Korean J Lab Med.
2010 Aug;30(4):339-344. 10.3343/kjlm.2010.30.4.339.
Flow-Assisted Differential Diagnosis of Hemolytic Anemia with Spherocytosis: A Case Report
- Affiliations
-
- 1Department of Laboratory Medicine, Kyungpook National University Hospital, Daegu, Korea. wondi@knu.ac.kr
- KMID: 1781638
- DOI: http://doi.org/10.3343/kjlm.2010.30.4.339
Abstract
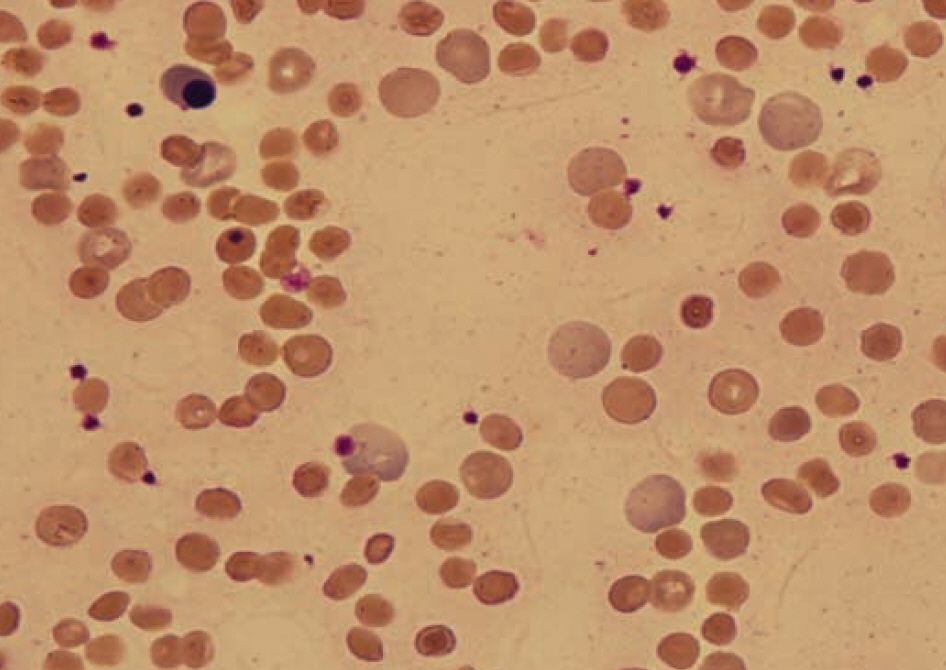
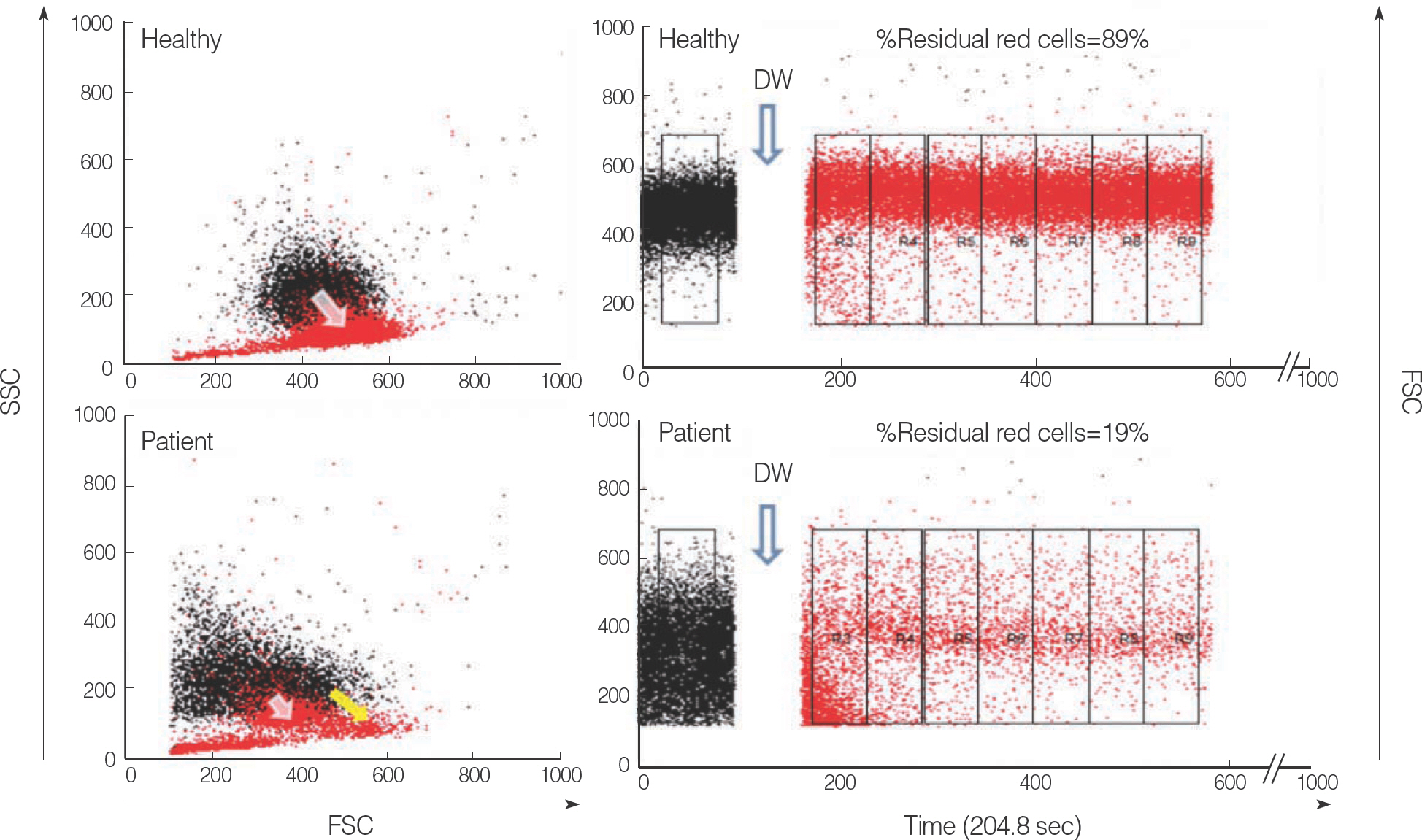
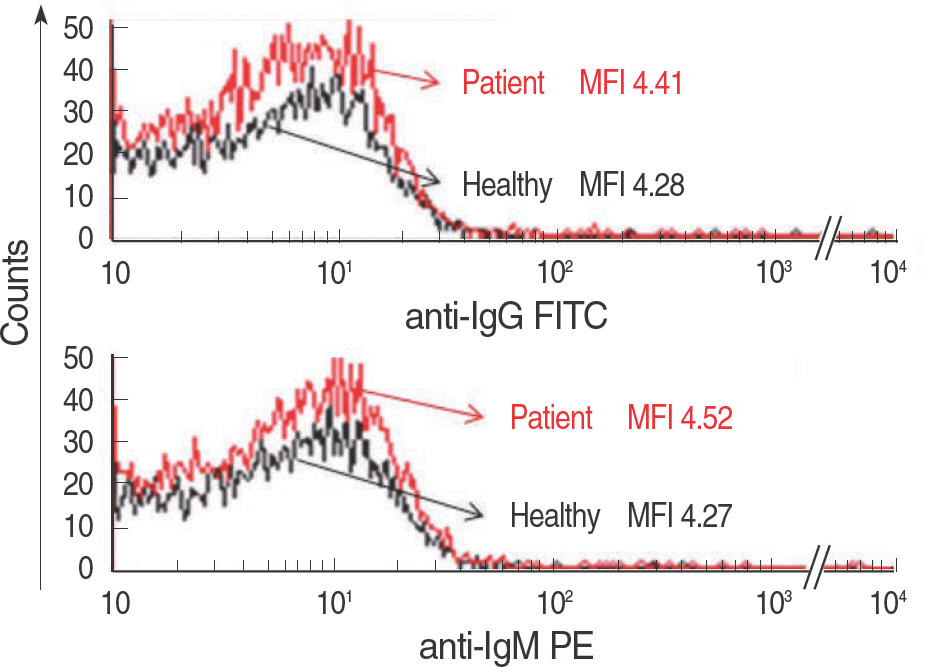
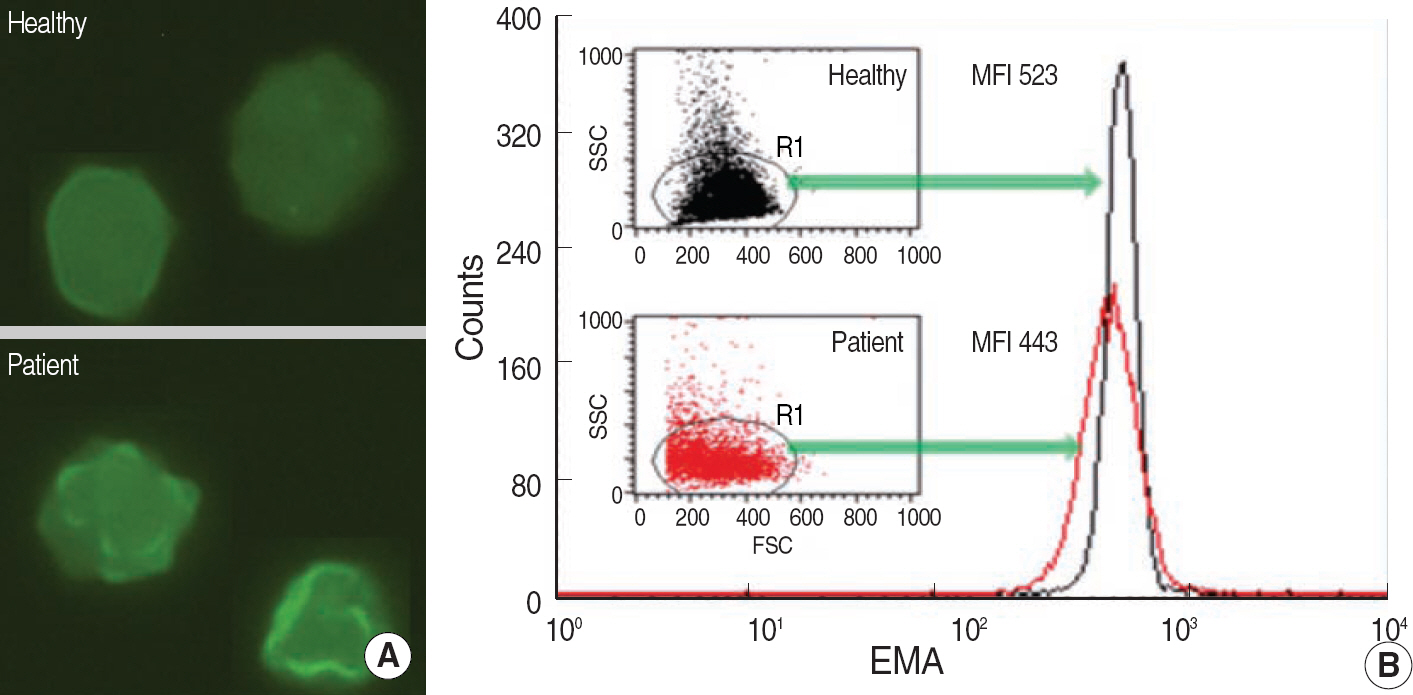
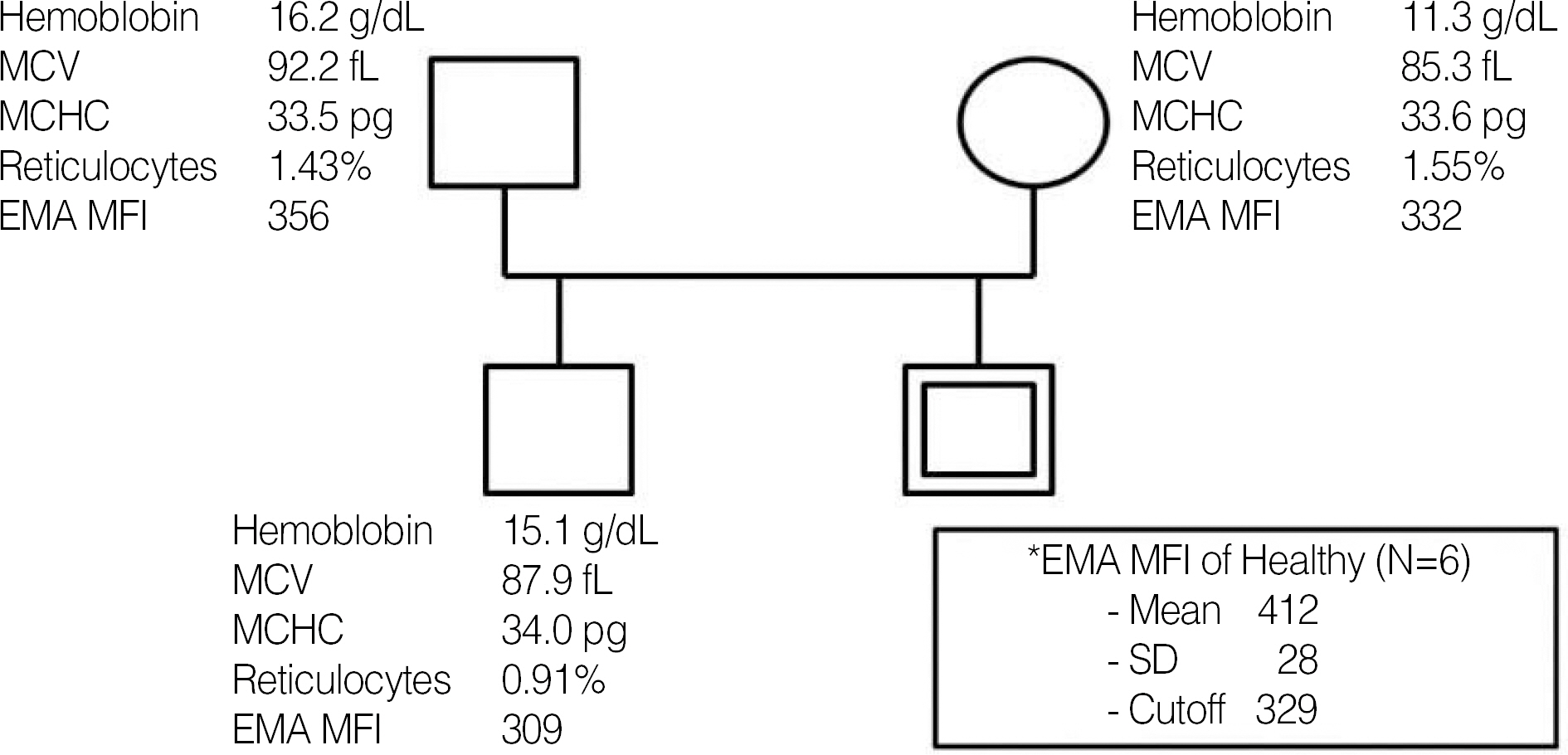
- In patients with hemolytic anemia associated with spherocytosis, differential diagnosis has to be made whether the hemolysis is immune-mediated or of non-immune origin. We report a case of hereditary spherocytosis in a 12-yr-old male child, in whom flow-assisted diagnosis was made. In this case, diagnosis was not determined because routine laboratory workups for hereditary spherocytosis yielded discrepant
RESULTS
positive osmotic fragility test, positive direct antiglobulin test, and normal result in the red cell membrane protein sodium dodecyl succinimide polyacrylamide gel electrophoresis. However, all flow cytometry-based tests, such as osmotic fragility, direct antiglobulin, and eosin 5-maleimide binding test, yielded results compatible with hereditary spherocytosis. Additionally, in family study, the results of eosin 5-maleimide binding test suggested his disease being hereditary. In cases with diagnostic difficulties, flow cytometry may be used as an alternative tool, which can provide additional information in the differential diagnosis of hemolytic anemia with spherocytosis.
MeSH Terms
Figure
Cited by 1 articles
-
Hereditary hemolytic anemia in Korea from 2007 to 2011: A study by the Korean Hereditary Hemolytic Anemia Working Party of the Korean Society of Hematology
Eun Sil Park, Hye Lim Jung, Hee-Jin Kim, Sung Sup Park, Soon Hwan Bae, Hee Young Shin, Sang Hoon Song, Kyung-Nam Koh, Chuhl Joo Lyu, Young Tak Lim, Dong Kyun Han, Jeong Ok Hah
Blood Res. 2013;48(3):211-216. doi: 10.5045/br.2013.48.3.211.
Reference
-
1.Gallagher PG., Forget BG., Lux SE. Disorders of the erythrocyte membrane. Nathan DG, Orkin SH, editors. Nathan and Oski's hematology of infancy and childhood. 5th ed.Philadelphia: WB Saunders;1998. p. 544–664.2.Bolton-Maggs PH, Stevens RF, Dodd NJ, Lamont G, Tittensor P, King MJ; General Haematology Task Force of the British Committee for Standards in Haematology. Guidelines for the diagnosis and management of hereditary spherocytosis. Br J Haematol. 2004. 126:455–74.3.Garratty G. The significance of IgG on the red cell surface. Transfus Med Rev. 1987. 1:47–57.
Article4.Lee YK., Cho HI., Park SS., Ra E., Chang YH., Hur M, et al. SDS-PAGE analysis of red cell membrane proteins in hereditary hemolytic anemia. Korean J Hematol. 1999. 34:559–67. (이영경, 조한익, 박성섭, 나은경, 장윤환, 허미나 등. 유전성용혈성빈혈에서 SDS-PAGE를 이용한 적혈구막 단백성분분석. 대한혈액학회지 1999;34:559-67.).5.King MJ., Telfer P., MacKinnon H., Langabeer L., McMahon C., Darbyshire P, et al. Using the eosin-5-maleimide binding test in the differential diagnosis of hereditary spherocytosis and hereditary pyropoikilocytosis. Cytometry B Clin Cytom. 2008. 74:244–50.
Article6.King MJ., Smythe JS., Mushens R. Eosin-5-maleimide binding to band 3 and Rh-related proteins forms the basis of a screening test for hereditary spherocytosis. Br J Haematol. 2004. 124:106–13.
Article7.Kedar PS., Colah RB., Kulkarni S., Ghosh K., Mohanty D. Experience with eosin-5′-maleimide as a diagnostic tool for red cell membrane cytoskeleton disorders. Clin Lab Haematol. 2003. 25:373–6.
Article8.King MJ., Behrens J., Rogers C., Flynn C., Greenwood D., Chambers K. Rapid flow cytometric test for the diagnosis of membrane cytoskeleton-associated haemolytic anaemia. Br J Haematol. 2000. 111:924–33.
Article9.Won DI., Suh JS. Flow cytometric detection of erythrocyte osmotic fragility. Cytometry B Clin Cytom. 2009. 76:135–41.
Article10.Won DI., Jung OJ., Lee YS., Kim SG., Suh JS. Flow cytometry antibody screening using pooled red cells. Cytometry B Clin Cytom. 2010. 78:96–104.
Article11.Kerr R., Rawlinson PS., Cachia PG. Direct antiglobulin test negative, non spherocytic autoimmune haemolytic anaemia. Clin Lab Haematol. 2000. 22:365–7.
Article12.Boyd AS. Hereditary spherocytosis. Am Fam Physician. 1989. 39:167–72.13.Ruddy S. Complement. Rose NR, de Macario EC, editors. Manual of clinical laboratory immunology. 4th ed.Washington, DC: American Society for Microbiology;1992. p. 114–23.14.Downes KA., Shulman IA. Pretransfusion testing. Roback JD, Combs MR, editors. Technical manual. 16th ed.Bethesda: American Association of Blood Banks;2008. p. 437–63.15.Leger RM. The positive direct antiglobulin test and immune-mediated hemolysis. Roback JD, Combs MR, editors. Technical manual. 16th ed.Bethesda: American Association of Blood Banks;2008. p. 499–521.16.Mentzer WC., Lubin BH. Red cell membrane abnormalities. Lilleyman JS, Hann IM, editors. Pediatric hematology. 2nd ed.London: Churchill Livingstone;1999. p. 257–83.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Hereditary Spherocytosis with Hemolytic Anemia due to Mycoplasma pneumonia
- Autoimmune Hemolytic Anemia after Aplastic Crisis due to Parvovirus B19 Infection in a Patient with Hereditary Spherocytosis
- Prolonged Extreme Thrombocytosis in a Postsplenectomy Patient with Hereditary Spherocytosis
- Coexistence of Gilbert Syndrome and Hereditary Spherocytosis in a Child Presenting with Extreme Jaundice
- Hereditary Spherocytosis