Korean J Physiol Pharmacol.
2010 Apr;14(2):113-118. 10.4196/kjpp.2010.14.2.113.
Effects of Saccharin Intake on Hippocampal and Cortical Plasticity in Juvenile and Adolescent Rats
- Affiliations
-
- 1Department of Physiology, Seoul National University School of Dentistry, Seoul 110-749, Korea. sychoi@snu.ac.kr
- 2Department of Oral and Maxillofacial Surgery, Dental Research Institute, Seoul National University School of Dentistry, Seoul 110-749, Korea.
- KMID: 1457677
- DOI: http://doi.org/10.4196/kjpp.2010.14.2.113
Abstract
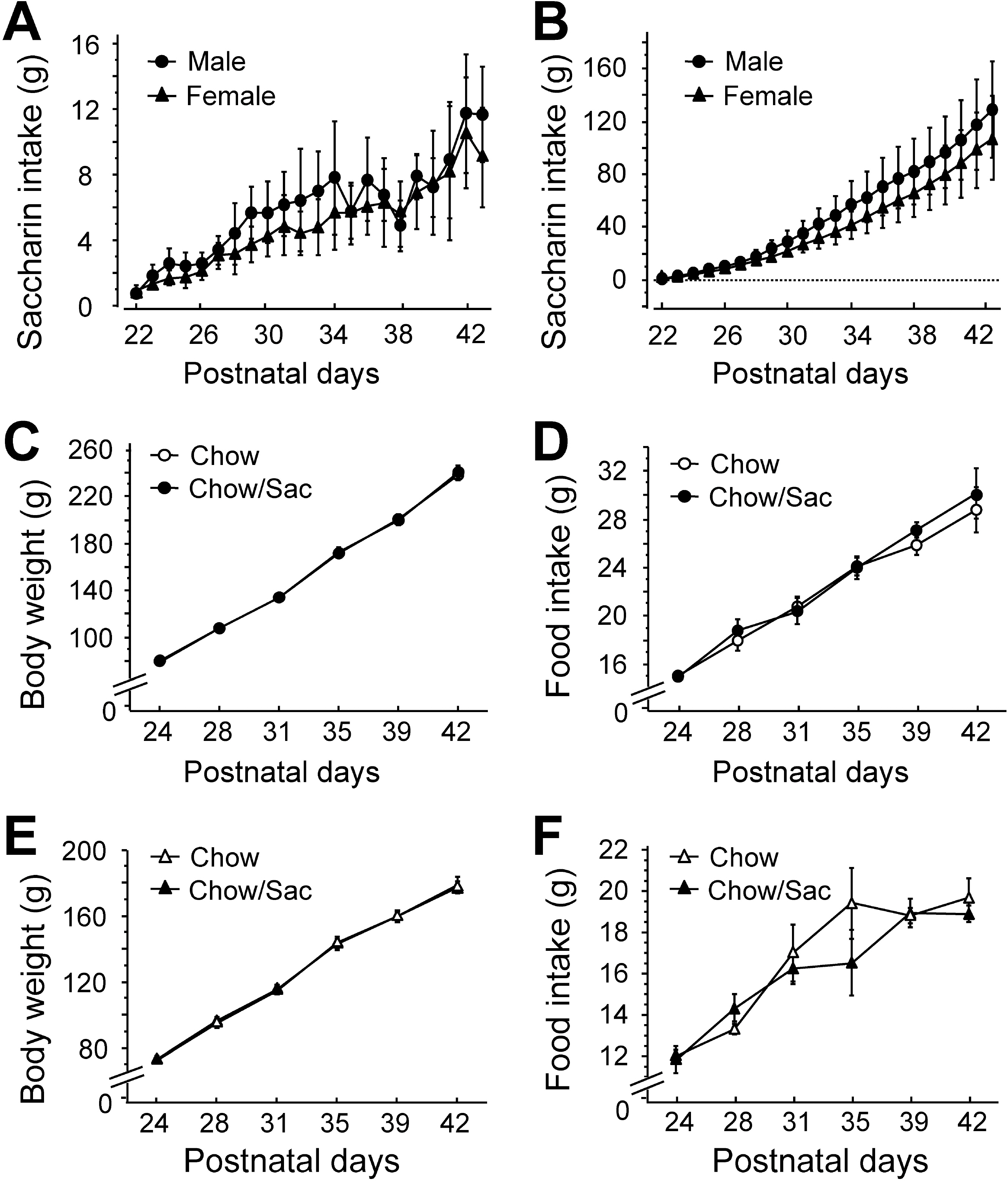
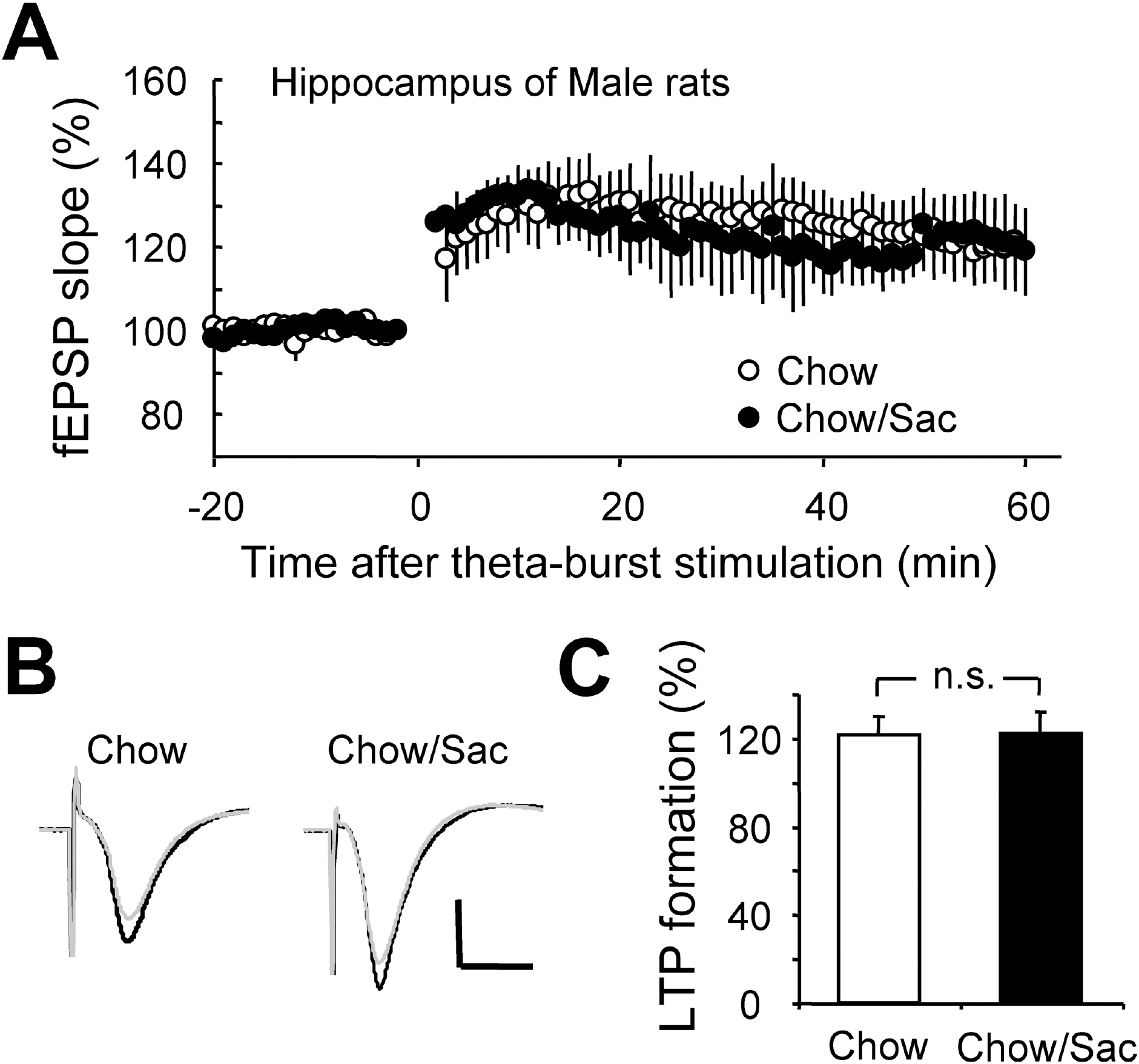
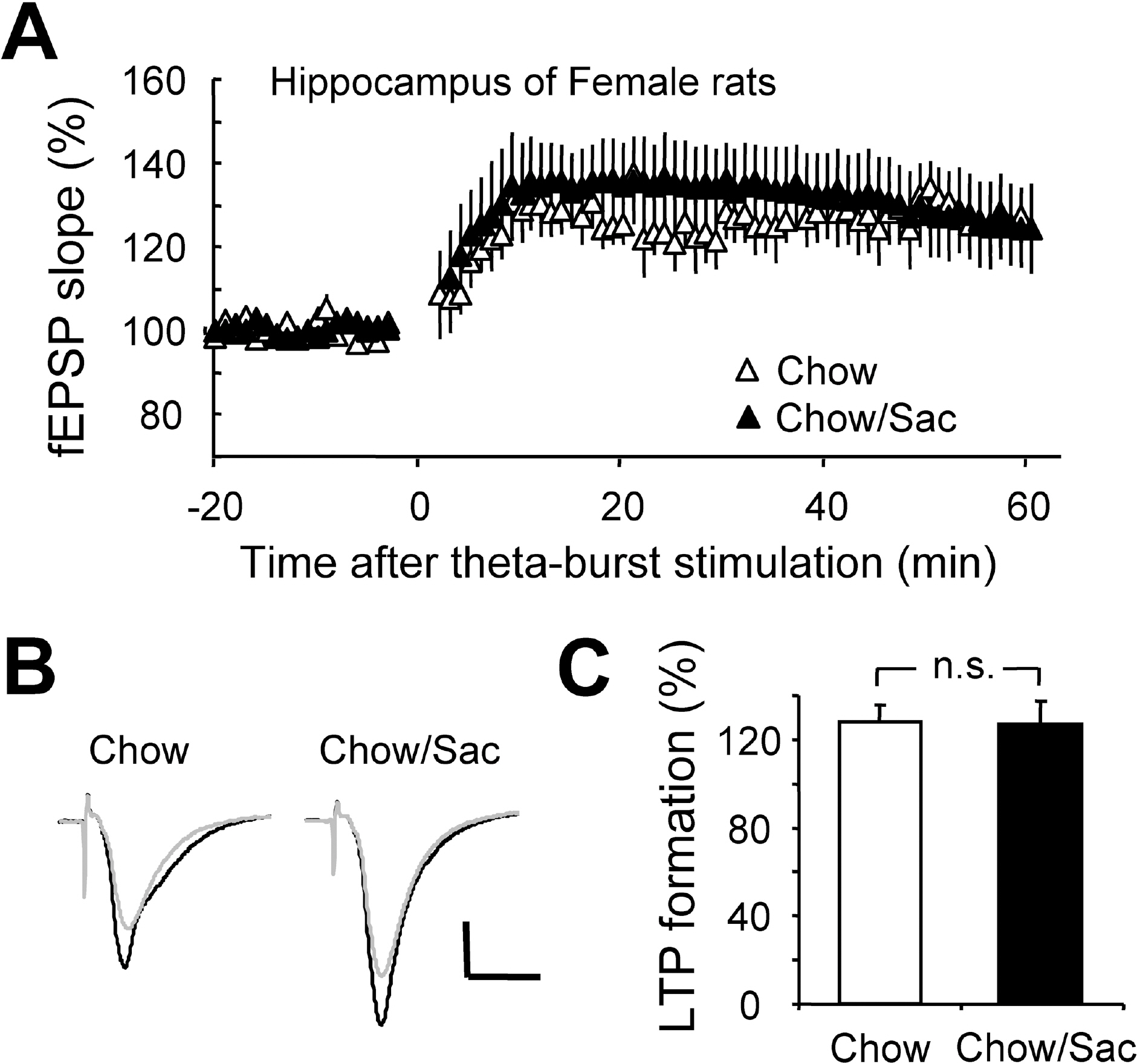
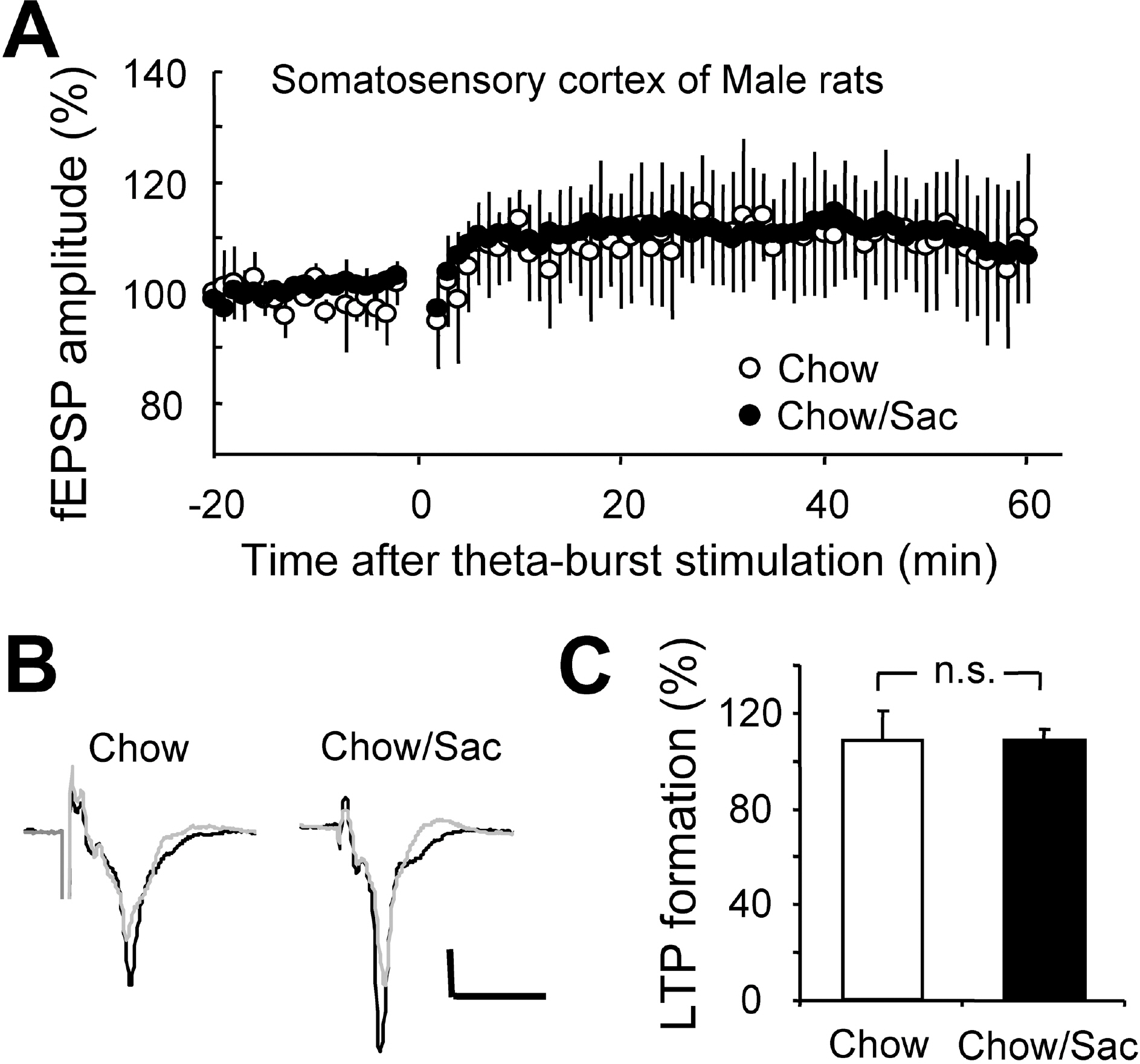
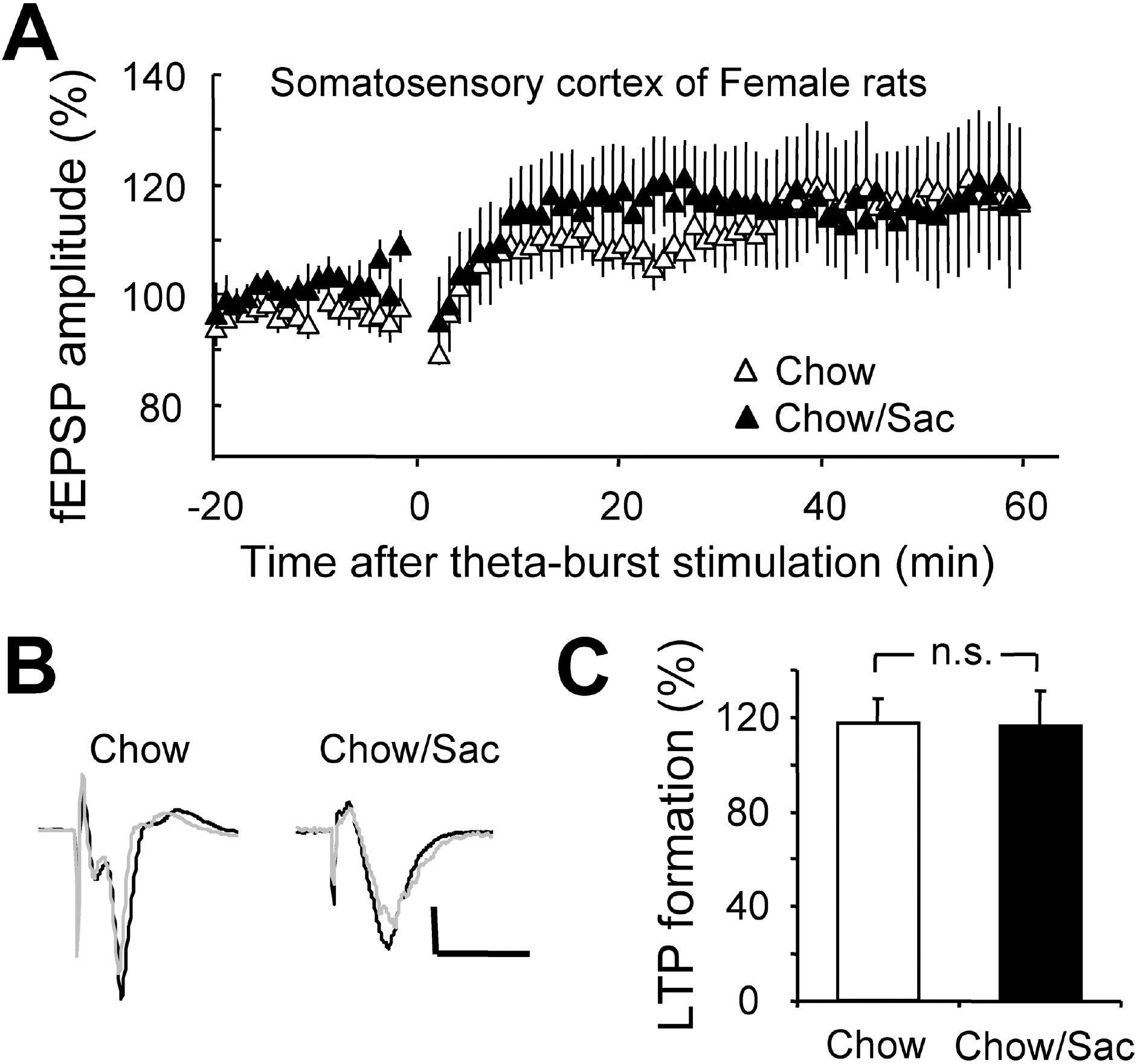
- The sensory system is developed and optimized by experiences given in the early phase of life in association with other regions of the nervous system. To date, many studies have revealed that deprivation of specific sensory experiences can modify the structure and function of the central nervous system; however, the effects of sensory overload remains unclear. Here we studied the effect of overloading the taste sense in the early period of life on the synaptic plasticity of rat hippocampus and somatosensory cortex. We prepared male and female Sprague Dawley rats with ad libitum access to a 0.1% saccharin solution for 2 hrs per day for three weeks after weaning on postnatal day 22. Saccharin consumption was slightly increased in males compared with females; however, saccharin intake did not affect chow intake or weight gain either in male or in female rats. We examined the effect of saccharin-intake on long term potentiation (LTP) formation in hippocampal Schaffer collateral pathway and somatosensory cortex layer IV - II/III pathways in the 6-week old saccharin-fed rats. There was no significant difference in LTP formation in the hippocampus between the control group and saccharin-treated group in both male and female rats. Also in the somatosensory cortex, we did not see a significant difference in LTP among the groups. Therefore, we conclude that saccharin-intake during 3~6 weeks may not affect the development of physiological function of the cortical and hippocampal synapses in rats.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Long-term Synaptic Plasticity: Circuit Perturbation and Stabilization
Joo Min Park, Sung-Cherl Jung, Su-Yong Eun
Korean J Physiol Pharmacol. 2014;18(6):457-460. doi: 10.4196/kjpp.2014.18.6.457.
Reference
-
References
1. Touhara K, Vosshall LB. Sensing odorants and pheromones with chemosensory receptors. Annu Rev Physiol. 2009; 71:307–332.
Article2. Burnstock G. Purines and sensory nerves. Handb Exp Pharmacol. 2009; 194:333–392.
Article3. Hubel DH, Wiesel TN. The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J Physiol. 1970; 206:419–436.
Article4. Draeger UC. Observations on monocular deprivation in mice. J Neurophysiol. 1978; 41:28–42.5. Berardi N, Pizzorusso T, Maffei L. Critical periods during sensory development. Curr Opin Neurobiol. 2000; 10:138–145.
Article6. Frenkel MY, Bear MF. How monocular deprivation shifts ocular dominance in visual cortex of young mice. Neuron. 2004; 44:917–923.
Article7. Hensch TK. Critical period mechanisms in developing visual cortex. Curr Top Dev Biol. 2005; 69:215–237.
Article8. Sotnikov OS. Primary sensory neurons in the central nervous system. Neurosci Behav Physiol. 2006; 36:541–548.
Article9. Johnson BA, Woo CC, Zeng Y, Xu Z, Hingco EE, Ong J, Leon M. Prolonged stimulus exposure reveals prolonged neurobehavioral response patterns. J Comp Neurol. 2010; 518:1617–1629.
Article10. Mitchell DE, Sengpiel F. Neural mechanisms of recovery following early visual deprivation. Philos Trans R Soc Lond B Biol Sci. 2009; 364:383–398.
Article11. Knudsen EI. Experience alters the spatial tuning of auditory units in the optic tectum during a sensitive period in the barn owl. J Neurosci. 1985; 5:3094–3109.
Article12. Nakahara H, Zhang LI, Merzenich MM. Specialization of primary auditory cortex processing by sound exposure in the “critical period”. Proc Natl Acad Sci U S A. 2004; 101:7170–7174.
Article13. Smith JC. Microstructure of the rat's intake of food, sucrose and saccharin in 24-hour tests. Neurosci Biobehav Rev. 2000; 24:199–212.
Article14. Choi SY, Chang J, Jiang B, Seol GH, Min SS, Han JS, Shin HS, Gallagher M, Kirkwood A. Multiple receptors coupled to phospholipase C gate long-term depression in visual cortex. J Neurosci. 2005; 25:11433–11443.
Article15. Kirkwood A, Bear MF. Hebbian synapses in visual cortex. J Neurosci. 1994; 14:1634–1645.
Article16. Porciatti V, Bonanni P, Fiorentini A, Guerrini R. Lack of cortical contrast gain control in human photosensitive epilepsy. Nat Neurosci. 2000; 3:259–263.
Article17. Chang EF, Merzenich MM. Environmental noise retards auditory cortical development. Science. 2003; 300:498–502.
Article18. Noreña AJ, Gourèvitch B, Aizawa N, Eggermont JJ. Spectrally enhanced acoustic environment disrupts frequency representation in cat auditory cortex. Nat Neurosci. 2006; 9:932–939.
Article19. Yarmolinsky DA, Zuker CS, Ryba NJ. Common sense about taste: from mammals to insects. Cell. 2009; 139:234–244.
Article20. Collier G, Novell K. Saccharin as a sugar surogate. J Comp Physiol Psychol. 1967; 64:404–408.21. Smith JC, Sclafani A. Saccharin as a sugar surogate revisited. Appetite. 2002; 38:155–160.22. Yamamoto T. Cnetral mechanisms of roles of taste in reward and eating. Acta Physiol Hung. 2008; 95:165–186.23. Ulrich-Lai YM, Ostrander MM, Thomas IM, Packard BA, Furay AR, Dolgas CM, Van Hooren DC, Figueiredo HF, Mueller NK, Choi DC, Herman JP. Daily limited access to sweetened drink attenuates hypothalamic-pituitary-adrenocortical axis stress responses. Endocrinology. 2007; 148:1823–1883.
Article24. Kinzig KP, Hargrave SL, Honors MA. Binge-type eating attenuates corticosterone and hypophagic responses to restraint stress. Physiol Behav. 2008; 95:108–113.
Article25. Zhang TY, Cho HJ, Lee S, Lee JH, Choi SH, Ryu V, Yoo SB, Lee JY, Kim DG, Jahng JW. Impairments in water maze learning of aged rats that received dextromethorphan repeatedly during adolescent period. Psychopharmacology. 2007; 191:171–179.
Article26. Matthews K, Dalley JW, Matthews C, Tsai TH, Robbins TW. Periodic maternal separation of neonatal rats produces region-and gender-specific effects on biogenic amine content in postmortem adult brain. Synapse. 2001; 40:1–10.27. Slotten HA, Kalinichev M, Hagan JJ, Marsden CA, Fone KCF. Long-lasting changes in behavioural and neuroendocrine indices in the rat following neonatal maternal separation: Gender-dependent effects. Brain Res. 2006; 1097:123–132.
Article28. Desbonnet L, Garrett L, Daly E, McDermott KW, Dinan TG. Sexually dimorphic effects of maternal separation stress on corticotrophin-releasing factor and vasopressin systems in the adult rat brain. Int J Devl Neurosci. 2008; 26:259–268.
Article29. Goel A, Jiang B, Xu LW, Song L, Kirkwood A, Lee HK. Cross-modal regulation of synaptic AMPA receptors in primary sensory cortices by visual experience. Nat Neurosci. 2006; 9:1001–1003.
Article30. Collignon O, Voss P, Lassonde M, Lepore F. Cross-modal plasticity for the spatial processing of sounds in visually deprived subjects. Exp Brain Res. 2009; 192:343–358.
Article31. Giza CC, Kolb B, Harris NG, Asarnow RF, Prins ML. Hitting a moving target: basic mechanisms of recovery from acquired developmental brain injury. Dev Neurorehabil. 2009; 12:255–268.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Saccharin Intake in Restraint-induced Stress Response Reduction in Rats
- A priming role of local estrogen on exogenous estrogen-mediated synaptic plasticity and neuroprotection
- Neuroprotection of the Developing Brain by Dexmedetomidine Is Mediated by Attenuating Single Propofol-induced Hippocampal Apoptosis and Synaptic Plasticity Deficits
- The Alleviating Effects of Sweet Drinks on Restraint Stress-Induced Anxiety and Depressive Behavior in Adolescent Rats
- Observation on the Hippocampal Mossy Fiber Sprouting After Hypoxic-Ischemic Neuronal Loss in Juvenile and Mature Rat Brain