Yonsei Med J.
2014 Nov;55(6):1648-1655. 10.3349/ymj.2014.55.6.1648.
Increased Expression of Cathelicidin by Direct Activation of Protease-Activated Receptor 2: Possible Implications on the Pathogenesis of Rosacea
- Affiliations
-
- 1Department of Dermatology, Cutaneous Biology Research Institute, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea. oddung93@yuhs.ac
- 2Department of Pathology, Brain Korea 21 Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2070215
- DOI: http://doi.org/10.3349/ymj.2014.55.6.1648
Abstract
- PURPOSE
Recent findings of increased cathelicidin protein and its proteolytic fragments in rosacea suggest a pathogenic role for cathelicidin in this disease. The relationship between cathelicidin and protease-activated receptor 2 (PAR-2) is therefore of interest, as PAR-2, expressed principally in keratinocytes, regulates pro-inflammatory cytokine expression in the skin. The purpose of this study was to determine the relationship between expression of PAR-2 and cathelicidin in rosacea and to test the effect of direct PAR-2 activation on cathelicidin expression in keratinocytes.
MATERIALS AND METHODS
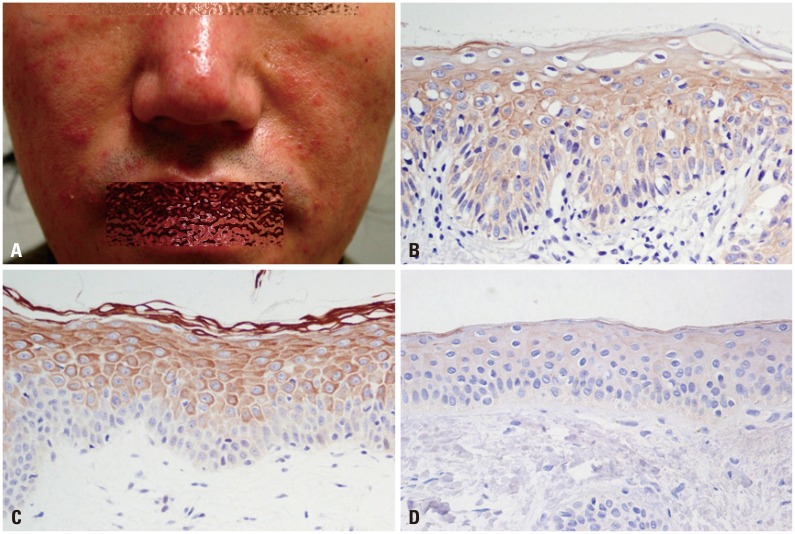
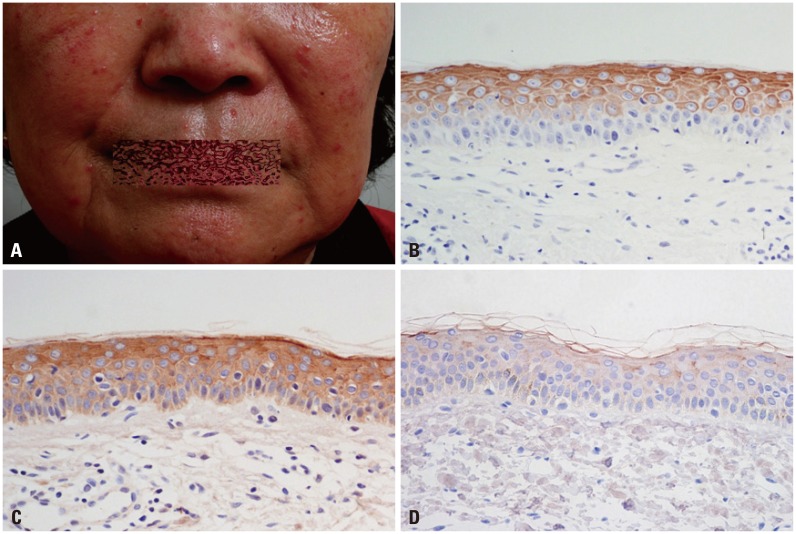
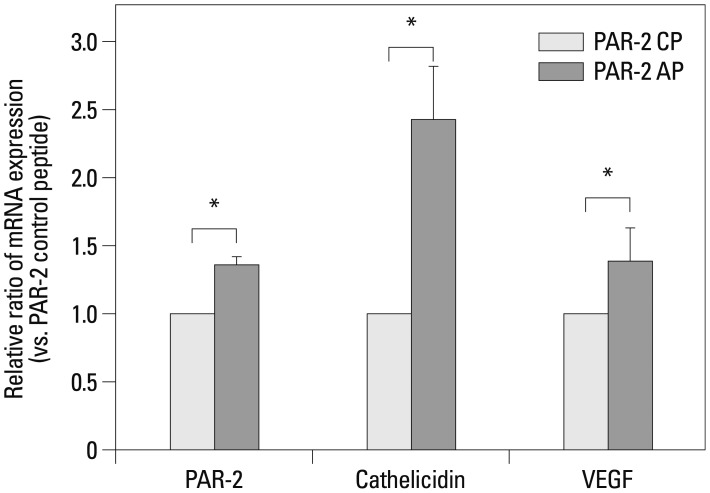
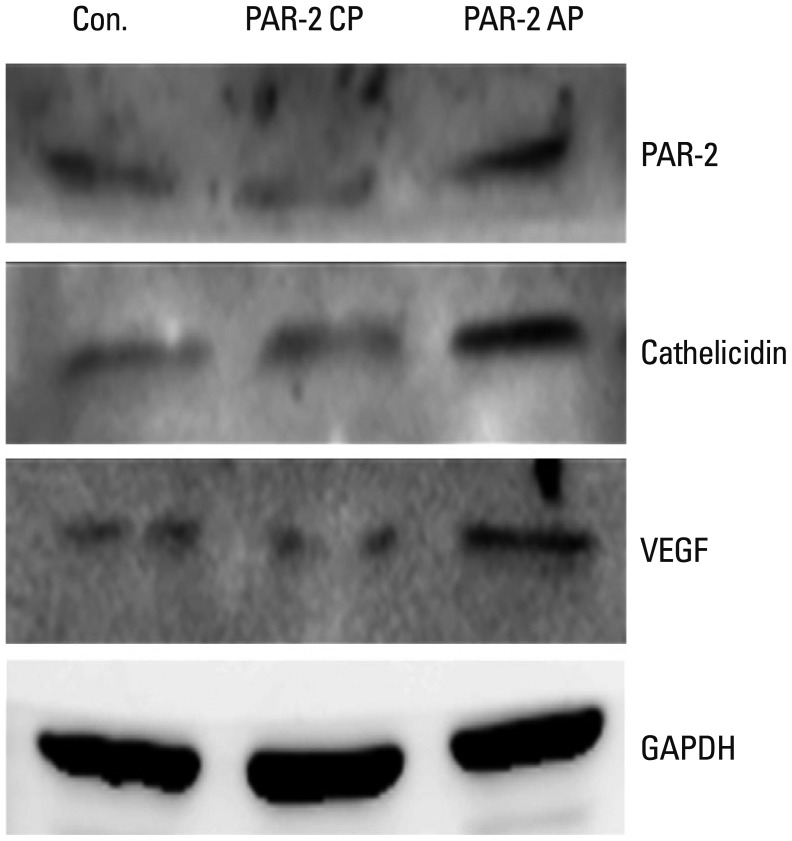
Samples from 40 patients with clinicopathologic diagnosis of rosacea and facial skin tissue samples from 20 patients with no specific findings or milium without inflammation were retrieved. Intensities of immunohistochemical staining for PAR-2 and cathelicidin were compared between normal and rosacea-affected skin tissues. Additionally, correlations between PAR-2 and cathelicidin staining intensities within rosacea patients were analyzed. In cultured keratinocytes, changes in PAR-2, cathelicidin, and vascular endothelial growth factor (VEGF) mRNA and protein were analyzed after treatment with PAR-2 activating peptide (AP).
RESULTS
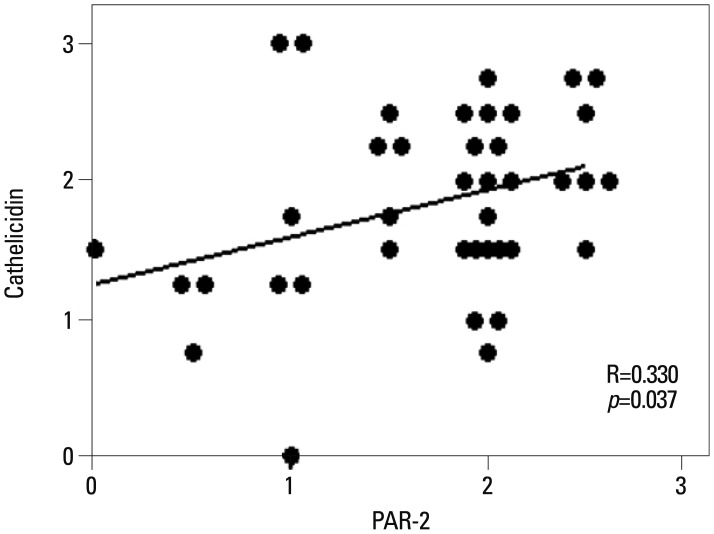
Cathelicidin expression was significantly higher in rosacea skin tissues than in normal tissues (p<0.001), while PAR-2 expression was not significantly higher in rosacea tissues than in normal skin tissues. A positive correlation between PAR-2 and cathelicidin within rosacea samples was observed (R=0.330, p=0.037). After treatment of PAR-2 AP, both mRNA and protein levels for PAR-2, cathelicidin, and VEGF significantly increased in cultured keratinocytes, compared with PAR-2 control peptide treatment.
CONCLUSION
PAR-2 may participate in the pathogenesis of rosacea through activation of cathelicidin LL-37, a mediator of innate immune responses in the skin.
Keyword
MeSH Terms
-
Adult
Aged
Antimicrobial Cationic Peptides/*metabolism
Cytokines/metabolism
Female
Humans
Immunity, Innate
Inflammation/metabolism
Keratinocytes/*metabolism
Male
Middle Aged
Receptor, PAR-2/*metabolism
Rosacea/*pathology
Skin/pathology
Vascular Endothelial Growth Factor A/*metabolism
Antimicrobial Cationic Peptides
Cytokines
Receptor, PAR-2
Vascular Endothelial Growth Factor A
Figure
Reference
-
1. Korting HC, Schöllmann C. Current topical and systemic approaches to treatment of rosacea. J Eur Acad Dermatol Venereol. 2009; 23:876–882. PMID: 19508315.
Article2. Wilkin J, Dahl M, Detmar M, Drake L, Feinstein A, Odom R, et al. Standard classification of rosacea: Report of the National Rosacea Society Expert Committee on the Classification and Staging of Rosacea. J Am Acad Dermatol. 2002; 46:584–587. PMID: 11907512.
Article3. Yamasaki K, Gallo RL. Rosacea as a disease of cathelicidins and skin innate immunity. J Investig Dermatol Symp Proc. 2011; 15:12–15.
Article4. Nizet V, Ohtake T, Lauth X, Trowbridge J, Rudisill J, Dorschner RA, et al. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature. 2001; 414:454–457. PMID: 11719807.
Article5. Zanetti M. The role of cathelicidins in the innate host defenses of mammals. Curr Issues Mol Biol. 2005; 7:179–196. PMID: 16053249.6. Koczulla R, von Degenfeld G, Kupatt C, Krötz F, Zahler S, Gloe T, et al. An angiogenic role for the human peptide antibiotic LL-37/hCAP-18. J Clin Invest. 2003; 111:1665–1672. PMID: 12782669.
Article7. Braff MH, Hawkins MA, Di Nardo A, Lopez-Garcia B, Howell MD, Wong C, et al. Structure-function relationships among human cathelicidin peptides: dissociation of antimicrobial properties from host immunostimulatory activities. J Immunol. 2005; 174:4271–4278. PMID: 15778390.
Article8. Yamasaki K, Di Nardo A, Bardan A, Murakami M, Ohtake T, Coda A, et al. Increased serine protease activity and cathelicidin promotes skin inflammation in rosacea. Nat Med. 2007; 13:975–980. PMID: 17676051.
Article9. Yamasaki K, Schauber J, Coda A, Lin H, Dorschner RA, Schechter NM, et al. Kallikrein-mediated proteolysis regulates the antimicrobial effects of cathelicidins in skin. FASEB J. 2006; 20:2068–2080. PMID: 17012259.
Article10. Detmar M, Brown LF, Claffey KP, Yeo KT, Kocher O, Jackman RW, et al. Overexpression of vascular permeability factor/vascular endothelial growth factor and its receptors in psoriasis. J Exp Med. 1994; 180:1141–1146. PMID: 8064230.
Article11. Hansen KK, Oikonomopoulou K, Li Y, Hollenberg MD. Proteinases, proteinase-activated receptors (PARs) and the pathophysiology of cancer and diseases of the cardiovascular, musculoskeletal, nervous and gastrointestinal systems. Naunyn Schmiedebergs Arch Pharmacol. 2008; 377:377–392. PMID: 17952408.
Article12. Coughlin SR. Protease-activated receptors start a family. Proc Natl Acad Sci U S A. 1994; 91:9200–9202. PMID: 7937741.
Article13. Rattenholl A, Steinhoff M. Proteinase-activated receptor-2 in the skin: receptor expression, activation and function during health and disease. Drug News Perspect. 2008; 21:369–381. PMID: 19259550.
Article14. Stefansson K, Brattsand M, Roosterman D, Kempkes C, Bocheva G, Steinhoff M, et al. Activation of proteinase-activated receptor-2 by human kallikrein-related peptidases. J Invest Dermatol. 2008; 128:18–25. PMID: 17625593.
Article15. Chung WO, Hansen SR, Rao D, Dale BA. Protease-activated receptor signaling increases epithelial antimicrobial peptide expression. J Immunol. 2004; 173:5165–5170. PMID: 15470061.
Article16. Lee SE, Kim JM, Jeong SK, Jeon JE, Yoon HJ, Jeong MK, et al. Protease-activated receptor-2 mediates the expression of inflammatory cytokines, antimicrobial peptides, and matrix metalloproteinases in keratinocytes in response to Propionibacterium acnes. Arch Dermatol Res. 2010; 302:745–756. PMID: 20697725.
Article17. Elewski BE, Fleischer AB Jr, Pariser DM. A comparison of 15% azelaic acid gel and 0.75% metronidazole gel in the topical treatment of papulopustular rosacea: results of a randomized trial. Arch Dermatol. 2003; 139:1444–1450. PMID: 14623704.
Article18. Crawford GH, Pelle MT, James WD. Rosacea: I. Etiology, pathogenesis, and subtype classification. J Am Acad Dermatol. 2004; 51:327–341. PMID: 15337973.
Article19. Neumann E, Frithz A. Capillaropathy and capillaroneogenesis in the pathogenesis of rosacea. Int J Dermatol. 1998; 37:263–266. PMID: 9585896.20. Lacey N, Delaney S, Kavanagh K, Powell FC. Mite-related bacterial antigens stimulate inflammatory cells in rosacea. Br J Dermatol. 2007; 157:474–481. PMID: 17596156.
Article21. McAleer MA, Fitzpatrick P, Powell FC. Papulopustular rosacea: prevalence and relationship to photodamage. J Am Acad Dermatol. 2010; 63:33–39. PMID: 20462665.
Article22. Selsted ME, Ouellette AJ. Mammalian defensins in the antimicrobial immune response. Nat Immunol. 2005; 6:551–557. PMID: 15908936.
Article23. Zaiou M, Gallo RL. Cathelicidins, essential gene-encoded mammalian antibiotics. J Mol Med (Berl). 2002; 80:549–561. PMID: 12226737.
Article24. Schröder JM, Harder J. Human beta-defensin-2. Int J Biochem Cell Biol. 1999; 31:645–651. PMID: 10404637.
Article25. Murakami M, Ohtake T, Dorschner RA, Schittek B, Garbe C, Gallo RL. Cathelicidin anti-microbial peptide expression in sweat, an innate defense system for the skin. J Invest Dermatol. 2002; 119:1090–1095. PMID: 12445197.
Article26. Wu WK, Wong CC, Li ZJ, Zhang L, Ren SX, Cho CH. Cathelicidins in inflammation and tissue repair: potential therapeutic applications for gastrointestinal disorders. Acta Pharmacol Sin. 2010; 31:1118–1122. PMID: 20676121.
Article27. Davidson DJ, Currie AJ, Reid GS, Bowdish DM, MacDonald KL, Ma RC, et al. The cationic antimicrobial peptide LL-37 modulates dendritic cell differentiation and dendritic cell-induced T cell polarization. J Immunol. 2004; 172:1146–1156. PMID: 14707090.
Article28. Zaiou M, Nizet V, Gallo RL. Antimicrobial and protease inhibitory functions of the human cathelicidin (hCAP18/LL-37) prosequence. J Invest Dermatol. 2003; 120:810–816. PMID: 12713586.
Article29. Steinmann J, Halldórsson S, Agerberth B, Gudmundsson GH. Phenylbutyrate induces antimicrobial peptide expression. Antimicrob Agents Chemother. 2009; 53:5127–5133. PMID: 19770273.
Article30. Gombart AF, O'Kelly J, Saito T, Koeffler HP. Regulation of the CAMP gene by 1,25(OH)2D3 in various tissues. J Steroid Biochem Mol Biol. 2007; 103:552–557. PMID: 17368184.
Article31. Yim S, Dhawan P, Ragunath C, Christakos S, Diamond G. Induction of cathelicidin in normal and CF bronchial epithelial cells by 1,25-dihydroxyvitamin D(3). J Cyst Fibros. 2007; 6:403–410. PMID: 17467345.
Article32. White JH. Vitamin D as an inducer of cathelicidin antimicrobial peptide expression: past, present and future. J Steroid Biochem Mol Biol. 2010; 121:234–238. PMID: 20302931.
Article33. Kawabata A. PAR-2: structure, function and relevance to human diseases of the gastric mucosa. Expert Rev Mol Med. 2002; 4:1–17. PMID: 14585156.
Article34. Sondell B, Thornell LE, Stigbrand T, Egelrud T. Immunolocalization of stratum corneum chymotryptic enzyme in human skin and oral epithelium with monoclonal antibodies: evidence of a proteinase specifically expressed in keratinizing squamous epithelia. J Histochem Cytochem. 1994; 42:459–465. PMID: 7510318.
Article35. Hachem JP, Man MQ, Crumrine D, Uchida Y, Brown BE, Rogiers V, et al. Sustained serine proteases activity by prolonged increase in pH leads to degradation of lipid processing enzymes and profound alterations of barrier function and stratum corneum integrity. J Invest Dermatol. 2005; 125:510–520. PMID: 16117792.
Article36. Buddenkotte J, Stroh C, Engels IH, Moormann C, Shpacovitch VM, Seeliger S, et al. Agonists of proteinase-activated receptor-2 stimulate upregulation of intercellular cell adhesion molecule-1 in primary human keratinocytes via activation of NF-kappa B. J Invest Dermatol. 2005; 124:38–45. PMID: 15654951.
Article37. Wakita H, Furukawa F, Takigawa M. Thrombin and trypsin induce granulocyte-macrophage colony-stimulating factor and interleukin-6 gene expression in cultured normal human keratinocytes. Proc Assoc Am Physicians. 1997; 109:190–207. PMID: 9069588.38. Hou L, Kapas S, Cruchley AT, Macey MG, Harriott P, Chinni C, et al. Immunolocalization of protease-activated receptor-2 in skin: receptor activation stimulates interleukin-8 secretion by keratinocytes in vitro. Immunology. 1998; 94:356–362. PMID: 9767417.39. Kawagoe J, Takizawa T, Matsumoto J, Tamiya M, Meek SE, Smith AJ, et al. Effect of protease-activated receptor-2 deficiency on allergic dermatitis in the mouse ear. Jpn J Pharmacol. 2002; 88:77–84. PMID: 11859856.
Article40. Gruber BL, Marchese MJ, Santiago-Schwarz F, Martin CA, Zhang J, Kew RR. Protease-activated receptor-2 (PAR-2) expression in human fibroblasts is regulated by growth factors and extracellular matrix. J Invest Dermatol. 2004; 123:832–839. PMID: 15482468.
Article41. Xiang Y, Masuko-Hongo K, Sekine T, Nakamura H, Yudoh K, Nishioka K, et al. Expression of proteinase-activated receptors (PAR)-2 in articular chondrocytes is modulated by IL-1beta, TNF-alpha and TGF-beta. Osteoarthritis Cartilage. 2006; 14:1163–1173. PMID: 16757188.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Study on Vitamin D and Cathelicidin Status in Patients with Rosacea: Serum Level and Tissue Expression
- Cathelicidin LL-37: An Antimicrobial Peptide with a Role in Inflammatory Skin Disease
- The Effect of Capsaicin on Neuroinflammatory Mediators of Rosacea
- Protease and Protease-Activated Receptor-2 Signaling in the Pathogenesis of Atopic Dermatitis
- Hyaluronan Oligosaccharides Improve Rosacea-Like Phenotype through Anti-Inflammatory and Epidermal Barrier-Improving Effects