Korean J Physiol Pharmacol.
2016 Jan;20(1):91-99. 10.4196/kjpp.2016.20.1.91.
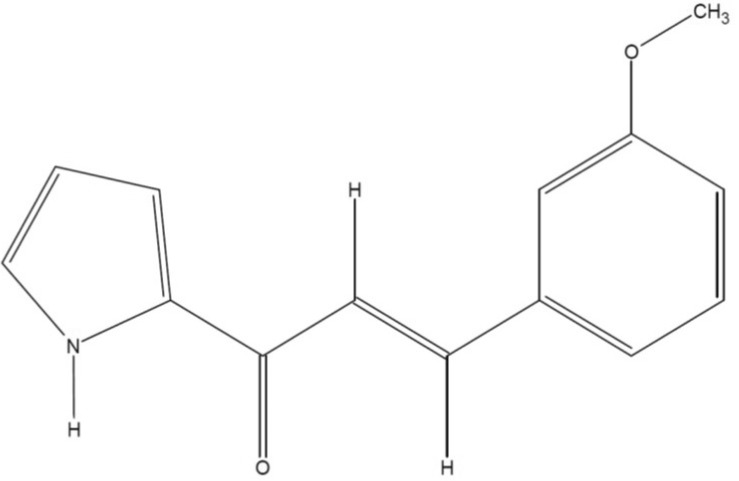
(E)-3-(3-methoxyphenyl)-1-(2-pyrrolyl)-2-propenone displays suppression of inflammatory responses via inhibition of Src, Syk, and NF-kappaB
- Affiliations
-
- 1Department of Genetic Engineering, Sungkyunkwan University, Suwon 16419, Korea. jaecho@skku.edu
- 2Department of Science Education, Kangwon National University, Chuncheon 24341, Korea. ishl@kangwon.ac.kr
- 3Department of Bioinformatics and Life Science, Soongsil University, Seoul 06978, Korea. kimmy@ssu.ac.kr
- KMID: 2150478
- DOI: http://doi.org/10.4196/kjpp.2016.20.1.91
Abstract
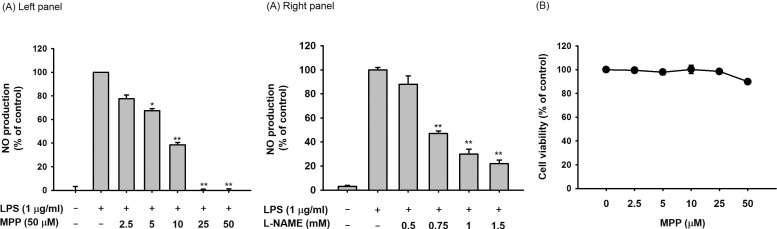
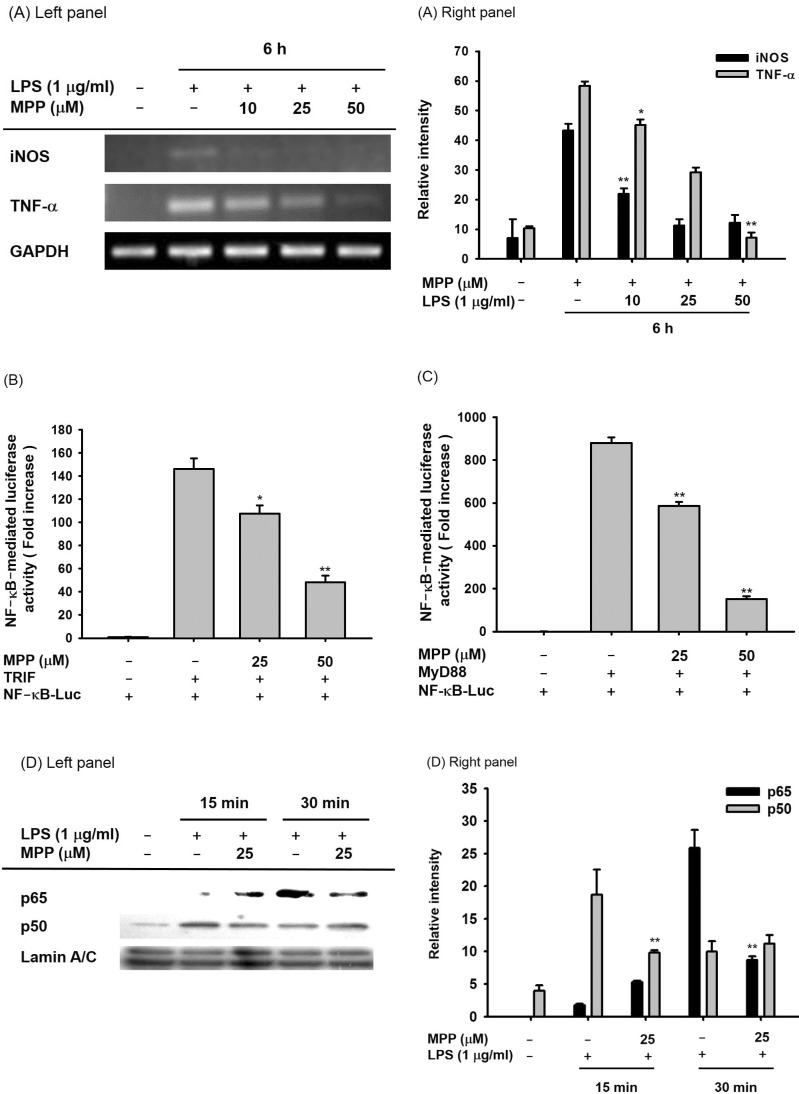
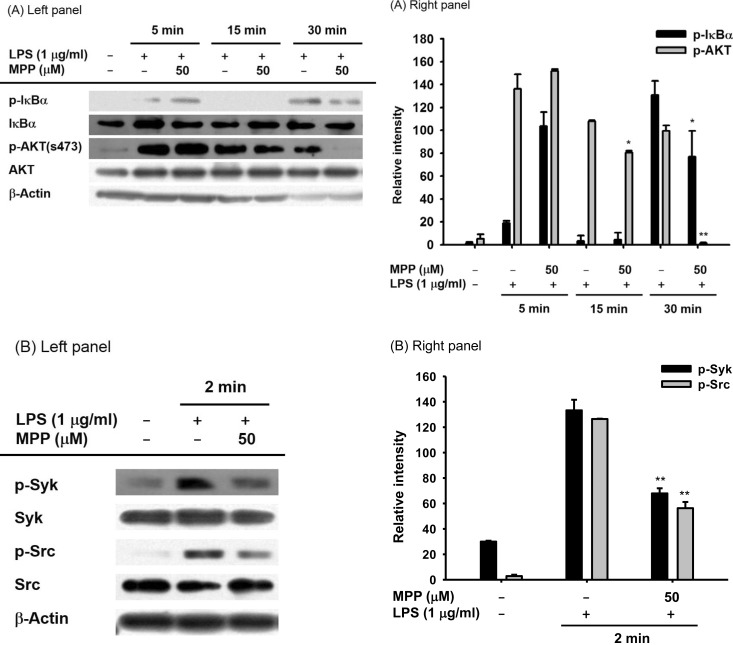
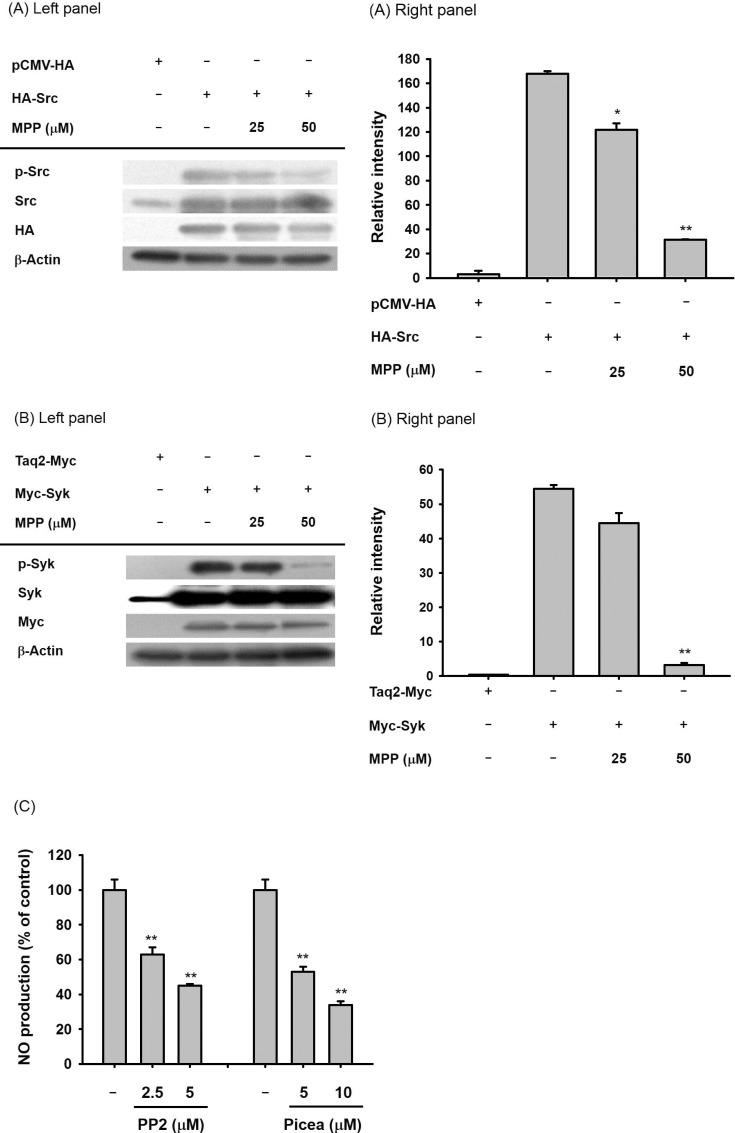
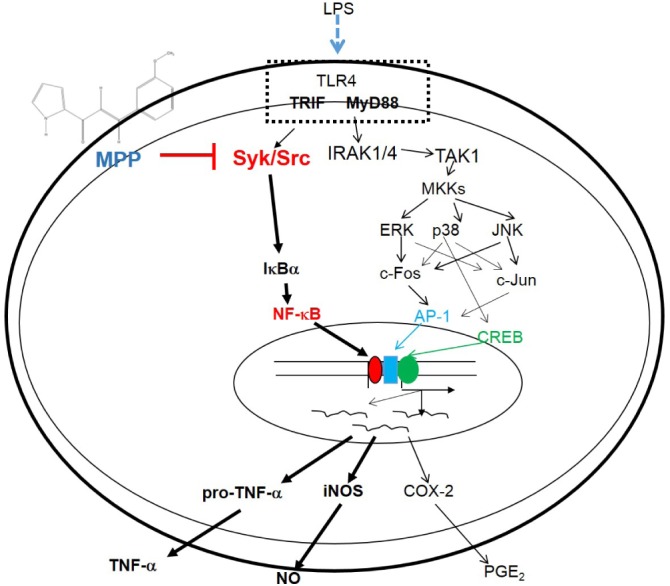
- (E)-3-(3-methoxyphenyl)-1-(2-pyrrolyl)-2-propenone (MPP) is an aldol condensation product resulting from pyrrole-2-carbaldehyde and m- and p- substituted acetophenones. However, its biological activity has not yet been evaluated. Since it has been reported that some propenone-type compounds display anti-inflammatory activity, we investigated whether MPP could negatively modulate inflammatory responses. To do this, we employed lipopolysaccharide (LPS)-stimulated macrophage-like RAW264.7 cells and examined the inhibitory levels of nitric oxide (NO) production and transcriptional activation, as well as the target proteins involved in the inflammatory signaling cascade. Interestingly, MPP was found to reduce the production of NO in LPS-treated RAW264.7 cells, without causing cytotoxicity. Moreover, this compound suppressed the mRNA levels of inflammatory genes, such as inducible NO synthase (iNOS) and tumor necrosis factor (TNF)-alpha. Using luciferase reporter gene assays performed in HEK293 cells and immunoblotting analysis with nuclear protein fractions, we determined that MPP reduced the transcriptional activation of nuclear factor (NF)-kappaB. Furthermore, the activation of a series of upstream signals for NF-kappaB activation, composed of Src, Syk, Akt, and IkappaBalpha, were also blocked by this compound. It was confirmed that MPP was able to suppress autophosphorylation of overexpressed Src and Syk in HEK293 cells. Therefore, these results suggest that MPP can function as an anti-inflammatory drug with NF-kappaB inhibitory properties via the suppression of Src and Syk.
Keyword
MeSH Terms
-
Acetophenones
Genes, Reporter
HEK293 Cells
Immunoblotting
Luciferases
Macrophages
NF-kappa B*
Nitric Oxide
Nitric Oxide Synthase
Nuclear Proteins
RNA, Messenger
Transcriptional Activation
Tumor Necrosis Factor-alpha
Acetophenones
Luciferases
NF-kappa B
Nitric Oxide
Nitric Oxide Synthase
Nuclear Proteins
RNA, Messenger
Tumor Necrosis Factor-alpha
Figure
Cited by 1 articles
-
JS-III-49, a hydroquinone derivative, exerts anti-inflammatory activity by targeting Akt and p38
Young-Su Yi, Mi-Yeon Kim, Jae Youl Cho
Korean J Physiol Pharmacol. 2017;21(3):345-352. doi: 10.4196/kjpp.2017.21.3.345.
Reference
-
1. Yu T, Li YJ, Bian AH, Zuo HB, Zhu TW, Ji SX, Kong F, Yin de Q, Wang CB, Wang ZF, Wang HQ, Yang Y, Yoo BC, Cho JY. The regulatory role of activating transcription factor 2 in inflammation. Mediators Inflamm. 2014; 2014:950472. PMID: 25049453.
Article2. Yi YS, Son YJ, Ryou C, Sung GH, Kim JH, Cho JY. Functional roles of Syk in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014; 2014:270302. PMID: 25045209.
Article3. Byeon SE, Yi YS, Oh J, Yoo BC, Hong S, Cho JY. The role of Src kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2012; 2012:512926. PMID: 23209344.
Article4. Shrimali D, Shanmugam MK, Kumar AP, Zhang J, Tan BK, Ahn KS, Sethi G. Targeted abrogation of diverse signal transduction cascades by emodin for the treatment of inflammatory disorders and cancer. Cancer Lett. 2013; 341:139–149. PMID: 23962559.
Article5. Li Q, Verma IM. NF-kappaB regulation in the immune system. Nat Rev Immunol. 2002; 2:725–734. PMID: 12360211.6. Lu H, Ouyang W, Huang C. Inflammation, a key event in cancer development. Mol Cancer Res. 2006; 4:221–233. PMID: 16603636.
Article7. Lee SK, Jeong HG, Lee ES, Jeong TC. Metabolism of FPP-3, an anti-inflammatory propenone compound, in rat by liquid chromatography-electrospray ionization tandem mass spectrometry. Biol Pharm Bull. 2007; 30:967–971. PMID: 17473444.
Article8. Park SY, Ku SK, Lee ES, Kim JA. 1,3-Diphenylpropenone ameliorates TNBS-induced rat colitis through suppression of NF-κB activation and IL-8 induction. Chem Biol Interact. 2012; 196:39–49. PMID: 22410118.
Article9. Lee ISH, Jeoung EJ, Lee CK. Synthesis and NMR Studies of (E)-1-Aryl-3-(2-pyrrolyl)-2-propenones and (E)-3-Aryl-1-(2-pyrrolyl)-2-propenones. Bull Korean Chem Soc. 2013; 34:936–942.10. Yang KJ, Shin S, Piao L, Shin E, Li Y, Park KA, Byun HS, Won M, Hong J, Kweon GR, Hur GM, Seok JH, Chun T, Brazil DP, Hemmings BA, Park J. Regulation of 3-phosphoinositide-dependent protein kinase-1 (PDK1) by Src involves tyrosine phosphorylation of PDK1 and Src homology 2 domain binding. J Biol Chem. 2008; 283:1480–1491. PMID: 18024423.
Article11. Byeon SE, Yu T, Yang Y, Lee YG, Kim JH, Oh J, Jeong HY, Hong S, Yoo BC, Cho WJ, Hong S, Cho JY. Hydroquinone regulates hemeoxygenase-1 expression via modulation of Src kinase activity through thiolation of cysteine residues. Free Radic Biol Med. 2013; 57:105–118. PMID: 23290930.
Article12. Shen T, Lee J, Park MH, Lee YG, Rho HS, Kwak YS, Rhee MH, Park YC, Cho JY. Ginsenoside Rp1, a Ginsenoside Derivative, Blocks Promoter Activation of iNOS and COX-2 Genes by Suppression of an IKKβ-mediated NF-XMLLink_XYZB Pathway in HEK293 Cells. J Ginseng Res. 2011; 35:200–208. PMID: 23717062.13. Song SB, Tung NH, Quang TH, Ngan NT, Kim KE, Kim YH. Inhibition of TNF-α-mediated NF-κB Transcriptional Activity in HepG2 Cells by Dammarane-type Saponins from Panax ginseng Leaves. J Ginseng Res. 2012; 36:146–152. PMID: 23717114.
Article14. Kim DH, Chung JH, Yoon JS, Ha YM, Bae S, Lee EK, Jung KJ, Kim MS, Kim YJ, Kim MK, Chung HY. Ginsenoside Rd inhibits the expressions of iNOS and COX-2 by suppressing NF-κB in LPS-stimulated RAW264.7 cells and mouse liver. J Ginseng Res. 2013; 37:54–63. PMID: 23717157.
Article15. Youn CK, Park SJ, Lee MY, Cha MJ, Kim OH, You HJ, Chang IY, Yoon SP, Jeon YJ. Silibinin Inhibits LPS-Induced Macrophage Activation by Blocking p38 MAPK in RAW 264.7 Cells. Biomol Ther (Seoul). 2013; 21:258–263. PMID: 24244809.
Article16. Oh CT, Park JI, Jung YR, Joo YA, Shin DH, Cho HJ, Ahn SM, Lim YH, Park CK, Hwang JS. Inhibitory effect of Korean Red Ginseng on melanocyte proliferation and its possible implication in GM-CSF mediated signaling. J Ginseng Res. 2013; 37:389–400. PMID: 24235857.
Article17. Pauwels R, Balzarini J, Baba M, Snoeck R, Schols D, Herdewijn P, Desmyter J, De Clercq E. Rapid and automated tetrazolium-based colorimetric assay for the detection of anti-HIV compounds. J Virol Methods. 1988; 20:309–321. PMID: 2460479.
Article18. Yayeh T, Jung KH, Jeong HY, Park JH, Song YB, Kwak YS, Kang HS, Cho JY, Oh JW, Kim SK, Rhee MH. Korean red ginseng saponin fraction downregulates proinflammatory mediators in LPS stimulated RAW264.7 cells and protects mice against endotoxic shock. J Ginseng Res. 2012; 36:263–269. PMID: 23717127.
Article19. Xu AN, Chen XH, Tan YH, Qi XL, Xu ZF, Zhang LL, Ren FG, Bian SC, Chen Y, Wang HW. Identification of a novel circularized transcript of the AML1 gene. BMB Rep. 2013; 46:163–168. PMID: 23527860.
Article20. Yang Y, Yang WS, Yu T, Yi YS, Park JG, Jeong D, Kim JH, Oh JS, Yoon K, Kim JH, Cho JY. Novel anti-inflammatory function of NSC95397 by the suppression of multiple kinases. Biochem Pharmacol. 2014; 88:201–215. PMID: 24468133.
Article21. Kim MY, Yoo BC, Cho JY. Ginsenoside-Rp1-induced apolipoprotein A-1 expression in the LoVo human colon cancer cell line. J Ginseng Res. 2014; 38:251–255. PMID: 25379004.
Article22. Byeon SE, Lee YG, Kim BH, Shen T, Lee SY, Park HJ, Park SC, Rhee MH, Cho JY. Surfactin blocks NO production in lipopolysaccharide-activated macrophages by inhibiting NF-kappaB activation. J Microbiol Biotechnol. 2008; 18:1984–1989. PMID: 19131703.23. Yang Y, Yang WS, Yu T, Yi YS, Park JG, Jeong D, Kim JH, Oh JS, Yoon K, Kim JH, Cho JY. Novel anti-inflammatory function of NSC95397 by the suppression of multiple kinases. Biochem Pharmacol. 2014; 88:201–215. PMID: 24468133.
Article24. Lee JA, Lee MY, Shin IS, Seo CS, Ha H, Shin HK. Anti-inflammatory effects of Amomum compactum on RAW 264.7 cells via induction of heme oxygenase-1. Arch Pharm Res. 2012; 35:739–746. PMID: 22553068.
Article25. Sung NY, Kim MY, Cho JY. Scutellarein Reduces Inflammatory Responses by Inhibiting Src Kinase Activity. Korean J Physiol Pharmacol. 2015; 19:441–449. PMID: 26330757.
Article26. Kim HG, Yang WS, Sung GH, Kim JH, Baek GS, Kim E, Yang S, Park YC, Sung JM, Yoon DH, Kim TW, Hong S, Kim JH, Cho JY. IKK β-Targeted Anti-Inflammatory Activities of a Butanol Fraction of Artificially Cultivated Cordyceps pruinosa Fruit Bodies. Evid Based Complement Alternat Med. 2014; 2014:562467. PMID: 25132860.27. Lee JO, Jeong D, Kim MY, Cho JY. ATP-Binding Pocket-Targeted Suppression of Src and Syk by Luteolin Contributes to Its Anti-Inflammatory Action. Mediators Inflamm. 2015; 2015:967053. PMID: 26236111.
Article28. Dung TT, Kim SC, Yoo BC, Sung GH, Yang WS, Kim HG, Park JG, Rhee MH, Park KW, Yoon K, Lee Y, Hong S, Kim JH, Cho JY. (5-Hydroxy-4-oxo-4H-pyran-2-yl)methyl 6-hydroxynaphthalene-2-carboxylate, a kojic acid derivative, inhibits inflammatory mediator production via the suppression of Syk/Src and NF-κB activation. Int Immunopharmacol. 2014; 20:37–45. PMID: 24583147.
Article29. Piechota-Polanczyk A, Fichna J. Review article: the role of oxidative stress in pathogenesis and treatment of inflammatory bowel diseases. Naunyn Schmiedebergs Arch Pharmacol. 2014; 387:605–620. PMID: 24798211.
Article30. Karin M. NF-kappaB as a critical link between inflammation and cancer. Cold Spring Harb Perspect Biol. 2009; 1:a000141. PMID: 20066113.31. Yu T, Lee S, Yang WS, Jang HJ, Lee YJ, Kim TW, Kim SY, Lee J, Cho JY. The ability of an ethanol extract of Cinnamomum cassia to inhibit Src and spleen tyrosine kinase activity contributes to its antiinflammatory action. J Ethnopharmacol. 2012; 139:566–573. PMID: 22155395.
Article32. Seok Yang W, Lee J, Woong Kim T, Hye Kim J, Lee S, Hee Rhee M, Hong S, Youl Cho J. Src/NF-κB-targeted inhibition of LPS-induced macrophage activation and dextran sodium sulphate-induced colitis by Archidendron clypearia methanol extract. J Ethnopharmacol. 2012; 142:287–293. PMID: 22537838.
Article33. Yoon JY, Jeong HY, Kim SH, Kim HG, Nam G, Kim JP, Yoon DH, Hwang H, Kimc TW, Hong S, Cho JY. Methanol extract of Evodia lepta displays Syk/Src-targeted anti-inflammatory activity. J Ethnopharmacol. 2013; 148:999–1007. PMID: 23747536.
Article34. Yang WS, Ko J, Kim E, Kim JH, Park JG, Sung NY, Kim HG, Yang S, Rho HS, Hong YD, Shin SS, Cho JY. 21-O-angeloyltheasapogenol E3, a novel triterpenoid saponin from the seeds of tea plants, inhibits macrophage-mediated inflammatory responses in a NF-κB-dependent manner. Mediators Inflamm. 2014; 2014:658351. PMID: 25477714.
Article35. Bustamante J, Boisson-Dupuis S, Jouanguy E, Picard C, Puel A, Abel L, Casanova JL. Novel primary immunodeficiencies revealed by the investigation of paediatric infectious diseases. Curr Opin Immunol. 2008; 20:39–48. PMID: 18083507.
Article36. Lee YG, Lee J, Byeon SE, Yoo DS, Kim MH, Lee SY, Cho JY. Functional role of Akt in macrophage-mediated innate immunity. Front Biosci (Landmark Ed). 2011; 16:517–530. PMID: 21196185.
Article37. Bai D, Ueno L, Vogt PK. Akt-mediated regulation of NFkappaB and the essentialness of NFkappaB for the oncogenicity of PI3K and Akt. Int J Cancer. 2009; 125:2863–2870. PMID: 19609947.38. Byeon SE, Yi YS, Oh J, Yoo BC, Hong S, Cho JY. The role of Src kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2012; 2012:512926. PMID: 23209344.
Article39. Yi YS, Son YJ, Ryou C, Sung GH, Kim JH, Cho JY. Functional roles of Syk in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014; 2014:270302. PMID: 25045209.
Article40. Lee YG, Chain BM, Cho JY. Distinct role of spleen tyrosine kinase in the early phosphorylation of inhibitor of kappaB alpha via activation of the phosphoinositide-3-kinase and Akt pathways. Int J Biochem Cell Biol. 2009; 41:811–821. PMID: 18775507.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pyrrole-Derivative of Chalcone, (E)-3-Phenyl-1-(2-Pyrrolyl)-2-Propenone, Inhibits Inflammatory Responses via Inhibition of Src, Syk, and TAK1 Kinase Activities
- Fisetin Suppresses Macrophage-Mediated Inflammatory Responses by Blockade of Src and Syk
- The Regulatory Mechanism of Src Familly Kinase in Lipopolysaccharide (LPS) induced HF-kappaB Activation Pathway
- Beauvericin, a cyclic peptide, inhibits inflammatory responses in macrophages by inhibiting the NF-κB pathway
- Differential Regulation of NF-kappaB Signaling during Human Cytomegalovirus Infection