J Korean Med Sci.
2014 Feb;29(2):183-189. 10.3346/jkms.2014.29.2.183.
In Vitro Bacteriostatic Effects of Rifampin on Orientia tsutsugamushi
- Affiliations
-
- 1Department of Internal Medicine, Inha University School of Medicine, Incheon, Korea. mhchungid@daum.net
- 2Translation Research Center, Inha University School of Medicine, Incheon, Korea.
- 3Department of Microbiology, Inha University School of Medicine, Incheon, Korea.
- KMID: 1789973
- DOI: http://doi.org/10.3346/jkms.2014.29.2.183
Abstract
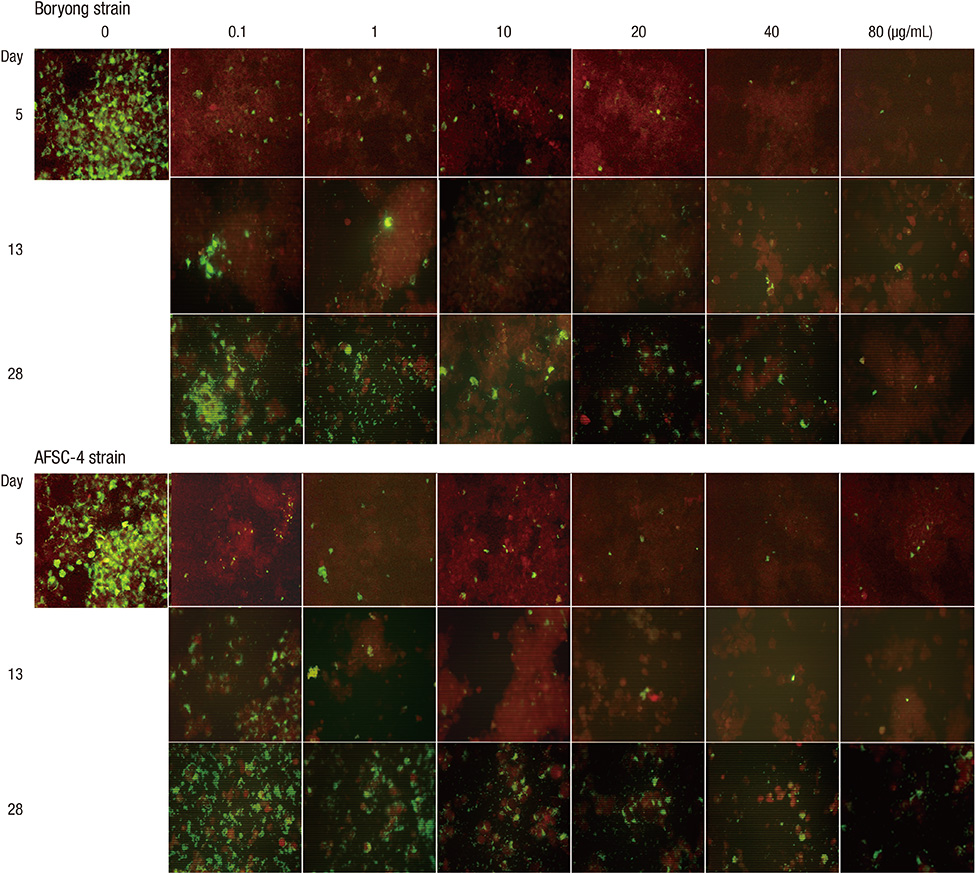
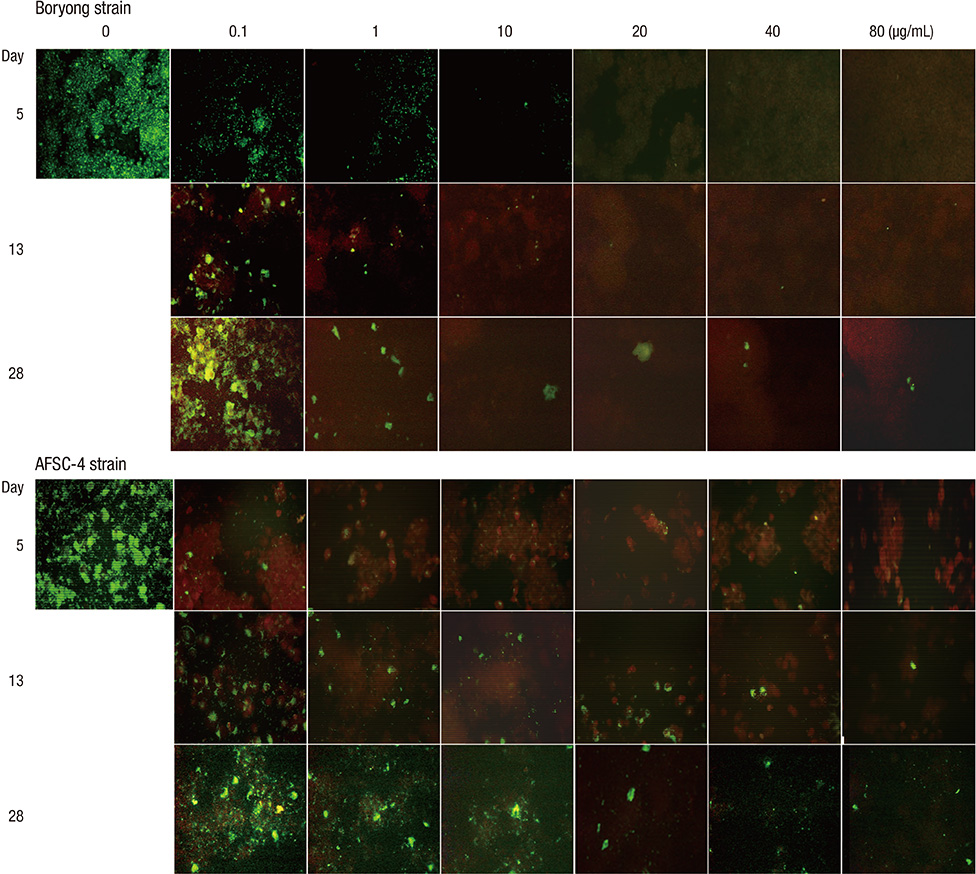
- We performed an in vitro cell culture experiment to ascertain whether rifampin exhibits bactericidal effects against Orientia tsutsugamushi, the causative agent of scrub typhus. ECV304 cells were infected with the Boryong or AFSC-4 strain of O. tsutsugamushi and then, the cultures were maintained in media with increasing concentrations of rifampin, azithromycin, doxycycline, or chloramphenicol for 4 days. On day 5, the media were replaced with fresh antibiotic-free medium and the cultures were maintained until day 28. On days 5, 13, and 28, immunofluorescence (IF) staining of O. tsutsugamushi was performed. IF staining on days 13 and 28 revealed increasing numbers of IF-positive foci in all cultures, even in cultures initially exposed to the highest concentration of rifampin (80 microg/mL), azithromycin (80 microg/mL), doxycycline (20 microg/mL), or chloramphenicol (100 microg/mL). The present study reveals that rifampin has no bactericidal effect against O. tsutsugamushi as observed for azithromycin, doxycycline, and chloramphenicol. A subpopulation of the bacteria that are not killed by high concentrations of the antibiotics may explain the persistence of O. tsutsugamushi in humans even after complete recovery from scrub typhus with antibiotic therapy.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
In vitro Synergism between Chloroquine and Antibiotics against Orientia tsutsugamushi
Dongwook Son, Moon-Hyun Chung
Infect Chemother. 2014;46(3):182-188. doi: 10.3947/ic.2014.46.3.182.In vitro Antagonism between Cefotaxime and Anti-Rickettsial Antibiotics against Orientia tsutsugamushi
Oh Hyun Lee, Ji Hyeon Baek, Jin-Soo Lee, Moon-Hyun Chung, Sun Myoung Lee, Jae-Seung Kang
Infect Chemother. 2014;46(3):189-193. doi: 10.3947/ic.2014.46.3.189.
Reference
-
1. Smadel JE, Bailey CA, Diercks FH. Chloramphenicol in the chemoprophylaxis of scrub typhus: IV. relapses of scrub typhus in treated volunteers and their prevention. Am J Epidemiol. 1950; 51:229–241.2. Sheehy TW, Hazlett D, Turk RE. Scrub typhus: a comparison of chloramphenicol and tetracycline in its treatment. Arch Intern Med. 1973; 132:77–80.3. Smadel JE, Jackson EB, Ley HL Jr. Terramycin as a rickettsiostatic agent and its usefulness in patients with scrub typhus. Ann N Y Acad Sci. 1950; 53:375–384.4. Chung MH, Lee JS, Baek JH, Kim M, Kang JS. Persistence of Orientia tsutsugamushi in humans. J Korean Med Sci. 2012; 27:231–235.5. Smadel JE, Jackson EB, Cruise AB. Chloromycetin in experimental rickettsial infections. J Immunol. 1949; 62:49–65.6. Finberg RW, Moellering RC, Tally FP, Craig WA, Pankey GA, Dellinger EP, West MA, Joshi M, Linden PK, Rolston KV, et al. The importance of bactericidal drugs: future directions in infectious disease. Clin Infect Dis. 2004; 39:1314–1320.7. Watt G, Kantipong P, Jongsakul K, Watcharapichat P, Phulsuksombati D, Strickman D. Doxycycline and rifampicin for mild scrub-typhus infections in northern Thailand: a randomised trial. Lancet. 2000; 356:1057–1061.8. Watt G, Chouriyagune C, Ruangweerayud R, Watcharapichat P, Phulsuksombati D, Jongsakul K, Teja-Isavadharm P, Bhodhidatta D, Corcoran KD, Dasch GA, et al. Scrub typhus infections poorly responsive to antibiotics in northern Thailand. Lancet. 1996; 348:86–89.9. Miyamura S, Sato N, Tamura A. In vitro susceptibility of recent clinical isolates of Rickettsia tsutsugamushi to chemotherapeutic agents. Kansenshogaku Zasshi. 1985; 59:486–488.10. Miyamura S, Ohta T, Tamura A. Comparison of in vitro susceptibilities of Rickettsia prowazekii, R. rickettsii, R. sibirica and R. tsutsugamushi to antimicrobial agents. Nihon Saikingaku Zasshi. 1989; 44:717–721.11. Urakami H, Tamura A, Miyamura S, Yamamoto S, Kawabata N. Susceptibilities of recent clinical isolates of Rickettsia tsutsugamushi to chemotherapeutic agents in vitro. Kansenshogaku Zasshi. 1988; 62:931–937.12. Kim ES, Kim MK, Lee HM, Chung MH, Lee JS, Park JE, Kang JS. In vitro antibiotic susceptibility of Orientia tsutsugamushi strain Boryong measured by flow cytometry. Infect Chemother. 2008; 40:212–217.13. Strickman D, Sheer T, Salata K, Hershey J, Dasch G, Kelly D, Kuschner R. In vitro effectiveness of azithromycin against doxycycline-resistant and -susceptible strains of Rickettsia tsutsugamushi, etiologic agent of scrub typhus. Antimicrob Agents Chemother. 1995; 39:2406–2410.14. Brown J, Reading SJ, Jones S, Fitchett CJ, Howl J, Martin A, Longland CL, Michelangeli F, Dubrova YE, Brown CA. Critical evaluation of ECV304 as a human endothelial cell model defined by genetic analysis and functional responses: a comparison with the human bladder cancer derived epithelial cell line T24/83. Lab Invest. 2000; 80:37–45.15. Kim MJ, Kim MK, Kang JS. Improved antibiotic susceptibility test of Orientia tsutsugamushi by flow cytometry using monoclonal antibody. J Korean Med Sci. 2007; 22:1–6.16. Amsden GW. Tables of antimicrobial agent pharmacology. In : Mandell GL, Bennett JE, Dolin R, editors. Principles and practice of infectious diseases. 6th ed. Philadelphia: Elsevier Churchill Livingstone;2005. p. 635–700.17. Darouiche RO, Hamill RJ. Antibiotic penetration of and bactericidal activity within endothelial cells. Antimicrob Agents Chemother. 1994; 38:1059–1064.18. Smadel JE. Influence of antibiotics on immunologic responses in scrub typhus. Am J Med. 1954; 17:246–258.19. Kim ES, Kim MK, Lee HM, Kil SH, Chung MH, Lee JS, Kang JS. Doxycycline resistance in Orientia tsutsugamushi isolated from Korean patients. Infect Chemother. 2008; 40:259–265.20. Tantibhedhyangkul W, Angelakis E, Tongyoo N, Newton PN, Moore CE, Phetsouvanh R, Raoult D, Rolain JM. Intrinsic fluoroquinolone resistance in Orientia tsutsugamushi. Int J Antimicrob Agents. 2010; 35:338–341.21. Jang HC, Choi SM, Jang MO, Ahn JH, Kim UJ, Kang SJ, Shin JH, Choy HE, Jung SI, Park KH. Inappropriateness of quinolone in scrub typhus treatment due to gyrA mutation in Orientia tsutsugamushi Boryong strain. J Korean Med Sci. 2013; 28:667–671.22. Levin BR, Rozen DE. Non-inherited antibiotic resistance. Nat Rev Microbiol. 2006; 4:556–562.23. Suchland RJ, Geisler WM, Stamm WE. Methodologies and cell lines used for antimicrobial susceptibility testing of Chlamydia spp. Antimicrob Agents Chemother. 2003; 47:636–642.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- In vitro Synergism between Chloroquine and Antibiotics against Orientia tsutsugamushi
- In vitro Antagonism between Cefotaxime and Anti-Rickettsial Antibiotics against Orientia tsutsugamushi
- In vitro Efficacy of Antibiotic Combinations against Orientia tsutsugamushi
- Leukocytoclastic Vasculitis Associated with Orientia tsutsugamushi Infection: A Report of Two Cases
- Genetic Heterogeneity in 56 kDa gene of Orientia tsutsugamushi Genotype Karp