Nutr Res Pract.
2008 Dec;2(4):322-325. 10.4162/nrp.2008.2.4.322.
Kaempferol induced the apoptosis via cell cycle arrest in human breast cancer MDA-MB-453 cells
- Affiliations
-
- 1Cancer Research Institute, The Catholic University of Korea, 505 Banpo-dong, Seocho-gu, Seoul 137-040, Korea. Ahnlab1@catholic.ac.kr
- KMID: 2139210
- DOI: http://doi.org/10.4162/nrp.2008.2.4.322
Abstract
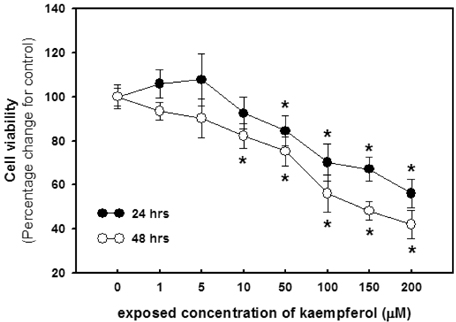
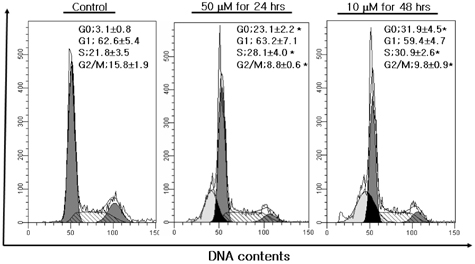
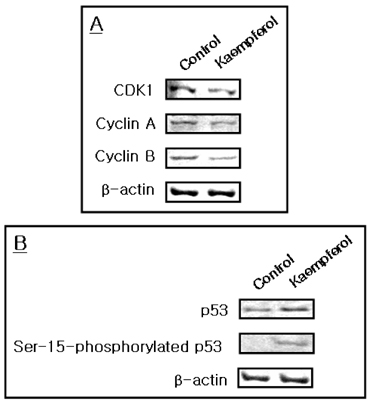
- The aim of present study was to investigate the effects of kaempferol on cellular proliferation and cell cycle arrest and explore the mechanism for these effects in human breast carcinoma MDA-MB-453 cells. Cells were treated with kaempferol at various concentrations (ranging from 1 to 200 microM) for 24 and 48 hrs. Kaempferol significantly inhibited cancer cell growth in cells exposed to 50 and 10 microM of kaempferol and incubated for 24 and 48 hrs, respectively. Exposure to kaempferol resulted in cell cycle arrest at the G2/M phase. Of the G2/M-phase related proteins, kaempferol down-regulated CDK1 and cyclin A and B in cells exposed to kaempferol. In addition, small DNA fragments at the sub-G0 phase were increased by up to 23.12 and 31.90% at 10 and 50 microM incubated for 24 and 48 hrs, respectively. The kaempferol-induced apoptosis was associated with the up-regulation of p53. In addition, the phosphorylation of p53 at the Ser-15 residue was observed with kaempferol. Kaempferol inhibits cell proliferation by disrupting the cell cycle, which is strongly associated with the induction of arrest at G2/M phase and may induce apoptosis via p53 phosphorylation in human breast carcinoma MDA-MB-453 cells.
MeSH Terms
Figure
Reference
-
1. Bestwick CS, Milne L, Duthie SJ. Kaempferol induced inhibition of HL-60 cell growth results from a heterogeneous response, dominated by cell cycle alterations. Chem Biol Interact. 2007. 170:76–85.
Article2. Casagrande F, Darbon JM. Effects of structurally related flavonoids on cell cycle progression of human melanoma cells: regulation of cyclin-dependent kinases CDK2 and CDK1. Biochem Pharmacol. 2001. 61:1205–1215.
Article3. Chen D, Daniel KG, Chen MS, Kuhn DJ, Landis-Piwowar KR, Dou QP. Dietary flavonoids as proteasome inhibitors and apoptosis inducers in human leukemia cells. Biochem Pharmacol. 2005. 69:1421–1432.
Article4. de Vries JH, Hollman PC, Meyboom S, Buysman MN, Zock PL, van Staveren WA, Katan MB. Plasma concentrations and urinary excretion of the antioxidant flavonols quercetin and kaempferol as biomarkers for dietary intake. Am J Clin Nutr. 1998. 68:60–65.
Article5. Fesik SW. Promoting apoptosis as a strategy for cancer drug discovery. Nat Rev Cancer. 2005. 5:876–885.
Article6. Galati G, O'Brien PJ. Potential toxicity of flavonoids and other dietary phenolics: significance for their chemopreventive and anticancer properties. Free Radic Biol Med. 2004. 37:287–303.
Article7. Guimaraes DP, Hainaut P. TP53: a key gene in human cancer. Biochimie. 2002. 84:83–93.
Article8. Hung H. Inhibition of estrogen receptor alpha expression and function in MCF-7 cells by kaempferol. J Cell Physiol. 2004. 198:197–208.
Article9. Ito K, Nakazato T, Yamato K, Miyakawa Y, Yamada T, Hozumi N, Segawa K, Ikeda Y, Kizaki M. Induction of apoptosis in leukemic cells by homovanillic acid derivative, capsaicin, through oxidative stress: implication of phosphorylation of p53 at Ser-15 residue by reactive oxygen species. Cancer Res. 2004. 64:1071–1078.
Article10. Jessus C, Beach D. Oscillation of MPF is accompanied by periodic association between cdc25 and cdc2-cyclin B. Cell. 1992. 68:323–332.
Article11. Jiang C, Hu H, Malewicz B, Wang Z, Lü J. Selenite-induced p53 Ser-15 phosphorylation and caspase-mediated apoptosis in LNCaP human prostate cancer cells. Mol Cancer Ther. 2004. 3:877–884.12. Knowles LM, Zigrossi DA, Tauber RA, Hightower C, Milner JA. Flavonoids suppress androgen-independent human prostate tumor proliferation. Nutr Cancer. 2000. 38:116–122.
Article13. Kuo PC, Liu HF, Chao JI. Survivin and p53 modulate quercetin-induced cell growth inhibition and apoptosis in human lung carcinoma cells. J Biol Chem. 2004. 279:55875–55885.
Article14. Leung HW, Lin CJ, Hour MJ, Yang WH, Wang MY, Lee HZ. Kaempferol induces apoptosis in human lung non-small carcinoma cells accompanied by an induction of antioxidant enzymes. Food Chem Toxicol. 2007. 45:2005–2013.
Article15. Marfak A, Trouillas P, Allais DP, Champavier Y, Calliste CA, Duroux JL. Radiolysis of kaempferol in water/methanol mixtures. Evaluation of antioxidant activity of kaempferol and products formed. J Agric Food Chem. 2003. 51:1270–1277.
Article16. Miean KH, Mohamed S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. J Agric Food Chem. 2001. 49:3106–3112.
Article17. Morla AO, Draetta G, Beach D, Wang JY. Reversible tyrosine phosphorylation of cdc2: dephosphorylation accompanies activation during entry into mitosis. Cell. 1989. 58:193–203.
Article18. Mota I, Wong D, Sadun EH. Mouse homocytotropic antibodies. I. Specific differentiation between mouse 7S gamma 1 and mouse reagin-like antibodies. Life Sci. 1968. 7:1289–1293.
Article19. Nguyen TT, Tran E, Ong CK, Lee SK, Do PT, Huynh TT, Nguyen TH, Lee JJ, Tan Y, Ong CS, Huynh H. Kaempferol-induced growth inhibition and apoptosis in A549 lung cancer cells is mediated by activation of MEK-MAPK. J Cell Physiol. 2003a. 197:110–121.
Article20. Nguyen TT, Tran E, Ong CK, Lee SK, Do PT, Huynh TT, Nguyen TH, Lee JJ, Tan Y, Ong CS, Huynh H. Kaempferol-induced growth inhibition and apoptosis in A549 lung cancer cells is mediated by activation of MEK-MAPK. J Cell Physiol. 2003b. 197:110–121.
Article21. Park JS, Rho HS, Kim DH, Chang IS. Enzymatic preparation of kaempferol from green tea seed and its antioxidant activity. J Agric Food Chem. 2006. 54:2951–2956.
Article22. Prouillet C, Mazière JC, Mazière C, Wattel A, Brazier M, Kamel S. Stimulatory effect of naturally occurring flavonols quercetin and kaempferol on alkaline phosphatase activity in MG-63 human osteoblasts through ERK and estrogen receptor pathway. Biochem Pharmacol. 2004. 67:1307–1313.
Article23. Ren W, Qiao Z, Wang H, Zhu L, Zhang L. Flavonoids: promising anticancer agents. Med Res Rev. 2003. 23:519–534.
Article24. Ross JA, Kasum CM. Dietary flavonoids: bioavailability, metabolic effects, and safety. Annu Rev Nutr. 2002. 22:19–34.
Article25. Sun SY, Hail N, Lotan R. Apoptosis as a novel target for cancer chemoprevention. J Natl Cancer Inst. 2004. 96:662–672.
Article26. Yoshida T, Konishi M, Horinaka M, Yasuda T, Goda AE, Taniguchi H, Yano K, Wakada M, Sakai T. Kaempferol sensitizes colon cancer cells to TRAIL-induced apoptosis. Biochem Biophys Res Commun. 2008. 375:129–133.
Article27. Zhang Q, Zhao XH, Wang ZJ. Flavones and flavonols exert cytotoxic effects on a human oesophageal adenocarcinoma cell line (OE33) by causing G2/M arrest and inducing apoptosis. Food Chem Toxicol. 2008. 46:2042–2053.
Article28. Zhang Z, Li M, Rayburn ER, Hill DL, Zhang R, Wang H. Oncogenes as novel targets for cancer therapy (part IV): regulators of the cell cycle and apoptosis. Am J Pharmacogenomics. 2005. 5:397–407.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Dioscin Decreases Breast Cancer Stem-like Cell Proliferation via Cell Cycle Arrest by Modulating p38 Mitogen-activated Protein Kinase and AKT/mTOR Signaling Pathways
- Imidazole Antifungal Drugs Inhibit the Cell Proliferation and Invasion of Human Breast Cancer Cells
- Peroxisome proliferator-activated receptor gamma activator inhibits cell growth of MDA-MB-231 breast cancer cells through induction of apoptosis
- Growth inhibition by fusidic acid in cervical, thyroid, and breast carcinoma cell lines
- Kaempferol Synergistically Enhances Cisplatin-induced Apoptosis and Cell Cycle Arrest in Colon Cancer Cells