Lab Anim Res.
2012 Sep;28(3):181-191. 10.5625/lar.2012.28.3.181.
Aqueous extract of Liriope platyphylla, a traditional Chinese medicine, significantly inhibits abdominal fat accumulation and improves glucose regulation in OLETF type II diabetes model rats
- Affiliations
-
- 1College of Natural Resources & Life Science, Pusan National University, Miryang, Korea. dyhwang@pusan.ac.kr
- 2Pusan National University-Wellbeing Products Center, Miryang, Korea.
- 3College of Human Ecology, Pusan National University, Pusan, Korea.
- 4Department of Experimental Animal Research, Clinical Research Institute, Seoul National University Hospital, Seoul, Korea.
- KMID: 1436723
- DOI: http://doi.org/10.5625/lar.2012.28.3.181
Abstract
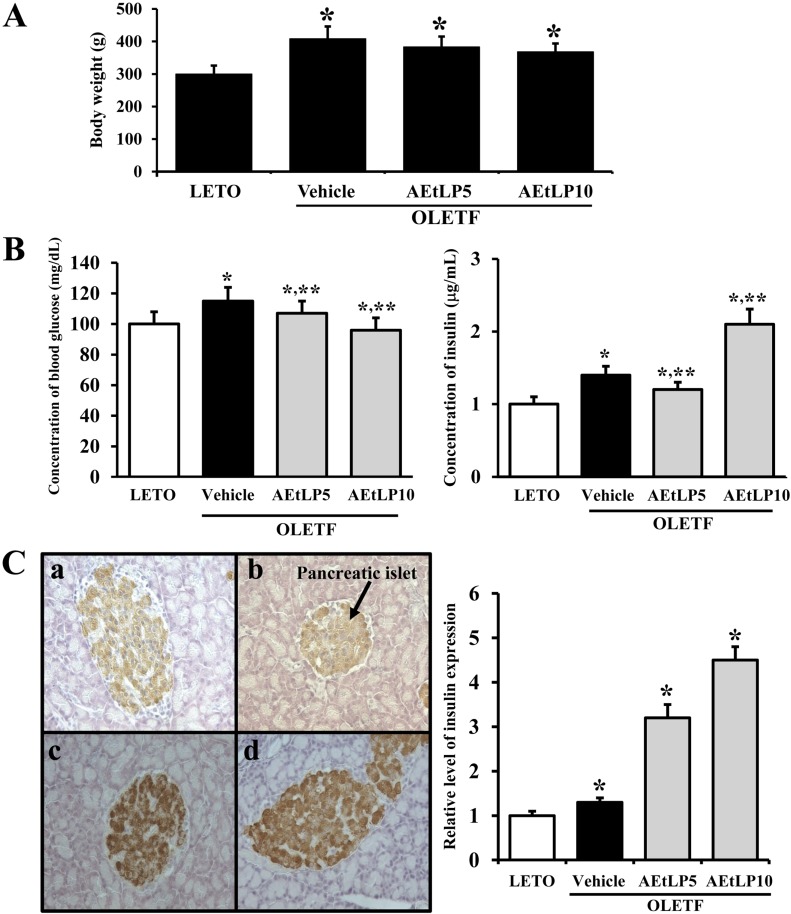
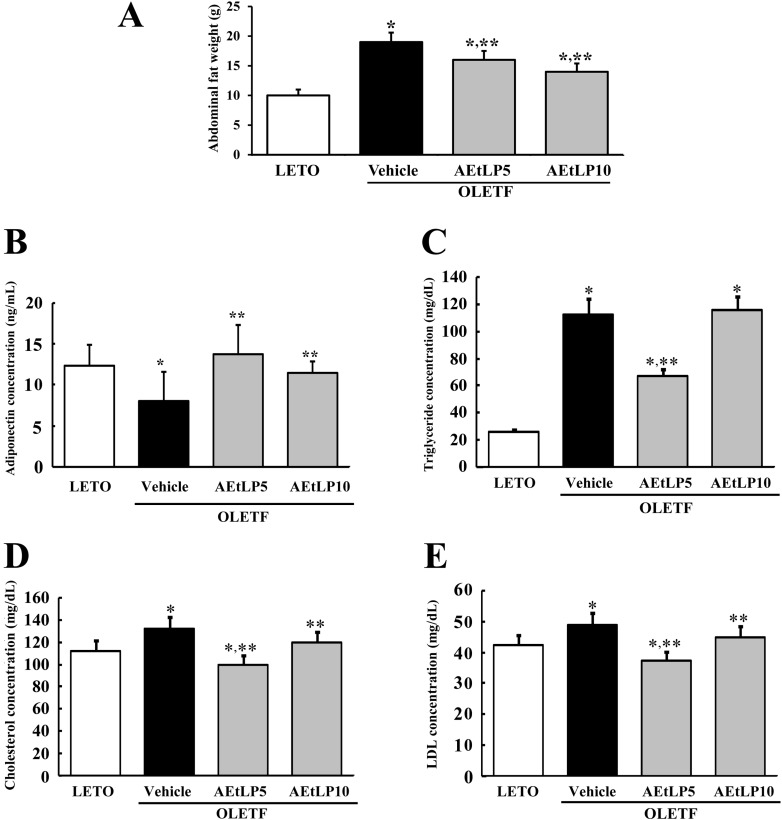
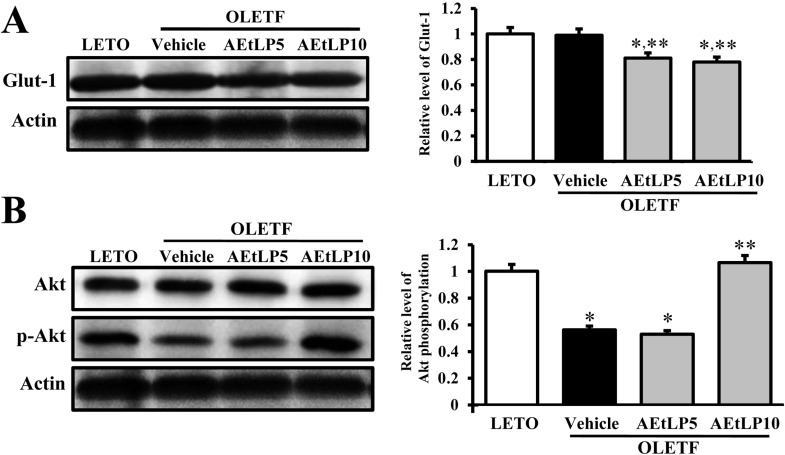
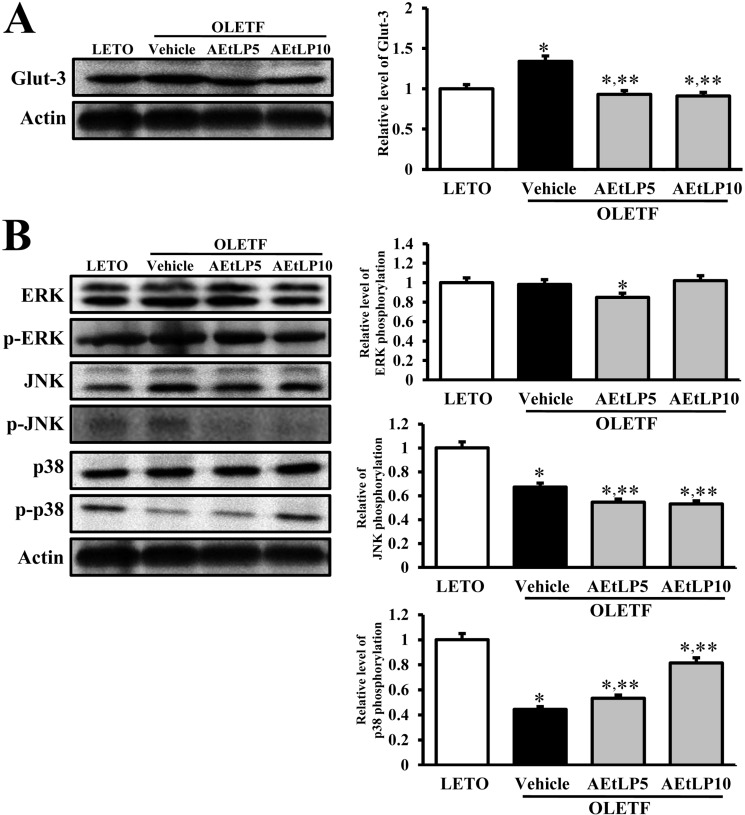
- Liriope platyphylla is a medical herb that has long been used in Korea and China to treat cough, sputum, neurodegenerative disorders, obesity, and diabetes. The aims of this study were to determine the antidiabetic and antiobesity effects of aqueous extract of L. platyphylla (AEtLP) through glucose and lipid regulation in both pre-diabetes and obesity stage of type II diabetes model. Two concentrations of AEtLP were orally administrated to OLETF (Otsuka Long-Evans Tokushima Fatty) rats once a day for 2 weeks, after which changes in glucose metabolism and fat accumulation were measured. Abdominal fat mass dramatically decreased in AEtLP-treated OLETF rats, whereas glucose concentration slightly decreased in all AEtLP-treated rats. However, compared to vehicle-treated OLETF rats, only AEtLP10 (10% concentration)-treated OLETF rats displayed significant induction of insulin production, whereas AEtLP5 (5% concentration)-treated OLETF rats showed a lower level of insulin. Although serum adiponectin level increased in only AEtLP5-treated rats, significant alteration of lipid concentration was detected in AEtLP5-treated OLETF rats. Expression of Glut-1 decreased in all AEtLP-treated rats, whereas Akt phosphorylation increased only in AEtLP10-treated OLETF rats. Furthermore, the pattern of Glut-3 expression was very similar with that of Glut-1 expression, which roughly corresponded with the phosphorylation of c-Jun N-teminal kinase (JNK) and p38 in the mitogen-activated protein kinase pathway. Therefore, these findings suggest that AEtLP should be considered as a therapeutic candidate during pre-diabetes and obesity stage capable of inducing insulin secretion from pancreatic beta-cells, glucose uptake in liver cells, as well as a decrease in fat and lipid accumulation.
MeSH Terms
-
Abdominal Fat
Adiponectin
Animals
China
Cough
Glucose
Glucose Transport Proteins, Facilitative
Insulin
Korea
Liver
Medicine, Chinese Traditional
Neurodegenerative Diseases
Obesity
Phosphorylation
Phosphotransferases
Protein Kinases
Rats
Rats, Inbred OLETF
Sputum
Adiponectin
Glucose
Glucose Transport Proteins, Facilitative
Insulin
Phosphotransferases
Protein Kinases
Figure
Reference
-
1. Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004; 27(5):1047–1053. PMID: 15111519.
Article2. Idris I, Donnelly R. Sodium-glucose co-transporter-2 inhibitors: an emerging new class of oral antidiabetic drug. Diabetes Obes Metab. 2009; 11(2):79–88. PMID: 19125776.
Article3. Kokil GR, Rewatkar PV, Verma A, Thareja S, Naik SR. Pharmacology and chemistry of diabetes mellitus and antidiabetic drugs: a critical review. Curr Med Chem. 2010; 17(35):4405–4423. PMID: 20939806.4. Jung M, Park M, Lee HC, Kang YH, Kang ES, Kim SK. Antidiabetic agents from medicinal plants. Curr Med Chem. 2006; 13(10):1203–1218. PMID: 16719780.
Article5. Prabhakar PK, Doble M. A target based therapeutic approach towards diabetes mellitus using medicinal plants. Curr Diabetes Rev. 2008; 4(4):291–308. PMID: 18991598.
Article6. Huh MK, Huh HW, Choi JS, Lee BK. Genetic diversity and population structure of Liriope platyphylla (Liliaceae) in Korea. J Life Sci. 2007; 17:328–333.7. Lee YC, Lee JC, Seo YB, Kook YB. Liriopis tuber inhibit OVA-induced airway inflammation and bronchial hyperresponsiveness in murine model of asthma. J Ethnopharmacol. 2005; 101(1-3):144–152. PMID: 15982838.8. Choi SB, Wha JD, Park S. The insulin sensitizing effect of homoisoflavone-enriched fraction in Liriope platyphylla Wang et Tang via PI3-kinase pathway. Life Sci. 2004; 75(22):2653–2664. PMID: 15369701.9. Hur J, Lee P, Kim J, Kim AJ, Kim H, Kim SY. Induction of nerve growth factor by butanol fraction of Liriope platyphylla in C6 and primary astrocyte cells. Biol Pharm Bull. 2004; 27(8):1257–1260. PMID: 15305032.10. Jeong S, Chae K, Jung YS, Rho YH, Lee J, Ha J, Yoon KH, Kim GC, Oh KS, Shin SS, Yoon M. The Korean traditional medicine Gyeongshingangjeehwan inhibits obesity through the regulation of leptin and PPARalpha action in OLETF rats. J Ethnopharmacol. 2008; 119(2):245–251. PMID: 18674606.11. Hur J, Lee P, Moon E, Kang I, Kim SH, Oh MS, Kim SY. Neurite outgrowth induced by spicatoside A, a steroidal saponin, via the tyrosine kinase A receptor pathway. Eur J Pharmacol. 2009; 620(1-3):9–15. PMID: 19695245.
Article12. Kim JE, Lee YK, Nam SH, Choi SI, Goo JS, Jang MJ, Lee HS, Son HJ, Lee CY, Hwang DY. The symptoms of atopic dermatitis in NC/Nga mice were significantly relieved by the water extract of Liriope platyphylla. Lab Anim Res. 2010; 26:377–384.13. Lee YK, Kim JE, Nam SH, Goo JS, Choi SI, Choi YH, Bae CJ, Woo JM, Cho JS, Hwang DY. Differential regulation of the biosynthesis of glucose transporters by the PI3-K and MAPK pathways of insulin signaling by treatment with novel compounds from Liriope platyphylla. Int J Mol Med. 2011; 27(3):319–327. PMID: 21165549.
Article14. Kim JE, Nam SH, Choi SI, Hwang IS, Lee HR, Jang MJ, Lee CY, Soon HJ, Lee HS, Kim HS, Kang BC, Hong JT, Hwang DY. Aqueous extracts of Liriopeplaty phylla are tightly-regulated by insulin secretion from pancreatic islets and by increased glucose uptake through glucose transporters expressed in liver hepatocytes. Biomol Ther. 2011; 19(3):348–356.15. Roy S, Dontamalla SK, Mondru AK, Sannigrahi S, Veerareddy PR. Downregulation of apoptosis and modulation of TGF-â1 by sodium selenate prevents streptozotocin-induced diabetic rat renal impairment. Biol Trace Elem Res. 2011; 139(1):55–71. PMID: 20174975.
Article16. Taha C, Klip A. The insulin signaling pathway. J Membrane Biol. 1999; 169:1–12. PMID: 10227847.
Article17. Kim SW, Chang IM, Oh KB. Inhibition of the bacterial surface protein anchoring transpeptidase sortase by medicinal plants. Biosci Biotechnol Biochem. 2002; 66(12):2751–2754. PMID: 12596883.
Article18. Nam SH, Choi SI, Goo JS, Kim JE, Hwang IS, Lee HR, Lee YJ, Lee HG, Choi YH, Hwang DY. LP-M, a novel butanol-extracts isolated from Liriopeplatyphylla, coul dinduce the neuronal cell survival an dneuritic outgrowth in hippocampus of mice through Akt/ERK activation on NGF signal pathway. J Life Sci. 2011; 21(9):1234–1242.19. Kim HJ, Kim J, Kim SJ, Lee SH, Park YS, Park BK, Kim BS, Kim SK, Cho SD, Jung JW, Nam JS, Choi CS, Jung JY. Anti-inflammatory effect of quercetin on picryl chloride-induced contact dermatitis in BALB/c mice. Lab Anim Res. 2010; 26:7–13.
Article20. Kawano K, Hirashima T, Mori S, Natori T. OLETF (Otsuka Long-Evans Tokushima Fatty) rat: a new NIDDM rat strain. Diabetes Res Clin Pract. 1994; 24:S317–S320. PMID: 7859627.
Article21. Moran TH. Unraveling the obesity of OLETF rats. Physiol Behav. 2008; 94(1):71–78. PMID: 18190934.
Article22. Mori YY. Similarity and dissimilarity between the OLETF rats and obese individuals with NIDDM. 1999. Amsterdam: Elsevier.23. Sharma SB, Gupta S, Ac R, Singh UR, Rajpoot R, Shukla SK. Antidiabetogenic action of Morus rubra L. leaf extract in streptozotocin-induced diabetic rats. J Pharm Pharmacol. 2010; 62(2):247–255. PMID: 20487205.24. Gupta S, Sharma SB, Singh UR, Bansal SK, Prabhu KM. Elucidation of mechanism of action of Cassia auriculata leaf extract for its antidiabetic activity in streptozotocin-induced diabetic rats. J Med Food. 2010; 13(3):528–534. PMID: 20521978.25. Diez JJ, Iglesias P. The role of the novel adipocyte-derived hormone adiponectin in human disease. Eur J Endocrinol. 2003; 148(3):293–300. PMID: 12611609.
Article26. Ukkola O, Santaniemi M. Adiponectin: a link between excess adiposity and associated comorbidities? J Mol Med. 2002; 80(11):696–702. PMID: 12436346.
Article27. Kimura M, Shinozaki T, Tateishi N, Yoda E, Yamauchi H, Suzuki M, Hosoyamada M, Shibasaki T. Adiponectin is regulated differently by chronic exercise than by weight-matched food restriction in hyperphagic and obese OLETF rats. Life Sci. 2006; 79:2105–2111. PMID: 16889803.
Article28. Choi KC, Ryu OH, Lee KW, Kim HY, Seo JA, Kim SG, Kim NH, Choi DS, Baik SH, Choi KM. Effect of PPAR-α and -γ agonist on the expression of visfatin, adiponectin, and TNF-α in visceral fat of OLETF rats. Biochem Biophys Res Commun. 2005; 336:747–753. PMID: 16157299.
Article29. Mori Y, Oana F, Matsuzawa A, Akahane S, Tajima N. Short-term effect of bezafibrate on the expression of adiponectin mRNA in the adipose tissues. Endocrine. 2004; 25(3):247–251. PMID: 15758252.30. Lee YK, Kim JE, Nam SH, Goo JS, Choi SI, Choi YH, Bae CJ, Woo JM, Cho JS, Hwang DY. Differential regulation of the biosynthesis of glucose transporters by the PI3-K and MAPK pathways of insulin signaling by treatment with novel compounds from Liriopeplatyphylla. Int J Mol Med. 2011; 27:319–327. PMID: 21165549.
Article31. Kaur HD, Bansal MP. Studies on HDL associated enzymes under experimental hypercholesterolemia: possible modulation on selenium supplementation. Lipids Health Dis. 2009; 8:55. PMID: 20015371.
Article32. Oka Y, Asano T, Shibasaki Y, Lin JL, Tsukuda K, Katagiri H, Akanuma Y, Takaku F. C-terminal truncated glucose transporter is locked into an inward-facing form without transport activity. Nature. 1990; 345(6275):550–553. PMID: 2348864.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Anti-diabetic effects of benfotiamine on an animal model of type 2 diabetes mellitus
- Ultrastructural Changes in Corneas of Diabetic Otsuka Long-Evans Tokushima Fatty (OLETF) Rats
- Red Liriope platyphylla stimulated the insulin secretion through the regulation of calcium concentration in rat insulinoma cells and animal models
- Angiotensin-Converting Enzyme Inhibitor Ramipril Improves Coronary Microvascular Remodeling in Type II Diabetic Rats
- Precautionary effects of Red Liriope platyphylla on NGF secretion and Abeta42 deposition under the preclinical stage of Alzheimer's disease in Tg2576 mice