J Vet Sci.
2013 Jun;14(2):167-173. 10.4142/jvs.2013.14.2.167.
Identification of differentially expressed genes in gauze-exposed omentum of dogs using differential display RT-PCR
- Affiliations
-
- 1Department of Veterinary Surgery, College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea. ohkweon@snu.ac.kr
- 2Department of Plant Science, Research Institute for College of Agriculture and Life Sciences, Seoul National University, Seoul 151-742, Korea.
- KMID: 1705518
- DOI: http://doi.org/10.4142/jvs.2013.14.2.167
Abstract
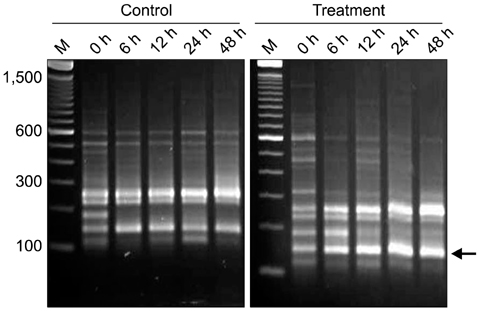
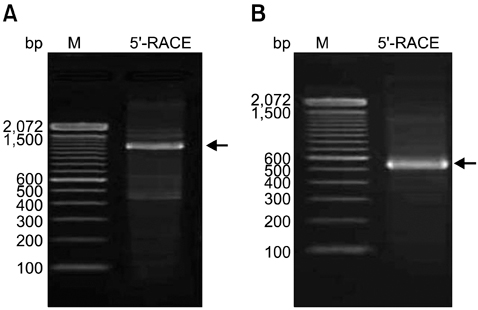
- Molecular mechanisms governing peritonitis caused by the presence of aseptic gauze have remained unclear. To identify the genes involved, sterile gauze-exposed omentum was collected at 0, 6, 12, 24, and 48 h intervals, and analyzed by differential display RT(reverse transcription)-PCR. Among over 1,200 bands, 230 bands were found differentially expressed. These bands represented the fragment sizes of approximately 200 to 1,500 bp. The eight fragments were expressed differentially in the treatment group but not in the control. The sequences of two bands were similar to those of genes associated with the inflammatory process and a band was related to repair and regeneration process. Another one was related with spermatogonia and the rest four were unknown. Additionally, amplicons corresponding to the full-length sequences of two inflammatory gene fragments were synthesized by rapid amplification of cDNA end PCR. One showed 99% similarity to the major histocompatibility complex class II dog leukocyte antigen-DR beta chain and the other was canis familiaris proteasome beta type 3. Results of the present study suggested that sterile gauze induced the differential expression of genes in the omentum involved in inflammation and healing process.
Keyword
MeSH Terms
-
Animals
*Bandages
Base Sequence
DNA, Complementary/analysis
Dogs/*genetics/metabolism
Gene Expression Profiling/veterinary
Gene Expression Regulation
Histocompatibility Antigens Class II/*genetics/metabolism
Molecular Sequence Data
Omentum/*metabolism
Proteasome Endopeptidase Complex/*genetics/metabolism
RNA, Messenger/analysis
Reverse Transcriptase Polymerase Chain Reaction/veterinary
*Wound Healing
DNA, Complementary
Histocompatibility Antigens Class II
Proteasome Endopeptidase Complex
RNA, Messenger
Figure
Reference
-
1. Azouz A, Razzaque MS, El-Hallak M, Taguchi T. Immunoinflammatory responses and fibrogenesis. Med Electron Microsc. 2004; 37:141–148.
Article2. Bush JM, Gardiner DW, Palmer JS, Rajpert-De Meyts E, Veeramachaneni DNR. Testicular germ cell tumors in dogs are predominantly of spermatocytic seminoma type and are frequently associated with somatic cell tumors. Int J Androl. 2011; 34(4 Pt 2):e288–e295.3. Chandra A, Srivastava RK, Kashyap MP, Kumar R, Srivastava RN, Pant AB. The anti-inflammatory and antibacterial basis of human omental defense: selective expression of cytokines and antimicrobial peptides. PLoS One. 2011; 6:e20446.
Article4. Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002; 82:373–428.
Article5. Haley B, Paunesku T, Protic M, Woloschak GE. Response of heterogenous ribonuclear proteins (hnRNP) to ionising radiation and their involvement in DNA damage repair. Int J Radiat Biol. 2009; 85:643–655.
Article6. Hwang IT, Kim YJ, Kim SH, Kwak CI, Gu YY, Chun JY. Annealing control primer system for improving specificity of PCR amplification. Biotechniques. 2003; 35:1180–1184.
Article7. Jani K, Saxena AK, Vaghasia R. Omental plugging for large-sized duodenal peptic perforations: a prospective randomized study of 100 patients. South Med J. 2006; 99:467–471.
Article8. Kanazawa S, Ota S, Sekine C, Tada T, Otsuka T, Okamoto T, Sønderstrup G, Peterlin BM. Aberrant MHC class II expression in mouse joints leads to arthritis with extraarticular manifestations similar to rheumatoid arthritis. Proc Natl Acad Sci U S A. 2006; 103:14465–14470.
Article9. Kao RH, Francia G, Poulsom R, Hanby AM, Hart IR. Application of differential display, with in situ hybridization verification, to microscopic samples of breast cancer tissue. Int J Exp Pathol. 2003; 84:207–212.
Article10. Kim YS, Do JH, Bae S, Bae DH, Ahn WS. Identification of differentially expressed genes using an annealing control primer system in stage III serous ovarian carcinoma. BMC Cancer. 2010; 10:576.
Article11. Liang P, Averboukh L, Pardee AB. Distribution and cloning of eukaryotic mRNAs by means of differential display: refinements and optimization. Nucleic Acids Res. 1993; 21:3269–3275.
Article12. Liang P, Pardee AB. Differential display. A general protocol. Mol Biotechnol. 1998; 10:261–267.
Article13. Maimunah M, Froemming GA, Nawawi H, Nafeeza MI, Effat O, Rosmadi MY, Mohamed Saifulaman MS. Identification of differentially expressed gene (DEG) in atherosclerotic lesion by annealing control primer (ACP)-based Genefishing™PCR. World Acad Sci Eng Technol. 2012; 67:194–199.14. Nagasaka T, Boulday G, Fraser CC, Coupel S, Coulon F, Tesson L, Heslan JM, Soulillou JP, Charreau B. Rapid selection of differentially expressed genes in TNFα-activated endothelial cells. Mol Med. 2002; 8:559–567.
Article15. Nathan C. Points of control in inflammation. Nature. 2002; 420:846–852.
Article16. Risher WH, McKinnon WM. Foreign body in the gastrointestinal tract: intraluminal migration of laparotomy sponge. South Med J. 1991; 84:1042–1045.17. Risom L, Møller P, Loft S. Oxidative stress-induced DNA damage by particulate air pollution. Mutat Res. 2005; 592:119–137.
Article18. Schreiber S, Gehrckens A, Raedler A. Activation of the major omentum-associated lymphoid tissue in Crohn disease. Zentralbl Chir. 1995; 120:135–144.19. Ting JP, Trowsdale J. Genetic control of MHC class II expression. Cell. 2002; 109:Suppl. S21–S33.
Article20. Whitcup SM, Nussenblatt RB, Price FW Jr, Chan CC. Expression of cell adhesion molecules in corneal graft failure. Cornea. 1993; 12:475–480.
Article21. Yoshikawa Y, Mukai H, Asada K, Hino F, Kato I. Differential display with carboxy-X-rhodamine-labeled primers and the selection of differentially amplified cDNA fragments without cloning. Anal Biochem. 1998; 256:82–91.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Identification of differentially expressed genes using an annealing control primer system in periodontitis
- Idenfitied Differentially Expressed Genes in Keratoconus
- Identification of Differentially Expressed Genes from Serum Deprived p388D1 Cells
- Identification of Differentially Expressed Genes in Murine Hippocampus by Modulation of Nitric Oxide in Kainic Acid-induced Neurotoxic Animal Model
- The Differentially Expressed Genes by Radiotherapy in the Patients with Uterine Cervix Cancer