Chonnam Med J.
2010 Dec;46(3):129-139. 10.4068/cmj.2010.46.3.129.
Mechanism of Ischemia and Reperfusion Injury to the Heart: From the Viewpoint of Nitric Oxide and Mitochondria
- Affiliations
-
- 1Cardiology Division, Department of Internal Medicine, Kyung Hee University Hospital, Seoul, Korea. mylovekw@hanmail.net
- KMID: 2116766
- DOI: http://doi.org/10.4068/cmj.2010.46.3.129
Abstract
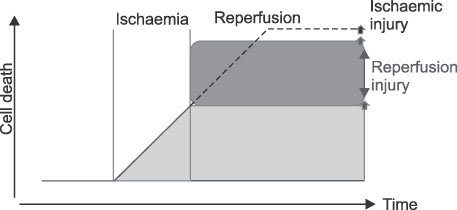
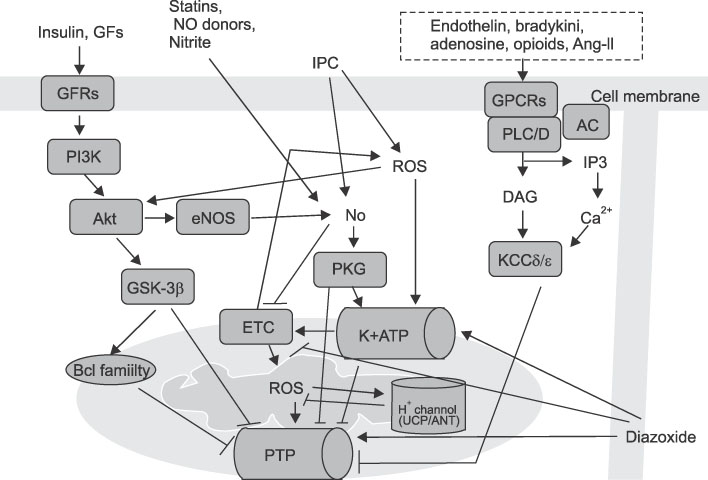
- After an acute myocardial infarction, early and successful myocardial reperfusion is the most effective strategy for reducing the size of a myocardial infarct and improving the clinical outcome. However, the process of restoring blood flow to the ischemic myocardium can induce injury. This phenomenon,termed myocardial reperfusion injury, can paradoxically reduce the beneficial effects of myocardial reperfusion and lead to lethal damage to myocardium. During cardiac ischemia-reperfusion (IR) injury, excessive generation of reactive oxygen species (ROS), overload of intracellular Ca2+, H+ leakage at the mitochondrial level, inflammation, and metabolic modulations lead to opening of the mitochondrial permeability transition pore(PTP) on reperfusion. This can result in the depletion of ATP, irreversible oxidation of proteins, lipids, and DNA within the cardiomyocyte, and can trigger apoptosis. In contrast, mitochondria also plays an important role in the cardioprotective signaling processes of ischemic preconditioning (IPC), to prevent IR injury. Nitric oxide (NO) generated constitutively within the heart has long been known to influence myocardial function. But, Nitric oxide (NO) has emerged as a potent effector molecule for a variety of cardioprotective strategies, including IPC. Whereas NO is most noted for its activation of the "classic" soluble guanylate cyclase (sGC) signaling pathway, emerging evidence indicates that NO can directly act on mitochondria, independent of the sGC pathway, affording acute cardioprotection against IR injury. These effects of NO on mitochondria and mitochondrial role during IR injury are the focus of this review.
MeSH Terms
-
Adenosine Triphosphate
Apoptosis
DNA
Guanylate Cyclase
Heart
Inflammation
Ischemia
Ischemic Preconditioning
Mitochondria
Myocardial Infarction
Myocardial Reperfusion
Myocardial Reperfusion Injury
Myocardium
Myocytes, Cardiac
Nitric Oxide
Permeability
Proteins
Reactive Oxygen Species
Reperfusion
Reperfusion Injury
Adenosine Triphosphate
DNA
Guanylate Cyclase
Nitric Oxide
Proteins
Reactive Oxygen Species
Figure
Reference
-
1. Garcia-Dorado D, Piper HM. Postconditioning: reperfusion of "reperfusion injury" after hibernation. Cardiovasc Res. 2006. 69:1–3.
Article2. Jennings RB, Sommers HM, Smyth GA, Flack HA, Linn H. Myocardial necrosis induced by temporary occlusion of a coronary artery in the dog. Arch Pathol. 1960. 70:68–78.3. Przyklenk K. Lethal Myocardial "Reperfusion Injury": The Opinions of Good Men. J Thromb Thrombolysis. 1997. 4:5–6.4. Braunwald E, Kloner RA. Myocardial reperfusion: a double-edged sword? J Clin Invest. 1985. 76:1713–1719.
Article5. Vinten-Johansen J, Johnston WE, Mills SA, Faust KB, Geisinger KR, DeMasi RJ, et al. Reperfusion injury after temporary coronary occlusion. J Thorac Cardiovasc Surg. 1988. 95:960–968.
Article6. Liu X, Kim CN, Yang J, Jemmerson R, Wang X. Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell. 1996. 86:147–157.
Article7. Bolli R. Cardioprotective function of inducible nitric oxide synthase and role of nitric oxide in myocardial ischemia and preconditioning: an overview of a decade of research. J Mol Cell Cardiol. 2001. 33:1897–1918.
Article8. Kelly RA, Balligand JL, Smith TW. Nitric oxide and cardiac function. Circ Res. 1996. 79:363–380.
Article9. Massion PB, Feron O, Dessy C, Balligand JL. Nitric oxide and cardiac function: ten years after, and continuing. Circ Res. 2003. 93:388–398.10. Schulz R, Rassaf T, Massion PB, Kelm M, Balligand JL. Recent advances in the understanding of the role of nitric oxide in cardiovascular homeostasis. Pharmacol Ther. 2005. 108:225–256.
Article11. Ziolo MT, Kohr MJ, Wang H. Nitric oxide signaling and the regulation of myocardial function. J Mol Cell Cardiol. 2008. 45:625–632.
Article12. Seddon M, Shah AM, Casadei B. Cardiomyocytes as effectors of nitric oxide signalling. Cardiovasc Res. 2007. 75:315–326.
Article13. Pinsky DJ, Patton S, Mesaros S, Brovkovych V, Kubaszewski E, Grunfeld S, et al. Mechanical transduction of nitric oxide synthesis in the beating heart. Circ Res. 1997. 81:372–379.
Article14. Irani K, Xia Y, Zweier JL, Sollott SJ, Der CJ, Fearon ER, et al. Mitogenic signaling mediated by oxidants in Ras-transformed fibroblasts. Science. 1997. 275:1649–1652.
Article15. Dhalla NS, Elmoselhi AB, Hata T, Makino N. Status of myocardial antioxidants in ischemia-reperfusion injury. Cardiovasc Res. 2000. 47:446–456.
Article16. Hess ML, Okabe E, Kontos HA. Proton and free oxygen radical interaction with the calcium transport system of cardiac sarcoplasmic reticulum. J Mol Cell Cardiol. 1981. 13:767–772.
Article17. Rosen H, Klebanoff SJ. Hydroxyl radical generation by polymorphonuclear leukocytes measured by electron spin resonance spectroscopy. J Clin Invest. 1979. 64:1725–1729.
Article18. Zweier JL, Talukder MA. The role of oxidants and free radicals in reperfusion injury. Cardiovasc Res. 2006. 70:181–190.
Article19. Piper HM, Garcia-Dorado D, Ovize M. A fresh look at reperfusion injury. Cardiovasc Res. 1998. 38:291–300.
Article20. Lemasters JJ, Bond JM, Chacon E, Harper IS, Kaplan SH, Ohata H, et al. The pH paradox in ischemia-reperfusion injury to cardiac myocytes. EXS. 1996. 76:99–114.
Article21. Bond JM, Herman B, Lemasters JJ. Protection by acidotic pH against anoxia/reoxygenation injury to rat neonatal cardiac myocytes. Biochem Biophys Res Commun. 1991. 179:798–803.
Article22. Kim JS, Jin Y, Lemasters JJ. Reactive oxygen species, but not Ca2+ overloading, trigger pH- and mitochondrial permeability transition-dependent death of adult rat myocytes after ischemia-reperfusion. Am J Physiol Heart Circ Physiol. 2006. 290:H2024–H2034.23. Vinten-Johansen J. Involvement of neutrophils in the pathogenesis of lethal myocardial reperfusion injury. Cardiovasc Res. 2004. 61:481–497.
Article24. Jonassen AK, Sack MN, Mjøs OD, Yellon DM. Myocardial protection by insulin at reperfusion requires early administration and is mediated via Akt and p70s6 kinase cell-survival signaling. Circ Res. 2001. 89:1191–1198.
Article25. Apstein CS, Opie LH. A challenge to the metabolic approach to myocardial ischaemia. Eur Heart J. 2005. 26:956–959.
Article26. Opie LH. The glucose hypothesis: relation to acute myocardial ischemia. J Mol Cell Cardiol. 1970. 1:107–115.27. Mehta SR, Yusuf S, Diaz R, Zhu J, Pais P, Xavier D, et al. Effect of glucose-insulin-potassium infusion on mortality in patients with acute ST-segment elevation myocardial infarction: the CREATE-ECLA randomized controlled trial. JAMA. 2005. 293:437–446.
Article28. Timmers L, Henriques JP, de Kleijn DP, Devries JH, Kemperman H, Steendijk P, et al. Exenatide reduces infarct size and improves cardiac function in a porcine model of ischemia and reperfusion injury. J Am Coll Cardiol. 2009. 53:501–510.
Article29. Facundo HT, Carreira RS, de Paula JG, Santos CC, Ferranti R, Laurindo FR, et al. Ischemic preconditioning requires increases in reactive oxygen release independent of mitochondrial K+ channel activity. Free Radic Biol Med. 2006. 40:469–479.
Article30. Sasaki N, Sato T, Ohler A, O'Rourke B, Marban E. Activation of mitochondrial ATP-dependent potassium channels by nitric oxide. Circulation. 2000. 101:439–445.
Article31. Costa AD, Garlid KD, West IC, Lincoln TM, Downey JM, Cohen MV, et al. Protein kinase G transmits the cardioprotective signal from cytosol to mitochondria. Circ Res. 2005. 97:329–336.
Article32. Kim JS, Ohshima S, Pediaditakis P, Lemasters JJ. Nitric oxide: a signaling molecule against mitochondrial permeability transition- and pH-dependent cell death after reperfusion. Free Radic Biol Med. 2004. 37:1943–1950.
Article33. Ardehali H, Chen Z, Ko Y, Mejia-Alvarez R, Marban E. Multiprotein complex containing succinate dehydrogenase confers mitochondrial ATP-sensitive K+ channel activity. Proc Natl Acad Sci USA. 2004. 101:11880–11885.
Article34. Lacza Z, Snipes JA, Miller AW, Szabó C, Grover G, Busija DW. Heart mitochondria contain functional ATP-dependent K+ channels. J Mol Cell Cardiol. 2003. 35:1339–1347.
Article35. Schäfer G, Wegener C, Portenhauser R, Bojanovski D. Diazoxide, an inhibitor of succinate oxidation. Biochem Pharmacol. 1969. 18:2678–2681.
Article36. Holmuhamedov EL, Jahangir A, Oberlin A, Komarov A, Colombini M, Terzic A. Potassium channel openers are uncoupling protonophores: implication in cardioprotection. FEBS Lett. 2004. 568:167–170.
Article37. Hanley PJ, Daut J. K(ATP) channels and preconditioning: a re-examination of the role of mitochondrial K(ATP) channels and an overview of alternative mechanisms. J Mol Cell Cardiol. 2005. 39:17–50.
Article38. Dröse S, Brandt U, Hanley PJ. K+-independent actions of diazoxide question the role of inner membrane KATP channels in mitochondrial cytoprotective signaling. J Biol Chem. 2006. 281:23733–23739.
Article39. Hanley PJ, Mickel M, Löffler M, Brandt U, Daut J. K(ATP) channel-independent targets of diazoxide and 5-hydroxydecanoate in the heart. J Physiol. 2002. 542:735–741.
Article40. Brdiczka DG, Zorov DB, Sheu SS. Mitochondrial contact sites: their role in energy metabolism and apoptosis. Biochim Biophys Acta. 2006. 1762:148–163.
Article41. Nadtochiy SM, Tompkins AJ, Brookes PS. Different mechanisms of mitochondrial proton leak in ischaemia/reperfusion injury and preconditioning: implications for pathology and cardioprotection. Biochem J. 2006. 395:611–618.
Article42. Gori T, Sicuro S, Dragoni S, Donati G, Forconi S, Parker JD. Sildenafil prevents endothelial dysfunction induced by ischemia and reperfusion via opening of adenosine triphosphate-sensitive potassium channels: a human in vivo study. Circulation. 2005. 111:742–746.
Article43. Hausenloy DJ, Yellon DM. The mitochondrial permeability transition pore: its fundamental role in mediating cell death during ischaemia and reperfusion. J Mol Cell Cardiol. 2003. 35:339–341.
Article44. Griffiths EJ, Halestrap AP. Mitochondrial non-specific pores remain closed during cardiac ischaemia, but open upon reperfusion. Biochem J. 1995. 307:93–98.
Article45. Kim YM, Bombeck CA, Billiar TR. Nitric oxide as a bifunctional regulator of apoptosis. Circ Res. 1999. 84:253–256.
Article46. Ramachandran A, Levonen AL, Brookes PS, Ceaser E, Shiva S, Barone MC, et al. Mitochondria, nitric oxide, and cardiovascular dysfunction. Free Radic Biol Med. 2002. 33:1465–1474.
Article47. Yabuki M, Tsutsui K, Horton AA, Yoshioka T, Utsumi K. Caspase activation and cytochrome c release during HL-60 cell apoptosis induced by a nitric oxide donor. Free Radic Res. 2000. 32:507–514.
Article48. Brookes PS, Salinas EP, Darley-Usmar K, Eiserich JP, Freeman BA, Darley-Usmar VM, et al. Concentration-dependent effects of nitric oxide on mitochondrial permeability transition and cytochrome c release. J Biol Chem. 2000. 275:20474–20479.
Article49. Haendeler J, Zeiher AM, Dimmeler S. Nitric oxide and apoptosis. Vitam Horm. 1999. 57:49–77.
Article50. Ramachandran A, Levonen AL, Brookes PS, Ceaser E, Shiva S, Barone MC, et al. Mitochondria, nitric oxide, and cardiovascular dysfunction. Free Radic Biol Med. 2002. 33:1465–1474.
Article51. Wang G, Liem DA, Vondriska TM, Honda HM, Korge P, Pantaleon DM, et al. Nitric oxide donors protect murine myocardium against infarction via modulation of mitochondrial permeability transition. Am J Physiol Heart Circ Physiol. 2005. 288:H1290–H1295.
Article52. Hoffmann J, Haendeler J, Aicher A, Rössig L, Vasa M, Zeiher AM, et al. Aging enhances the sensitivity of endothelial cells toward apoptotic stimuli: important role of nitric oxide. Circ Res. 2001. 89:709–715.
Article53. Hardy DL, Clark JB, Darley-Usmar VM, Smith DR. Reoxygenation of the hypoxic myocardium causes a mitochondrial complex I defect. Biochem Soc Trans. 1990. 18:549.
Article54. Lesnefsky EJ, Moghaddas S, Tandler B, Kerner J, Hoppel CL. Mitochondrial dysfunction in cardiac disease: ischemia--reperfusion, aging, and heart failure. J Mol Cell Cardiol. 2001. 33:1065–1089.
Article55. Paradies G, Ruggiero FM, Petrosillo G, Quagliariello E. Peroxidative damage to cardiac mitochondria: cytochrome oxidase and cardiolipin alterations. FEBS Lett. 1998. 424:155–158.
Article56. Asimakis GK, Conti VR. Myocardial ischemia: correlation of mitochondrial adenine nucleotide and respiratory function. J Mol Cell Cardiol. 1984. 16:439–447.
Article57. Shug AL, Subramanian R. Modulation of adenine nucleotide translocase activity during myocardial ischemia. Z Kardiol. 1987. 76:Suppl 5. 26–33.58. Lucas DT, Szweda LI. Declines in mitochondrial respiration during cardiac reperfusion: age-dependent inactivation of alpha-ketoglutarate dehydrogenase. Proc Natl Acad Sci USA. 1999. 96:6689–6693.
Article59. Sadek HA, Humphries KM, Szweda PA, Szweda LI. Selective inactivation of redox-sensitive mitochondrial enzymes during cardiac reperfusion. Arch Biochem Biophys. 2002. 406:222–228.
Article60. Lesnefsky EJ, Chen Q, Moghaddas S, Hassan MO, Tandler B, Hoppel CL. Blockade of electron transport during ischemia protects cardiac mitochondria. J Biol Chem. 2004. 279:47961–47967.
Article61. Lesnefsky EJ, Slabe TJ, Stoll MS, Minkler PE, Hoppel CL. Myocardial ischemia selectively depletes cardiolipin in rabbit heart subsarcolemmal mitochondria. Am J Physiol Heart Circ Physiol. 2001. 280:H2770–H2778.
Article62. Borutaite V, Mildaziene V, Brown GC, Brand MD. Control and kinetic analysis of ischemia-damaged heart mitochondria: which parts of the oxidative phosphorylation system are affected by ischemia? Biochim Biophys Acta. 1995. 1272:154–158.
Article63. Brookes PS, Darley-Usmar VM. Role of calcium and superoxide dismutase in sensitizing mitochondria to peroxynitrite-induced permeability transition. Am J Physiol Heart Circ Physiol. 2004. 286:H39–H46.
Article64. Darley-Usmar VM, Smith DR, O'Leary VJ, Stone D, Hardy DL, Clark JB. Hypoxia-reoxygenation induced damage in the myocardium: the role of mitochondria. Biochem Soc Trans. 1990. 18:526–528.
Article65. Di Lisa F, Canton M, Menabo R, Dodoni G, Bernardi P. Mitochondria and reperfusion injury. The role of permeability transition. Basic Res Cardiol. 2003. 98:235–241.66. Garcia-Rivas Gde J, Carvajal K, Correa F, Zazueta C. Ru360, a specific mitochondrial calcium uptake inhibitor, improves cardiac post-ischaemic functional recovery in rats in vivo. Br J Pharmacol. 2006. 149:829–837.67. Halestrap AP. Calcium, mitochondria and reperfusion injury: a pore way to die. Biochem Soc Trans. 2006. 34:232–237.
Article68. Hardy DL, Clark JB, Darley-Usmar VM, Smith DR. Reoxygenation of the hypoxic myocardium causes a mitochondrial complex I defect. Biochem Soc Trans. 1990. 18:549.
Article69. Nadtochiy SM, Burwell LS, Brookes PS. Cardioprotection and mitochondrial S-nitrosation: effects of S-nitroso-2-mercaptopropionyl glycine (SNO-MPG) in cardiac ischemia-reperfusion injury. J Mol Cell Cardiol. 2007. 42:812–825.
Article70. Stone D, Darley-Usmar V, Smith DR, O'Leary V. Hypoxia-reoxygenation induced increase in cellular Ca2+ in myocytes and perfused hearts: the role of mitochondria. J Mol Cell Cardiol. 1989. 21:963–973.
Article71. Stone D, Darley-Usmar VM, Martin JF. Parratt JR, editor. Calcium fluxes and reperfusion damage: the role of mitochondria. Myocardial Response to Acute Injury. 1992. Basingstoke, UK: Macmillan;67–79.
Article72. Tompkins AJ, Burwell LS, Digerness SB, Zaragoza C, Holman WL, Brookes PS. Mitochondrial dysfunction in cardiac ischemia-reperfusion injury: ROS from complex I, without inhibition. Biochim Biophys Acta. 2006. 1762:223–231.
Article73. Turrens JF, Beconi M, Barilla J, Chavez UB, McCord JM. Mitochondrial generation of oxygen radicals during reoxygenation of ischemic tissues. Free Radic Res Commun. 1991. 12-13(pt 2):681–689.
Article74. Lee GJ, Chae SJ, Jeong JH, Lee SR, Ha SJ, Pak YK, et al. Characterization of mitochondria isolated from normal and ischemic hearts in rats utilizing atomic force microscopy. Micron. [Epub ahead of print].
Article75. Brady NR, Hamacher-Brady A, Gottlieb RA. Proapoptotic BCL-2 family members and mitochondrial dysfunction during ischemia/reperfusion injury, a study employing cardiac HL-1 cells and GFP biosensors. Biochim Biophys Acta. 2006. 1757:667–678.
Article76. Ong SB, Subrayan S, Lim SY, Yellon DM, Davidson SM, Hausenloy DJ. Inhibiting mitochondrial fission protects the heart against ischemia/reperfusion injury. Circulation. 2010. 121:2012–2022.
Article77. Plotnikov EY, Vasileva AK, Arkhangelskaya AA, Pevzner IB, Skulachev VP, Zorov DB. Interrelations of mitochondrial fragmentation and cell death under ischemia/reoxygenation and UV-irradiation: protective effects of SkQ1, lithium ions and insulin. FEBS Lett. 2008. 582:3117–3124.
Article78. Chen Q, Camara AK, Stowe DF, Hoppel CL, Lesnefsky EJ. Modulation of electron transport protects cardiac mitochondria and decreases myocardial injury during ischemia and reperfusion. Am J Physiol Cell Physiol. 2007. 292:C137–C147.
Article79. Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986. 74:1124–1136.
Article80. Yellon DM, Downey JM. Preconditioning the myocardium: from cellular physiology to clinical cardiology. Physiol Rev. 2003. 83:1113–1151.
Article81. Kwok WM, Aizawa K. Preconditioning of the myocardium by volatile anesthetics. Curr Med Chem Cardiovasc Hematol Agents. 2004. 2:249–255.
Article82. Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, et al. Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol. 2003. 285:H579–H588.
Article83. Hausenloy D, Wynne A, Duchen M, Yellon D. Transient mitochondrial permeability transition pore opening mediates preconditioning-induced protection. Circulation. 2004. 109:1714–1717.
Article84. Hausenloy DJ, Duchen MR, Yellon DM. Inhibiting mitochondrial permeability transition pore opening at reperfusion protects against ischaemia-reperfusion injury. Cardiovasc Res. 2003. 60:617–625.
Article85. Honda HM, Ping P. Mitochondrial permeability transition in cardiac cell injury and death. Cardiovasc Drugs Ther. 2006. 20:425–432.
Article86. Jones SP, Bolli R. The ubiquitous role of nitric oxide in cardioprotection. J Mol Cell Cardiol. 2006. 40:16–23.
Article87. Hausenloy D, Wynne A, Duchen M, Yellon D. Transient mitochondrial permeability transition pore opening mediates preconditioning-induced protection. Circulation. 2004. 109:1714–1717.
Article88. Sun JZ, Tang XL, Park SW, Qiu Y, Turrens JF, Bolli R. Evidence for an essential role of reactive oxygen species in the genesis of late preconditioning against myocardial stunning in conscious pigs. J Clin Invest. 1996. 97:562–576.
Article89. Nadtochiy SM, Tompkins AJ, Brookes PS. Different mechanisms of mitochondrial proton leak in ischaemia/reperfusion injury and preconditioning: implications for pathology and cardioprotection. Biochem J. 2006. 395:611–618.
Article90. Minners J, van den Bos EJ, Yellon DM, Schwalb H, Opie LH, Sack MN. Dinitrophenol, cyclosporin A, and trimetazidine modulate preconditioning in the isolated rat heart: support for a mitochondrial role in cardioprotection. Cardiovasc Res. 2000. 47:68–73.
Article91. Pain T, Yang XM, Critz SD, Yue Y, Nakano A, Liu GS, et al. Opening of mitochondrial K(ATP) channels triggers the preconditioned state by generating free radicals. Circ Res. 2000. 87:460–466.
Article92. Alderton WK, Cooper CE, Knowles RG. Nitric oxide synthases: structure, function and inhibition. Biochem J. 2001. 357:593–615.
Article93. Brunner F, Maier R, Andrew P, Wolkart G, Zechner R, Mayer B. Attenuation of myocardial ischemia/reperfusion injury in mice with myocyte-specific overexpression of endothelial nitric oxide synthase. Cardiovasc Res. 2003. 57:55–62.
Article94. Jones SP, Greer JJ, Kakkar AK, Ware PD, Turnage RH, Hicks M, et al. Endothelial nitric oxide synthase overexpression attenuates myocardial reperfusion injury. Am J Physiol Heart Circ Physiol. 2004. 286:H276–H282.
Article95. Jones SP, Greer JJ, van Haperen R, Duncker DJ, de Crom R, Lefer DJ. Endothelial nitric oxide synthase overexpression attenuates congestive heart failure in mice. Proc Natl Acad Sci USA. 2003. 100:4891–4896.
Article96. Jones SP, Girod WG, Palazzo AJ, Granger DN, Grisham MB, Jourd'Heuil D, et al. Myocardial ischemia-reperfusion injury is exacerbated in absence of endothelial cell nitric oxide synthase. Am J Physiol. 1999. 276:H1567–H1573.97. Sharp BR, Jones SP, Rimmer DM, Lefer DJ. Differential response to myocardial reperfusion injury in eNOS-deficient mice. Am J Physiol Heart Circ Physiol. 2002. 282:H2422–H2426.98. Jones SP, Bolli R. The ubiquitous role of nitric oxide in cardioprotection. J Mol Cell Cardiol. 2006. 40:16–23.
Article99. Tsang A, Hausenloy DJ, Mocanu MM, Yellon DM. Postconditioning: a form of "modified reperfusion" protects the myocardium by activating the phosphatidylinositol 3-kinase-Akt pathway. Circ Res. 2004. 95:230–232.
Article100. Cross HR, Murphy E, Steenbergen C. Ca(2+) loading and adrenergic stimulation reveal male/female differences in susceptibility to ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol. 2002. 283:H481–H489.101. Ghafourifar P, Sen CK. Mitochondrial nitric oxide synthase. Front Biosci. 2007. 12:1072–1078.
Article102. Kato K, Giulivi C. Critical overview of mitochondrial nitric-oxide synthase. Front Biosci. 2006. 11:2725–2738.
Article103. Valdez LB, Boveris A. Mitochondrial nitric oxide synthase, a voltage-dependent enzyme, is responsible for nitric oxide diffusion to cytosol. Front Biosci. 2007. 12:1210–1219.
Article104. Brookes PS. Mitochondrial nitric oxide synthase. Mitochondrion. 2004. 3:187–204.
Article105. Lacza Z, Pankotai E, Csordas A, Gero D, Kiss L, Horvath EM, et al. Mitochondrial NO and reactive nitrogen species production: does mtNOS exist? Nitric Oxide. 2006. 14:162–168.
Article106. Guo FQ, Crawford NM. Arabidopsis nitric oxide synthase1 is targeted to mitochondria and protects against oxidative damage and dark-induced senescence. Plant Cell. 2005. 17:3436–3450.
Article107. Zemojtel T, Wade RC, Dandekar T. In search of the prototype of nitric oxide synthase. FEBS Lett. 2003. 554:1–5.
Article108. Crawford NM, Galli M, Tischner R, Heimer YM, Okamoto M, Mack A. Response to Zemojtel et al: plant nitric oxide synthase: back to square one. Trends Plant Sci. 2006. 11:526–527.
Article109. Guo FQ. Response to Zemojtel et al: plant nitric oxide synthase: AtNOS1 is just the beginning. Trends Plant Sci. 2006. 11:527–528.
Article110. Zemojtel T, Frohlich A, Palmieri MC, Kolanczyk M, Mikula I, Wyrwicz LS, et al. Plant nitric oxide synthase: a never-ending story? Trends Plant Sci. 2006. 11:524–525.
Article111. Feng Q, Lu X, Jones DL, Shen J, Arnold JM. Increased inducible nitric oxide synthase expression contributes to myocardial dysfunction and higher mortality after myocardial infarction in mice. Circulation. 2001. 104:700–704.
Article112. Ramachandran A, Levonen AL, Brookes PS, Ceaser E, Shiva S, Barone MC, et al. Mitochondria, nitric oxide, and cardiovascular dysfunction. Free Radic Biol Med. 2002. 33:1465–1474.
Article113. Trochu JN, Bouhour JB, Kaley G, Hintze TH. Role of endothelium-derived nitric oxide in the regulation of cardiac oxygen metabolism: implications in health and disease. Circ Res. 2000. 87:1108–1117.
Article114. Brookes PS, Levonen AL, Shiva S, Sarti P, Darley-Usmar VM. Mitochondria: regulators of signal transduction by reactive oxygen and nitrogen species. Free Radic Biol Med. 2002. 33:755–764.
Article115. Granger DL, Taintor RR, Cook JL, Hibbs JB Jr. Injury of neoplastic cells by murine macrophages leads to inhibition of mitochondrial respiration. J Clin Invest. 1980. 65:357–370.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Protective mechanism of nitric oxide and mucus against ischemia/reperfusion-induced gastric mucosal injury
- Changes of Nitric Oxide Currents and Morphologies in Rat Gastrocnemius Muscle during Ischemia-Reperfusion
- Changes of Hemodynamics and Nitric Oxide during Liver Ischemia/Reperfusion of Pig
- Neuroprotection of Dexmedetomidine against Cerebral Ischemia-Reperfusion Injury in Rats: Involved in Inhibition of NF-κB and Inflammation Response
- Expression Pattern of Nitric Oxide Synthases in Rat Skeletal Muscle after Cyclic Episodes of Short Ischemia and Reperfusion