Lab Med Online.
2012 Jul;2(3):166-169.
Autoimmune Hemolytic Anemia after Aplastic Crisis due to Parvovirus B19 Infection in a Patient with Hereditary Spherocytosis
- Affiliations
-
- 1Department of Laboratory Medicine, Inje University College of Medicine, Busan, Korea. jeong418@medimail.co.kr
Abstract
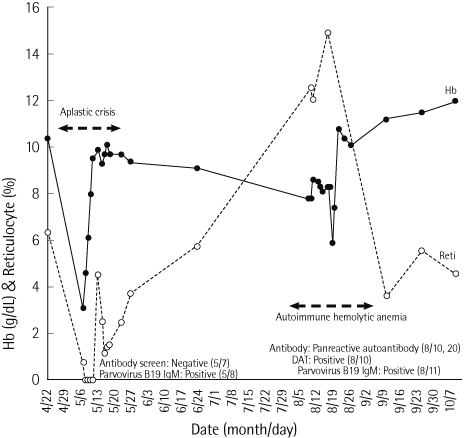
- Hereditary spherocytosis (HS) is a genetic disorder characterized by the production and destruction of spherocytes due to a deficiency of red cell membrane cytoskeletal proteins, resulting in the clinical presentation of chronic hemolytic anemia. This disease can be accompanied by an aplastic crisis due to parvovirus B19 infection. Parvovirus B19 infection causes diseases such as erythema infectiosum and arthritis, and can also trigger various autoimmune diseases, including autoimmune hemolytic anemia (AIHA). Here, we report a rare case of AIHA developing 3 months after an aplastic crisis due to parvovirus B19 infection in an 11-year-old boy with HS and provide the relevant literature review.
MeSH Terms
Figure
Reference
-
1. Perrotta S, Gallagher PG, Mohandas N. Hereditary spherocytosis. Lancet. 2008. 372:1411–1426.
Article2. Lee YK, Cho HI, Park SS, Lee YJ, Ra E, Chang YH, et al. Abnormalities of erythrocyte membrane proteins in Korean patients with hereditary spherocytosis. J Korean Med Sci. 2000. 15:284–288.
Article3. Cho HS, Hah JO, Kang IJ, Kang HJ, Kwak JY, Koo HH, et al. Hereditary hemolyltic anemia in Korea : a retrospective study from 1997 to 2006. Korean J Hematol. 2007. 42:197–205.
Article4. Park YJ, Koh DK, Oh JH. Aplastic crisis secondary to parvovirus B19 infection. J Korean Pediatr Soc. 2003. 46:1139–1142.5. Oh SE, Kim JH, Choi CH, Park KH, Jung JY, Park Y, et al. An adult with aplastic crisis induced by human parvovirus B19 as an initial presentation of hereditary spherocytosis. Korean J Intern Med. 2005. 20:96–99.
Article6. Young N. Hematologic and hematopoietic consequences of B19 parvovirus infection. Semin Hematol. 1988. 25:159–172.7. Jonte F, Ramírez A, Medina J, Garcia Gala J, Rosón C, Corte JR, et al. Hereditary spherocytosis: clinical characteristics and treatment with splenectomy. Sangre (Barc). 1995. 40:45–48.8. Yoshida H, Ishida H, Yoshihara T, Oyamada T, Kuwana M, Imamura T, et al. Complications of Evans' syndrome in an infant with hereditary spherocytosis: a case report. J Hematol Oncol. 2009. 2:40.
Article9. Smith MA, Shah NS, Lobel JS. Parvovirus B19 infection associated with reticulocytopenia and chronic autoimmune hemolytic anemia. Am J Pediatr Hematol Oncol. 1989. 11:167–169.10. Heegaard ED, Myhre J, Hornsleth A, Gundestrup M, Boye H. Parvovirus B19 infections in patients with chronic anemia. Haematologica. 1997. 82:402–405.11. Heegaard ED, Hornsleth A. Parvovirus: the expanding spectrum of disease. Acta Paediatr. 1995. 84:109–117.
Article12. de la Rubia J, Moscardó F, Arriaga F, Monteagudo E, Carreras C, Marty ML. Acute parvovirus B19 infection as a cause of autoimmune hemolytic anemia. Haematologica. 2000. 85:995–997.13. Cossart YE, Field AM, Cant B, Widdows D. Parvovirus-like particles in human sera. Lancet. 1975. 1:72–73.
Article14. Paver WK, Caul EO, Clarke SK. Letter: Parvovirus-like particles in human sera. Lancet. 1975. 1:232.15. Cohen BJ, Mortimer PP, Pereira MS. Diagnostic assays with monoclonal antibodies for the human serum parvovirus-like virus (SPLV). J Hyg (Lond). 1983. 91:113–130.
Article16. Gallinella G, Zuffi E, Gentilomi G, Manaresi E, Venturoli S, Bonvicini F, et al. Relevance of B19 markers in serum samples for a diagnosis of parvovirus B19-correlated diseases. J Med Virol. 2003. 71:135–139.
Article17. Slavov SN, Kashima S, Pinto AC, Covas DT. Human parvovirus B19: general considerations and impact on patients with sickle-cell disease and thalassemia and on blood transfusions. FEMS Immunol Med Microbiol. 2011. 62:247–262.
Article18. Lefrère JJ, Servant-Delmas A, Candotti D, Mariotti M, Thomas I, Brossard Y, et al. Persistent B19 infection in immunocompetent individuals: implications for transfusion safety. Blood. 2005. 106:2890–2895.
Article19. Chehal A, Sharara AI, Haidar HA, Haidar J, Bazarbachi A. Acute viral hepatitis A and parvovirus B19 infections complicated by pure red cell aplasia and autoimmune hemolytic anemia. J Hepatol. 2002. 37:163–165.
Article20. Beauchamp-Nicoud A, Morle L, Lutz HU, Stammler P, Agulles O, Petermann-Khder R, et al. Heavy transfusions and presence of an antiprotein 4.2 antibody in 4. 2(-) hereditary spherocytosis (949delG). Haematologica. 2000. 85:19–24.21. Kerr JR, Boyd N. Autoantibodies following parvovirus B19 infection. J Infect. 1996. 32:41–47.
Article22. Lehmann HW, von Landenberg P, Modrow S. Parvovirus B19 infection and autoimmune disease. Autoimmun Rev. 2003. 2:218–223.
Article23. Scheurlen W, Ramasubbu K, Wachowski O, Hemauer A, Modrow S. Chronic autoimmune thrombopenia/neutropenia in a boy with persistent parvovirus B19 infection. J Clin Virol. 2001. 20:173–178.
Article24. Hod EA, Arinsburg SA, Francis RO, Hendrickson JE, Zimring JC, Spitalnik SL. Use of mouse models to study the mechanisms and consequences of RBC clearance. Vox Sang. 2010. 99:99–111.
Article25. Montes CL, Acosta-Rodríguez EV, Merino MC, Bermejo DA, Gruppi A. Polyclonal B cell activation in infections: infectious agents' devilry or defense mechanism of the host? J Leukoc Biol. 2007. 82:1027–1032.
Article26. Stellrecht KA. Evidence for polyclonal B cell activation as the mechanism for LCMV-induced autoimmune hemolytic anemia. Immunol Lett. 1992. 31:273–277.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Aplastic Crisis Secondary to Parvovirus B19 Infection
- An Adult with Aplastic Crisis induced by Human Parvovirus B19 as an Initial Presentation of Hereditary Spherocytosis
- The Aplastic Crisis of Hereditary Spherocytosis Due to Parvovirus B19 Infection
- A Case of Hereditary Spherocytosis Initially Manifested as an Aplastic Crisis Caused by Parvovirus B19 Infection
- Clinical Characteristics of Human Parvovirus B19 Infection in Children