J Korean Med Sci.
2007 Apr;22(2):311-317. 10.3346/jkms.2007.22.2.311.
Methylation Profiles of CpG Island Loci in Major Types of Human Cancers
- Affiliations
-
- 1Department of Pathology, Seoul National University Hospital, 28 Yeongeon-dong, Jongno-gu, Seoul, Korea. ghkang@snu.ac.kr
- 2Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- 3Department of Pathology, Kyung Hee University College of Medicine, Seoul, Korea.
- KMID: 1713193
- DOI: http://doi.org/10.3346/jkms.2007.22.2.311
Abstract
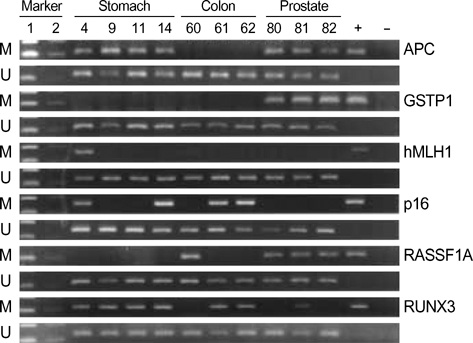
- Several reports have described aberrant methylation in various types of human cancers. However, the interpretation of methylation frequency in various human cancers has some limitations because of the different materials and methods used for methylation analysis. To gain an insight into the role of DNA hypermethylation in human cancers and allow direct comparison of tissue specific methylation, we generated methylation profiles in 328 human cancers, including 24 breast, 48 colon, 61 stomach, 48 liver, 37 larynx, 24 lung, 40 prostate, and 46 uterine cervical cancer samples by analyzing CpG island hypermethylation of 13 genes using methylation-specific PCR. The mean numbers of methylated genes were 6.5, 4.4, 3.6, 3.4, 3.1, 3.1, 3.1, and 2.1 in gastric, liver, prostate, larynx, colon, lung, uterine cervix, and in breast cancer samples, respectively. The number of genes that were methylated at a frequency of more than 40% in each tumor type ranged from nine (stomach) to one (breast). Generally genes frequently methylated in a specific cancer type differed from those methylated in other cancer types. The findings indicate that aberrant CpG island hypermethylation is a frequent finding in human cancers of various tissue types, and each tissue type has its own distinct methylation pattern.
MeSH Terms
Figure
Reference
-
1. Larsen F, Gundersen G, Lopez R, Prydz H. CpG islands as gene markers in the human genome. Genomics. 1992. 13:1095–1107.
Article2. Antequera F, Bird A. Number of CpG islands and genes in human and mouse. Proc Natl Acad Sci USA. 1993. 90:11995–11999.
Article3. Ponger L, Duret L, Mouchiroud D. Determinants of CpG islands: expression in early embryo and isochore structure. Genome Res. 2001. 11:1854–1860.
Article4. Nan X, Ng HH, Johnson CA, Laherty CD, Turner BM, Eisenman RN, Bird A. Transcriptio-nal repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature. 1998. 393:386–389.
Article5. Jones PL, Veenstra GJ, Wade PA, Vermaak D, Kass SU, Landsberger N, Strouboulis J, Wolffe AP. Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nat Genet. 1998. 19:187–191.
Article6. Bird AP, Wolffe AP. Methylation-induced repression-belts, braces, and chromatin. Cell. 1999. 99:451–454.7. Jones PA, Takai D. The role of DNA methylation in mammalian epigenetics. Science. 2001. 293:1068–1070.
Article8. Baylin SB, Herman JG, Graff JR, Vertino PM, Issa JP. Alterations in DNA methylation: A functional aspect of neoplasia. Adv Cancer Res. 1998. 72:141–196.9. Jones PA, Laird PW. Cancer epigenetic comes of age. Nat Genet. 1999. 21:163–167.10. Costello JF, Fruhwald MC, Smiraglia DJ, Rush LJ, Robertson GP, Gao X, Wright FA, Feramisco JD, Peltomaki P, Lang JC, Schuller DE, Yu L, Bloomfield CD, Caligiuri MA, Yates A, Nishikawa R, Su Huang H, Petrelli NJ, Zhang X, O'Dorisio MS, Held WA, Cavenee WK, Plass C. Aberrant CpG-island methylation has non-random and tumour-type-specific patterns. Nat Genet. 2000. 24:132–138.
Article11. Esteller M, Corn PG, Baylin SB, Herman JG. A gene hypermethylation profile of human cancer. Cancer Res. 2001. 61:3225–3229.12. Parkin DM, Pisani P, Ferlay J. Global cancer statistics. Ca Cancer J Clin. 1999. 49:33–64.
Article13. Herman JG, Graff JR, Myohanen S, Nelkin BD, Baylin SB. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci USA. 1996. 93:9821–9826.
Article14. Toyota M, Ahuja N, Ohe-Toyota M, Herman JG, Baylin SB, Issa JP. CpG island methylator phenotype in colorectal cancer. Proc Natl Acad Sci USA. 1999. 96:8681–8686.
Article15. Toyota M, Ahuja N, Suzuki H, Itoh F, Ohe-Toyota M, Imai K, Baylin SB, Issa JP. Aberrant methylation in gastric cancer associated with the CpG island methylator phenotype. Cancer Res. 1999. 59:5438–5442.16. Kang GH, Lee HJ, Hwang KS, Lee S, Kim JH, Kim JS. Aberrant CpG island hypermethylation of chronic gastritis, in relation to aging, gender, intestinal metaplasia, and chronic inflammation. Am J Pathol. 2003. 163:1551–1556.
Article17. Bai H, Gu L, Zhou J, Deng D. p16 hypermethylation during gastric carcinogenesis of Wistar rats by N-methyl-N'-nitro-N-nitrosoguanidine. Mutat Res. 2003. 535:73–78.
Article18. Lee YW, Klein CB, Kargacin B, Salnikow K, Kitahara J, Dowjat K, Zhitkovich A, Christie NT, Costa M. Carcinogenic nickel silences gene expression by chromatin condensation and DNA methylation: a new model for epigenetic carcinogens. Mol Cell Biol. 1995. 15:2547–2557.
Article19. Sutherland JE, Peng W, Zhang QW, Costa M. The histone deacetylases inhibitor A reduces nickel-induced gene silencing in yeast and mammalian cells. Mutat Res. 2001. 479:225–233.20. Maekita T, Nakazawa K, Mihara M, Nakajima T, Yanaoka K, Iguchi M, Arii K, Kaneda A, Tsukamoto T, Tatematsu M, Tamura G, Saito D, Sugimura T, Ichinose M, Ushijima T. High levels of aberrant DNA methylation in Helicobacter pylori-infected gastric mucosae and its possible association with gastric cancer risk. Clin Cancer Res. 2006. 12:989–995.
Article21. Maekita T, Nakazawa K, Mihara M, Nakajima T, Yanaoka K, Iguchi M, Arii K, Kaneda A, Tsukamoto T, Tatematsu M, Tamura G, Saito D, Sugimura T, Ichinose M, Ushijima T. High levels of aberrant DNA methylation in Helicobacter pylori-infected gastric mucosae and its possible association with gastric cancer risk. Clin Cancer Res. 2006. 12:989–995.
Article22. Ahuja N, Li Q, Mohan AL, Baylin SB, Issa JP. Aging and DNA methylation in colorectal mucosa and cancer. Cancer Res. 1998. 58:5489–5494.23. Esteller M, Garcia-Foncillas J, Andion E, Goodman SN, Hidalgo OF, Vanaclocha V, Baylin SB, Herman JG. Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N Engl J Med. 2000. 343:1350–1354.24. Esteller M, Gaidano G, Goodman SN, Zagonel V, Capello D, Botto B, Rossi D, Gloghini A, Vitolo U, Carbone A, Baylin SB, Herman JG. Hypermethylation of the DNA repair gene O(6)-methylguanine DNA methyltransferase and survival of patients with diffuse large B-cell lymphoma. J Natl Cancer Inst. 2002. 94:26–32.
Article25. Li QL, Ito K, Sakakura C, Fukamachi H, Inoue K, Chi XZ, Lee KY, Nomura S, Lee CW, Han SB, Kim HM, Kim WJ, Yamamoto H, Yamashita N, Yano T, Ikeda T, Itohara S, Inazawa J, Abe T, Hagiwara A, Yamagishi H, Ooe A, Kaneda A, Sugimura T, Ushijima T, Bae SC, Ito Y. Causal relationship between the loss of RUNX3 expression and gastric cancer. Cell. 2002. 109:113–124.
Article26. Ku JL, Kang SB, Shin YK, Kang HC, Hong SH, Kim IJ, Shin JH, Han IO, Park JG. Promoter hypermethylation downregulates RUNX3 gene expression in colorectal cancer cell lines. Oncogene. 2004. 23:6736–6742.
Article27. Mori T, Nomoto S, Koshikawa K, Fujii T, Sakai M, Nishikawa Y, Inoue S, Takeda S, Kaneko T, Nakao A. Decreased expression and frequent allelic inactivation of the RUNX3 gene at 1p36 in human hepatocellular carcinoma. Liver Int. 2005. 25:380–388.
Article28. Park WS, Cho YG, Kim CJ, Song JH, Lee YS, Kim SY, Nam SW, Lee SH, Yoo NJ, Lee JY. Hypermethylation of the RUNX3 gene in hepatocellular carcinoma. Exp Mol Med. 2005. 37:276–281.
Article29. Li QL, Kim HR, Kim WJ, Choi JK, Lee YH, Kim HM, Li LS, Kim H, Chang J, Ito Y, Youl Lee K, Bae SC. Transcriptional silencing of the RUNX3 gene by CpG hypermethylation is associated with lung cancer. Biochem Biophys Res Commun. 2004. 314:223–228.
Article30. Wada M, Yazumi S, Takaishi S, Hasegawa K, Sawada M, Tanaka H, Ida H, Sakakura C, Ito K, Ito Y, Chiba T. Frequent loss of RUNX3 gene expression in human bile duct and pancreatic cancer cell lines. Oncogene. 2004. 23:2401–2407.
Article31. Sakakura C, Hasegawa K, Miyagawa K, Nakashima S, Yoshikawa T, Kin S, Nakase Y, Yazumi S, Yamagishi H, Okanoue T, Chiba T, Hagiwara A. Possible involvement of RUNX3 silencing in the peritoneal metastases of gastric cancers. Clin Cancer Res. 2005. 11:6479–6488.
Article32. Kang GH, Lee S, Lee HJ, Hwang KS. Aberrant CpG island hypermethylation of multiple genes in prostate cancer and prostatic intraepithelial neoplasia. J Pathol. 2004. 202:233–240.
Article33. Kim WJ, Kim EJ, Jeong P, Quan C, Kim J, Li QL, Yang JO, Ito Y, Bae SC. RUNX3 inactivation by point mutations and aberrant DNA methylation in bladder tumors. Cancer Res. 2005. 65:9347–9354.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- CpG Island Hypermethylation in Gastric Carcinoma and Its Premalignant Lesions
- DNA Methylation Changes in Human Cancers
- Chromosomal Losses are Associated with Hypomethylation of the Gene-Control Regions in the Stomach with a Low Number of Active Genes
- Diffuse Large B Cell Lymphoma Shows Distinct Methylation Profiles of the Tumor Suppressor Genes among the Non-Hodgkin's Lymphomas
- The Relationship between the Methylenetetrahydrofolate Reductase Genotypes and the Methylation Status of the CpG Island Loci, LINE-1 and Alu in Prostate Adenocarcinoma