF, Hediger MA. A family of mammalian Na+-dependent L-ascorbic acid transporters. Nature. 1999. 399:70–75.
Article36. Daruwala R, Song J, Koh WS, Rumsey SC, Levine M. Cloning and functional characterization of the human sodium-dependent vitamin C transporters hSVCT1 and hSVCT2. FEBS Lett. 1999. 460:480–484.
Article37. Kim H, Bae S, Yu Y, Kim Y, Kim HR, Hwang YI, Kang JS, Lee WJ. The analysis of vitamin C concentration in organs of gulo(-/-) mice upon vitamin C withdrawal. Immune Netw. 2012. 12:18–26.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of dendritic cell markers on cultured neutrophils and its modulation by anti-apoptotic and pro-apoptotic compounds
- Expression of cytokines and co-stimulatory molecules in the Toxoplasma gondii-infected dendritic cells of C57BL/6 and BALB/c mice
- Induction of Functional Changes of Dendritic Cells by Silica Nanoparticles
- Transcription of class II MHC gene by interferon-gamma in FRTL-5 cells
- Cross-linking of CD80 and CD86 Diminishes Expression of CD54 on EBV-transformed B Cells through Inactivation of RhoA and Ras