J Adv Prosthodont.
2015 Apr;7(2):166-171. 10.4047/jap.2015.7.2.166.
Evaluation of antibacterial activity and osteoblast-like cell viability of TiN, ZrN and (Ti(1-x)Zr(x))N coating on titanium
- Affiliations
-
- 1Department of Prosthodontics, School of Dentistry, Chonnam National University, Gwangju, Republic of Korea. mcnihil@jnu.ac.kr
- 2Department of Materials Science and Engineering, Chonnam National University, Gwangju, Republic of Korea.
- 3Department of Oral Microbiology, School of Dentistry, Chonnam National University, Gwangju, Republic of Korea.
- 4RIS Foundation for Advanced Biomaterials, Chonnam National University, Gwangju, Republic of Korea.
- KMID: 2118248
- DOI: http://doi.org/10.4047/jap.2015.7.2.166
Abstract
- PURPOSE
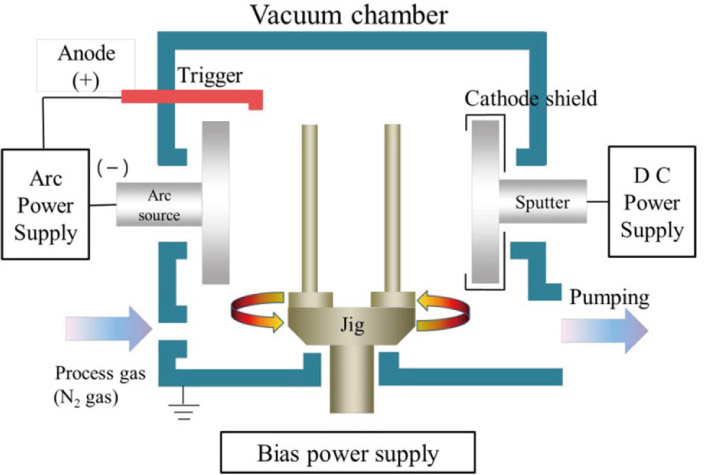
The aim of this study was to evaluate antibacterial activity and osteoblast-like cell viability according to the ratio of titanium nitride and zirconium nitride coating on commercially pure titanium using an arc ion plating system.
MATERIALS AND METHODS
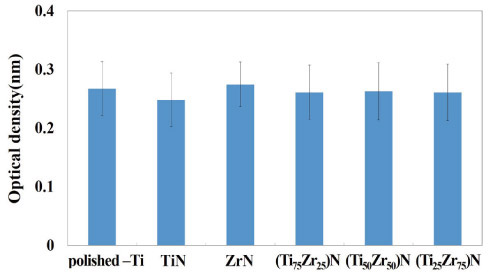
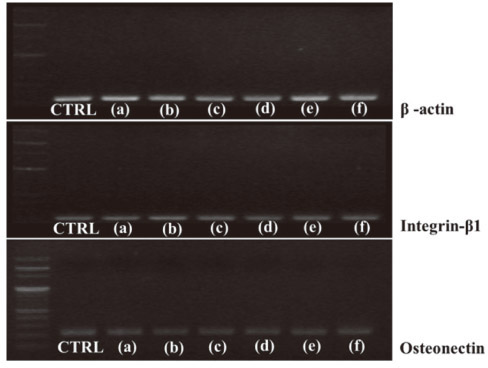
Polished titanium surfaces were used as controls. Surface topography was observed by scanning electron microscopy, and surface roughness was measured using a two-dimensional contact stylus profilometer. Antibacterial activity was evaluated against Streptococcus mutans and Porphyromonas gingivalis with the colony-forming unit assay. Cell compatibility, mRNA expression, and morphology related to human osteoblast-like cells (MG-63) on the coated specimens were determined by the XTT assay and reverse transcriptase-polymerase chain reaction.
RESULTS
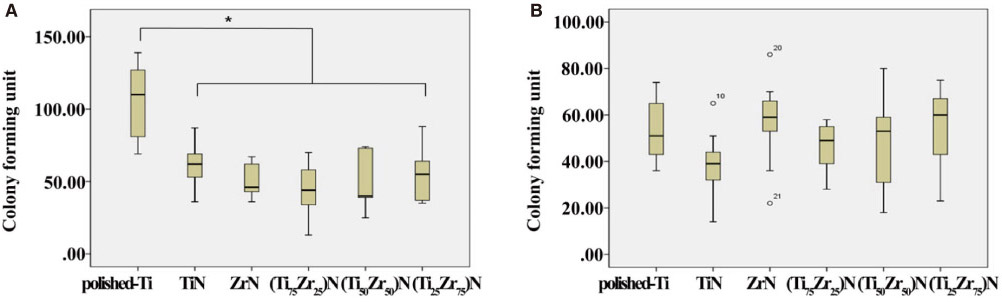
The number of S. mutans colonies on the TiN, ZrN and (Ti(1-x)Zr(x))N coated surface decreased significantly compared to those on the non-coated titanium surface (P<0.05).
CONCLUSION
The number of P. gingivalis colonies on all surfaces showed no significant differences. TiN, ZrN and (Ti(1-x)Zr(x))N coated titanium showed antibacterial activity against S. mutans related to initial biofilm formation but not P. gingivalis associated with advanced periimplantitis, and did not influence osteoblast-like cell viability.
Keyword
MeSH Terms
Figure
Reference
-
1. Niinomi M. Metallic biomaterials. J Artif Organs. 2008; 11:105–110.2. Grössner-Schreiber B, Griepentrog M, Haustein I, Müller WD, Lange KP, Briedigkeit H, Göbel UB. Plaque formation on surface modified dental implants. An in vitro study. Clin Oral Implants Res. 2001; 12:543–551.3. Watzak G, Zechner W, Ulm C, Tangl S, Tepper G, Watzek G. Histologic and histomorphometric analysis of three types of dental implants following 18 months of occlusal loading: a preliminary study in baboons. Clin Oral Implants Res. 2005; 16:408–416.4. Isidor F. Influence of forces on peri-implant bone. Clin Oral Implants Res. 2006; 17:8–18.5. Becker W, Becker BE, Newman MG, Nyman S. Clinical and microbiologic findings that may contribute to dental implant failure. Int J Oral Maxillofac Implants. 1990; 5:31–38.6. Jung CW. Peri-implant disease and GBR. 1st ed. Seoul: Narae publishing;2001. p. 2–7.7. Elias CN, Figueira DC, Rios PR. Influence of the coating material on the loosing of dental implant abutment screw joints. Mater Sci Eng C. 2006; 26:1361–1366.8. Chou WJ, Yu GP, Huang JH. Corrosion resistance of ZrN films on AISI 304 stainless steel substrate. Surf Coat Technol. 2003; 167:59–67.9. Moon BH, Choe HC, Brantley WA. Surface characteristics of TiN/ZrN coated nanotubular structure on the Ti-35Ta-xHf alloy for bio-implant applications. Appl Surf Sci. 2012; 258:2088–2092.10. Damaschek R, Strydom IL, Bergmann H. Improved adhesion of TiN deposited on prenitrided steels. Surf Eng. 1997; 13:128–132.11. Groessner-Schreiber B, Neubert A, Müller WD, Hopp M, Griepentrog M, Lange KP. Fibroblast growth on surfacemodified dental implants: an in vitro study. J Biomed Mater Res A. 2003; 64:591–599.12. Chollet L, Perry AJ. The stress in ion-plated HfN and TiN coatings. Thin Solid Films. 1985; 123:223–234.13. Jeong YH, Kwag DM, Chung CH, Kim WG, Choe HC. Corrosion characteristics and surface morphologies of TiN and ZrN film on the abutment screw by Arc-ion coating(2). Corrosion Sci Technol. 2011; 10:212–217.14. Nakazato G, Tsuchiya H, Sato M, Yamauchi M. In vivo plaque formation on implant materials. Int J Oral Maxillofac Implants. 1989; 4:321–326.15. Mombelli A, Lang NP. Microbial aspects of implant dentistry. Periodontol 2000. 1994; 4:74–80.16. Tai CN, Koh ES, Akari K. Macroparticles on TiN films prepared by the arc ion plating process. Surf Coat Technol. 1990; 43/44:324–335.17. Quirynen M, van der Mei HC, Bollen CM, Schotte A, Marechal M, Doornbusch GI, Naert I, Busscher HJ, van Steenberghe D. An in vivo study of the influence of the surface roughness of implants on the microbiology of supraand subgingival plaque. J Dent Res. 1993; 72:1304–1309.18. Lin NM, Huang XB, Zou JJ, Zhang XY, Qin L, Fan AL, Tang B. Effects of plasma nitriding and multiple arc ion plating TiN coating on bacterial adhesion of commercial pure titanium via in vitro investigations. Surf Coat Technol. 2012; 209:212–215.19. Bollen CM, Lambrechts P, Quirynen M. Comparison of surface roughness of oral hard materials to the threshold surface roughness for bacterial plaque retention: a review of the literature. Dent Mater. 1997; 13:258–269.20. Ryan V, Hart TR, Schiller R. Size determination of Streptococcus mutans 10499 by laser light scattering. Biophys J. 1980; 31:313–324.21. Jeyachandran YL, Narayandass SK, Mangalaraj D, Bao CY, Martin PJ. The effect of surface composition of titanium films on bacterial adhesion. Biomed Mater. 2006; 1:L1–L5.22. Jeyachandran YL, Venkatachalam S, Karunagaran B, Narayandass SK, Mangalaraj D, Bao CY, Zhang CL. Bacterial adhesion studies on titanium, titanium nitride and modified hydroxyapatite thin films. Mater Sci Eng C. 2007; 27:35–41.23. Yoshinari M, Oda Y, Kato T, Okuda K. Influence of surface modifications to titanium on antibacterial activity in vitro. Biomaterials. 2001; 22:2043–2048.24. Ata-Ali J, Candel-Marti ME, Flichy-Fernández AJ, Peñarrocha-Oltra D, Balaguer-Martinez JF, Peñarrocha Diago M. Peri-implantitis: associated microbiota and treatment. Med Oral Patol Oral Cir Bucal. 2011; 16:e937–e943.25. Anselme K. Osteoblast adhesion on biomaterials. Biomaterials. 2000; 21:667–681.26. Schwartz Z, Lohmann CH, Vocke AK, Sylvia VL, Cochran DL, Dean DD, Boyan BD. Osteoblast response to titanium surface roughness and 1alpha,25-(OH)(2)D(3) is mediated through the mitogen-activated protein kinase (MAPK) pathway. J Biomed Mater Res. 2001; 56:417–426.27. Anselme K. Osteoblast adhesion on biomaterials. Biomaterials. 2000; 21:667–681.28. Roach HI. Why does bone matrix contain non-collagenous proteins? The possible roles of osteocalcin, osteonectin, osteopontin and bone sialoprotein in bone mineralisation and resorption. Cell Biol Int. 1994; 18:617–628.29. zur Nieden NI, Kempka G, Ahr HJ. In vitro differentiation of embryonic stem cells into mineralized osteoblasts. Differentiation. 2003; 71:18–27.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A study on mechanical properties of TiN, ZrN and WC coated film on the titanium alloy surface
- Effects of surface coating on the screw release of dental implant screw
- Osteoblastic behavior to zirconium coating on Ti-6Al-4V alloy
- The effect of Zirconium Nitride coating on shear bond strength with denture base resin in Co-Cr alloy and titanium alloy
- Mechanical properties of tin coated film with various coating thickness on titanium alloy