Immune Netw.
2010 Dec;10(6):239-246. 10.4110/in.2010.10.6.239.
Characterization of a Novel Monoclonal Antibody (27H2) Recognizing Human CD34 Class III Epitope
- Affiliations
-
- 1Department of Pathology, College of Medicine and Medical Research Institute, Chungbuk National University, Cheongju 361-763, Korea. hgsong@chungbuk.ac.kr
- 2Research Institute, DiNonA Inc, Iksan 570-912, Korea.
- 3Department of Laboratory Medicine, College of Medicine and Medical Research Institute, Chungbuk National University, Cheongju 361-763, Korea.
- 4Department of Plastic and Reconstructive Surgery, College of Medicine and Medical Research Institute, Chungbuk National University, Cheongju 361-763, Korea.
- KMID: 2150680
- DOI: http://doi.org/10.4110/in.2010.10.6.239
Abstract
- BACKGROUND
Monoclonal antibodies (mAbs) recognizing Class III epitope of CD34 are essential for flow cytometric diagnosis of leukemia.
METHODS
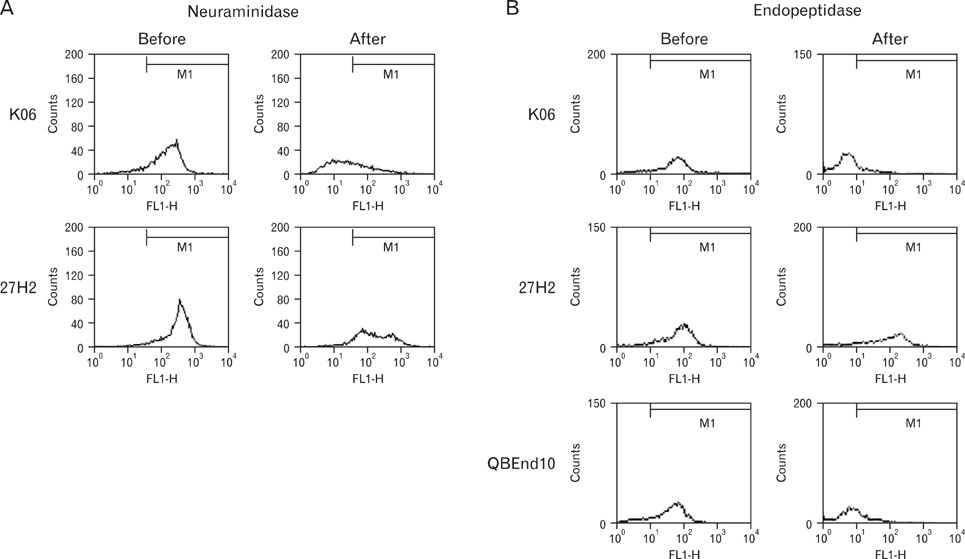
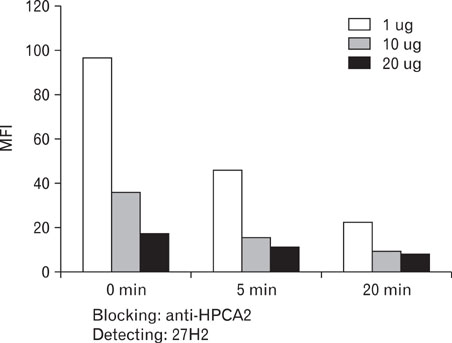
27H2 mAb was developed from a mouse alternatively immunized with human acute leukemia cell lines, KG1 and Molm-1. Using flow cytometric analysis of various leukemic cell lines and peripheral blood, immunohistochemical study of frozen tonsil, we characterized 27H2 mAb. Antigen immunoprecipitated with 27H2 mAb immunobloted with anti-CD34 mAb. A case of bone marrow sample of acute lymphoblastic leukemia (ALL) patient was obtained at CBNU Hospital. For epitope identification enzyme treatment with neuraminidase and O-sialoglycoprotein endopeptidase (OSGE) and blocking assay with known classIII mAb (HPCA-2) were done.
RESULTS
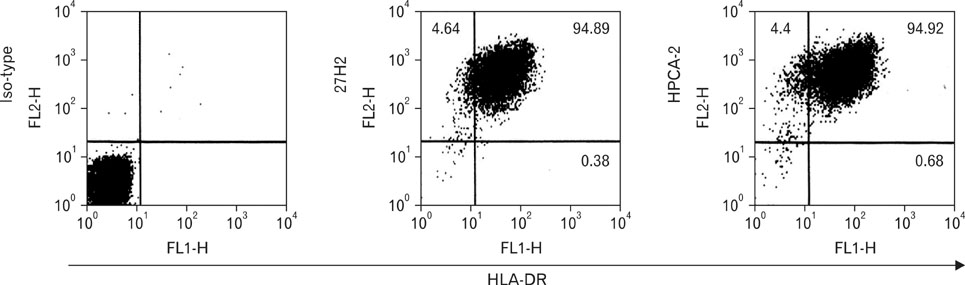
Only KG1 and Molm-1 revealed positive immunoreactivity. Immunohistochemical staining disclosed strong membranous immunoreactivity on high endothelial venules. Antigen immunoprecipitated by 27H2 mAb showed approximately 100 kDa sized band immunoblotted with anti-CD34 under non-reducing conditions. Epitope recognized by 27H2 mAb disclosed resistancy to both neuraminidase and OSGE treatment and completely blocked with known class III mAb preincubation. CD34 positive leukemic cells in BM of pre B cell ALL patient detected by FITC-conjugated 27H2 and HPCA-2 were identified with similar sensitivity.
CONCLUSION
A novel murine mAb recognizing class III epitope of human CD34 with high affinity, which is useful for flow cytometric diagnosis of leukemia, was developed.
Keyword
MeSH Terms
Figure
Reference
-
1. Civin CI, Strauss LC, Brovall C, Fackler MJ, Schwartz JF, Shaper JH. Antigenic analysis of hematopoiesis. III. A hematopoietic progenitor cell surface antigen defined by a monoclonal antibody raised against KG-1a cells. J Immunol. 1984. 133:157–165.2. Tindle RW, Nichols RA, Chan L, Campana D, Catovsky D, Birnie GD. A novel monoclonal antibody BI-3C5 recognises myeloblasts and non-B non-T lymphoblasts in acute leukaemias and CGL blast crises, and reacts with immature cells in normal bone marrow. Leuk Res. 1985. 9:1–9.
Article3. Watt SM, Karhi K, Gatter K, Furley AJ, Katz FE, Healy LE, Altass LJ, Bradley NJ, Sutherland DR, Levinsky R, et al. Distribution and epitope analysis of the cell membrane glycoprotein (HPCA-1) associated with human hemopoietic progenitor cells. Leukemia. 1987. 1:417–426.4. Beschorner WE, Civin CI, Strauss LC. Localization of hematopoietic progenitor cells in tissue with the anti-My-10 monoclonal antibody. Am J Pathol. 1985. 119:1–4.5. Fina L, Molgaard HV, Robertson D, Bradley NJ, Monaghan P, Delia D, Sutherland DR, Baker MA, Greaves MF. Expression of the CD34 gene in vascular endothelial cells. Blood. 1990. 75:2417–2426.
Article6. Civin CI, Small D. Purification and expansion of human hematopoietic stem/progenitor cells. Ann N Y Acad Sci. 1995. 770:91–98.
Article7. Greaves MF, Brown J, Molgaard HV, Spurr NK, Robertson D, Delia D, Sutherland DR. Molecular features of CD34: a hemopoietic progenitor cell-associated molecule. Leukemia. 1992. 6:Suppl 1. 31–36.8. Sutherland DR, Keating A. The CD34 antigen: structure, biology, and potential clinical applications. J Hematother. 1992. 1:115–129.
Article9. Sutherland DR, Anderson L, Keeney M, Nayar R, Chin-Yee I. The ISHAGE guidelines for CD34+ cell determination by flow cytometry. International Society of Hematotherapy and Graft Engineering. J Hematother. 1996. 5:213–226.10. Siena S, Bregni M, Brando B, Belli N, Lansdorp PM, Bonadonna G, Gianni M. Flow cytometry to estimate circulating hematopoietic progenitors for autologous transplantation: comparative analysis of different CD34 monoclonal antibodies. Haematologica. 1991. 76:330–333.11. Katz FE, Tindle R, Sutherland DR, Greaves MF. Identification of a membrane glycoprotein associated with haemopoietic progenitor cells. Leuk Res. 1985. 9:191–198.
Article12. Shpall EJ, Jones RB, Franklin W. Transplantation of autologous CD34+ hematopoietic progenitor cells into breast cancer patients following high-dose chemotherapy. J Clin Oncol. 1994. 12:28–32.13. Vescio R, Schiller G, Stewart AK, Ballester O, Noga S, Rugo H, Freytes C, Stadtmauer E, Tarantolo S, Sahebi F, Stiff P, Meharchard J, Schlossman R, Brown R, Tully H, Benyunes M, Jacobs C, Berenson R, DiPersio J, Anderson K, Berenson J. Multicenter phase III trial to evaluate CD34(+) selected versus unselected autologous peripheral blood progenitor cell transplantation in multiple myeloma. Blood. 1999. 93:1858–1868.14. Civin CI, Trischmann TM, Fackler MJ, Bernstein ID, Bühring HJ, Campos L, et al. In : Knapp W, Dörken B, Gilks WR, Rieber EP, Schmidt RE, Stein H, editors. M7.1. Report on the CD34 cluster workshop. Leukocyte typing IV. White cell differentiation antigens. 1989. Proceedings of the 4th International Workshop and Conference; 1989 Feb 21-25; Vienna, Austria. New York, Tokyo: Oxford University Press;818–825.15. Greaves MF, Titley I, Colman SM, Buhring H-J, Campos L, Castoldi GL, et al. Schlossman S, editor. Report on the CD34 cluster Workshop. Leukocyte Typing V. 1995. Oxford University Press;840–846.16. Nishio H, Tada J, Hashiyama M, Hirn J, Ingles-Esteve J, Suda T. In : Kishimoto T, Kikutani H, von dem Borne AEG, Goyert SM, Mason DY, Miyasaka M, editors. MC7. CD34 workshop panel report. Leucocyte typing VI. White cell differentiation antigens. 1997. Proceedings of the 6th International Workshop and Conference; 1996 Nov 10-14; Kobe, Japan. New York, London: Garland Publishing Inc.;974–984.17. Titley I, Healy LE, Scott M, Amos TA, Gordon MY. Extent of variability inherent in measurements of CD34-positive cells in different human haemopoietic tissues. Bone Marrow Transplant. 1995. 16:611–616.18. Titley I, Healy LE, Scott M, Amos TA, Gordon MY. Extent of variability inherent in measurements of CD34-positive cells in different human haemopoietic tissues. Bone Marrow Transplant. 1995. 16:611–616.19. Cannovo N, Bossy D, Hirn J. Significant diversity of the reactivity of monoclonal CD34 antibodies requires an extended classification of the CD34 epitopes. Lab Hematol. 1997. 3:253–260.20. Serke S, Huhn D. Expression of class I, II and III epitopes of the CD34 antigen by normal and leukemic hemopoietic cells. Cytometry. 1996. 26:154–160.
Article21. Steen R, Tjønnfjord GE, Gaudernack G, Brinch L, Egeland T. Differences in the distribution of CD34 epitopes on normal haemopoietic progenitor cells and leukaemic blast cells. Br J Haematol. 1996. 94:597–605.
Article22. Lanza F, Moretti S, Castagnari B, Latorraca A, Rigolin GM, Bardi A, Castoldi G. CD34+ leukemic cells assessed by different CD34 monoclonal antibodies. Leuk Lymphoma. 1995. 18:Suppl 1. 25–30.
Article23. Weinberg DS, Benjamin RJ. QBEnd10 (CD34) antibody is unsuitable for routine use in the ISHAGE CD34+ cell determination assay. J Hematother. 1996. 5:599–603.24. Macey MG, McCarthy DA, vanAgthoven A, Newland AC. How should CD34+ cells be analysed? A study of three classes of antibody and five leucocyte preparation procedures. J Immunol Methods. 1997. 204:175–188.
Article25. Sovalat H, Racadot E, Hénon P, Fuchs P, Lewandowski H, Billot M. Comparative analysis of class I, II and III epitope-detecting CD34 monoclonal antibodies by quantitative flow cytometry. Hematol Cell Ther. 1998. 40:259–268.26. Sutherland DR, Anderson L, Nayar R, Keeney M, Chin Yee I. Ball E, Lister J, editors. The ISHAGE Guidelines for CD34+ cell determination: Applications in autologous and allogenic blood and marrow transplantation. Bone Marrow Transplantation: A Practical Approach. 2000. Churchill Livingstone;239–246.27. Lanza F, Healy L, Sutherland DR. Structural and functional features of the CD34 antigen: an update. J Biol Regul Homeost Agents. 2001. 15:1–13.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development of Monoclonal Antibodies Recognizing Human Peripheral Blood T Lymphocytes Cytoplasmic Proteins Induced upon Activation
- Characterization of the Monoclonal Antibody Specific to Human S100A6 Protein
- Production and characterization of monoclonal antibodies to oxidized LDL
- Generation and Characterization of Anti - Human CTLA - 4 Monoclonal Antibodies
- Production and characterization of cross-reactive anti-Porphyromonas gingivalis heat shock protein 60 monoclonal antibody