Korean J Physiol Pharmacol.
2022 Nov;26(6):531-540. 10.4196/kjpp.2022.26.6.531.
Group 1 metabotropic glutamate receptor 5 is involved in synaptically-induced Ca2+ -spikes and cell death in cultured rat hippocampal neurons
- Affiliations
-
- 1Department of Physiology, College of Medicine, The Catholic University of Korea, Seoul 06591, Korea
- 2Catholic Neuroscience Institute, The Catholic University of Korea, Seoul 06591, Korea
- KMID: 2534528
- DOI: http://doi.org/10.4196/kjpp.2022.26.6.531
Abstract
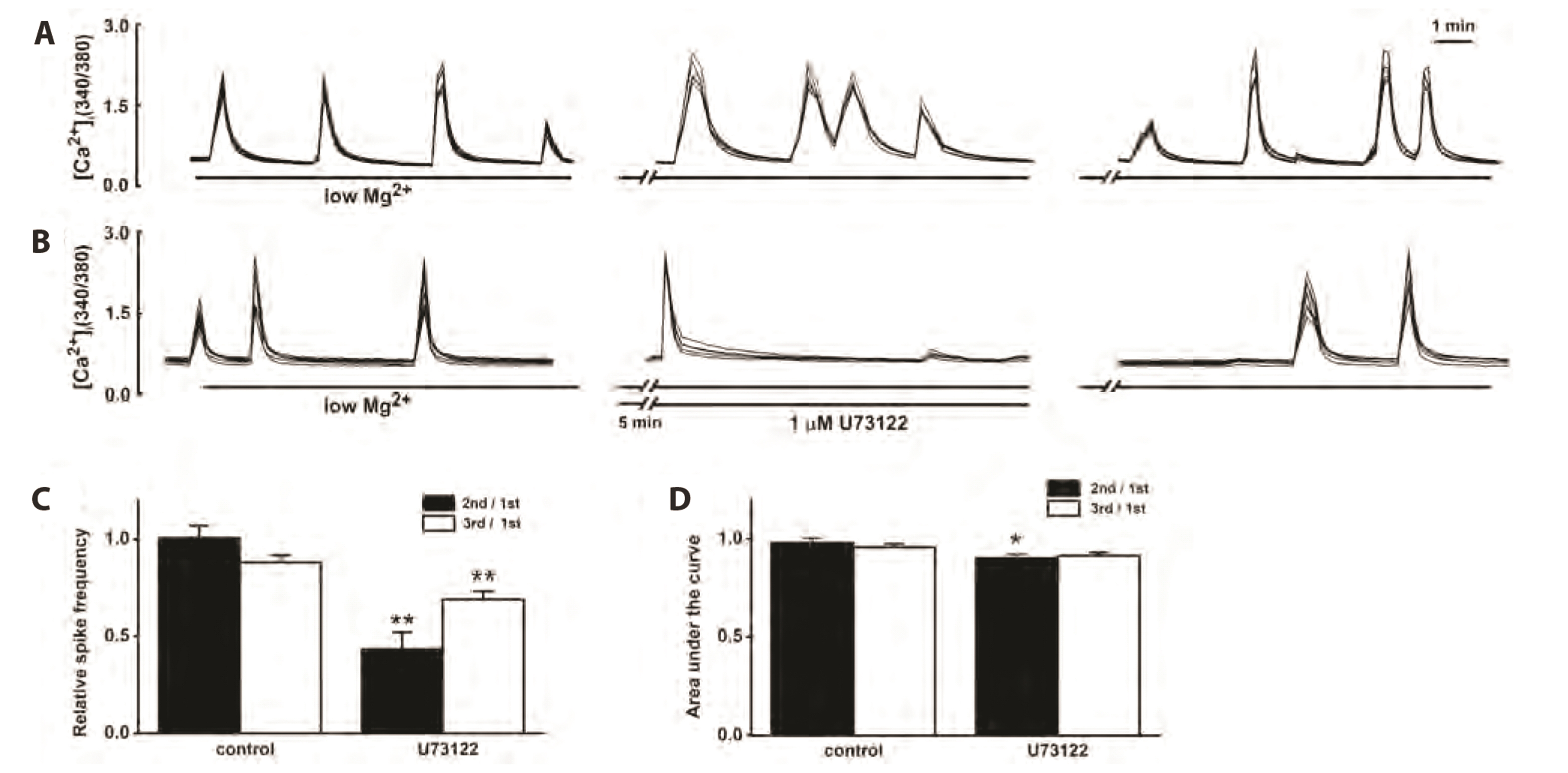
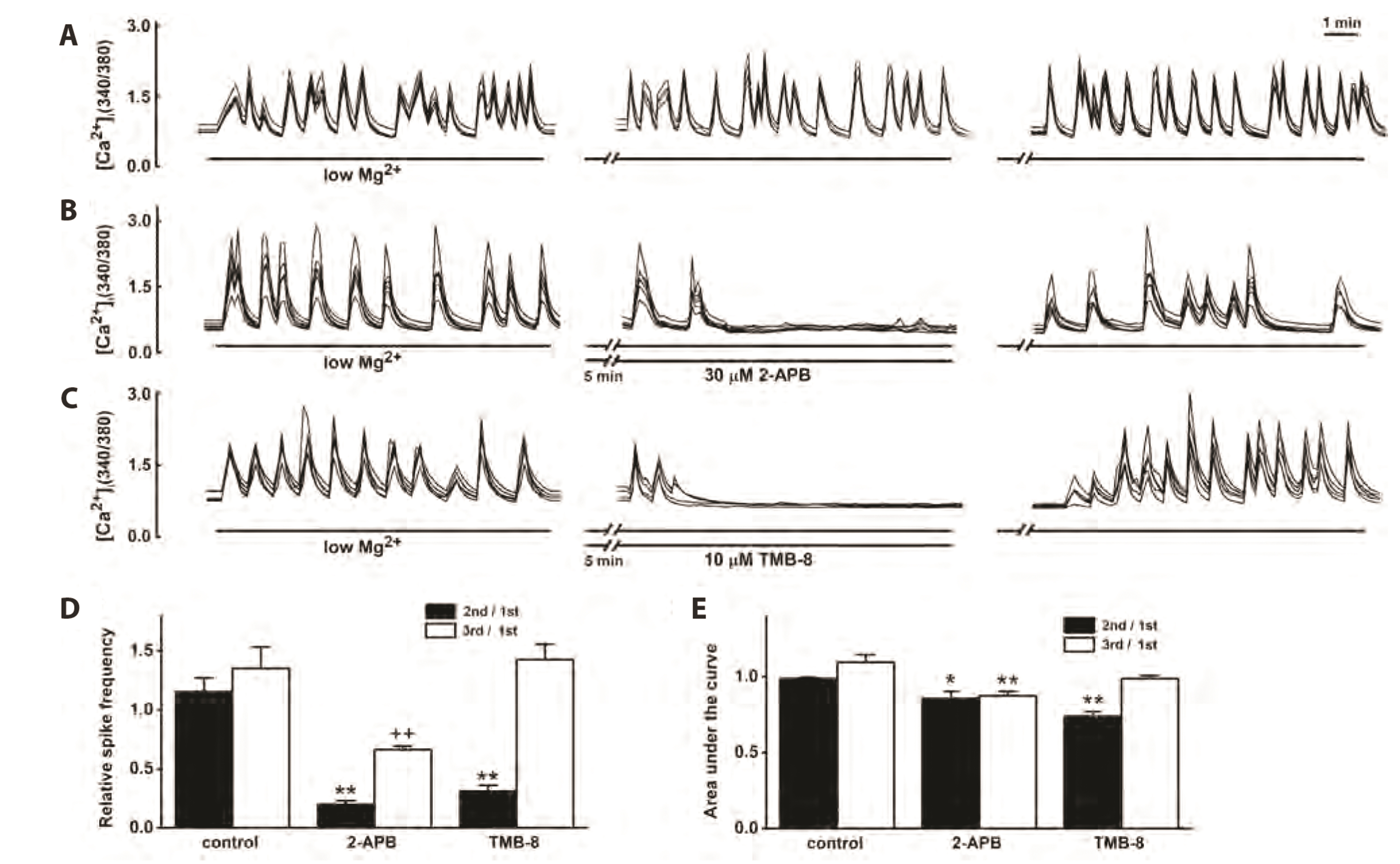
- Group 1 metabotropic glutamate receptors (mGluRs) can positively affect postsynaptic neuronal excitability and epileptogenesis. The objective of the present study was to determine whether group 1 mGluRs might be involved in synapticallyinduced intracellular free Ca2+ concentration ([Ca2+ ] i ) spikes and neuronal cell death induced by 0.1 mM Mg2+ and 10 µM glycine in cultured rat hippocampal neurons from embryonic day 17 fetal Sprague–Dawley rats using imaging methods for Ca2+ and 3-(4,5- dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assays for cell survival. Reduction of extracellular Mg2+ concentration ([Mg2+ ] o ) to 0.1 mM induced repetitive [Ca2+ ] i spikes within 30 sec at day 11.5. The mGluR5 antagonist 6-Methyl-2-(phenylethynyl) pyridine (MPEP) almost completely inhibited the [Ca2+ ] i spikes, but the mGluR1 antagonist LY367385 did not. The group 1 mGluRs agonist, 3,5-dihydroxyphenylglycine (DHPG), significantly increased the [Ca2+ ] i spikes. The phospholipase C inhibitor U73122 significantly inhibited the [Ca2+ ] i spikes in the absence or presence of DHPG. The IP3 receptor antagonist 2-aminoethoxydiphenyl borate or the ryanodine receptor antagonist 8-(diethylamino)octyl 3,4,5-trimethoxybenzoate also significantly inhibited the [Ca2+ ] i spikes in the absence or presence of DHPG. The TRPC channel inhibitors SKF96365 and flufenamic acid significantly inhibited the [Ca2+ ] i spikes in the absence or presence of DHPG. The mGluR5 antagonist MPEP significantly increased the neuronal cell survival, but mGluR1 antagonist LY367385 did not. These results suggest a possibility that mGluR5 is involved in synapticallyinduced [Ca2+ ] i spikes and neuronal cell death in cultured rat hippocampal neurons by releasing Ca2+ from IP3 and ryanodine-sensitive intracellular stores and activating TRPC channels.
Figure
Reference
-
1. Simpson PB, Challiss RA, Nahorski SR. 1995; Neuronal Ca2+ stores: activation and function. Trends Neurosci. 18:299–306. DOI: 10.1016/0166-2236(95)93919-O. PMID: 7571010. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0028979468&origin=inward.2. Abele AE, Scholz KP, Scholz WK, Miller RJ. 1990; Excitotoxicity induced by enhanced excitatory neurotransmission in cultured hippocampal pyramidal neurons. Neuron. 4:413–419. DOI: 10.1016/0896-6273(90)90053-I. PMID: 1690567. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0025218770&origin=inward.
Article3. Shen M, Piser TM, Seybold VS, Thayer SA. 1996; Cannabinoid receptor agonists inhibit glutamatergic synaptic transmission in rat hippocampal cultures. J Neurosci. 16:4322–4334. DOI: 10.1523/JNEUROSCI.16-14-04322.1996. PMID: 8699243. PMCID: PMC6578864. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0029937282&origin=inward.
Article4. Kim HJ, Yang JS, Yoon SH. 2016; Brief low [Mg2+]o-induced Ca2+ spikes inhibit subsequent prolonged exposure-induced excitotoxicity in cultured rat hippocampal neurons. Korean J Physiol Pharmacol. 20:101–109. DOI: 10.4196/kjpp.2016.20.1.101. PMID: 26807029. PMCID: PMC4722183. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84956938170&origin=inward.
Article5. McLeod JR Jr, Shen M, Kim DJ, Thayer SA. 1998; Neurotoxicity mediated by aberrant patterns of synaptic activity between rat hippocampal neurons in culture. J Neurophysiol. 80:2688–2698. DOI: 10.1152/jn.1998.80.5.2688. PMID: 9819273. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0031759749&origin=inward.
Article6. Kim HJ, Kim TH, Choi SJ, Hong YJ, Yang JS, Sung KW, Rhie DJ, Hahn SJ, Yoon SH. 2013; Fluoxetine suppresses synaptically induced [Ca2+]i spikes and excitotoxicity in cultured rat hippocampal neurons. Brain Res. 1490:23–34. DOI: 10.1016/j.brainres.2012.10.062. PMID: 23131584. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84871334105&origin=inward.
Article7. Mody I, Lambert JD, Heinemann U. 1987; Low extracellular magnesium induces epileptiform activity and spreading depression in rat hippocampal slices. J Neurophysiol. 57:869–888. DOI: 10.1152/jn.1987.57.3.869. PMID: 3031235. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0023149047&origin=inward.
Article8. Sombati S, Delorenzo RJ. 1995; Recurrent spontaneous seizure activity in hippocampal neuronal networks in culture. J Neurophysiol. 73:1706–1711. DOI: 10.1152/jn.1995.73.4.1706. PMID: 7643176. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0028898916&origin=inward.
Article9. Gibbs JW 3rd, Sombati S, DeLorenzo RJ, Coulter DA. 1997; Physiological and pharmacological alterations in postsynaptic GABAA receptor function in a hippocampal culture model of chronic spontaneous seizures. J Neurophysiol. 77:2139–2152. DOI: 10.1152/jn.1997.77.4.2139. PMID: 9114261. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0030949817&origin=inward.
Article10. Shigemoto R, Kinoshita A, Wada E, Nomura S, Ohishi H, Takada M, Flor PJ, Neki A, Abe T, Nakanishi S, Mizuno N. 1997; Differential presynaptic localization of metabotropic glutamate receptor subtypes in the rat hippocampus. J Neurosci. 17:7503–7522. DOI: 10.1523/JNEUROSCI.17-19-07503.1997. PMID: 9295396. PMCID: PMC6573434. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0030821474&origin=inward.
Article11. Pin JP, Duvoisin R. 1995; The metabotropic glutamate receptors: structure and functions. Neuropharmacology. 34:1–26. DOI: 10.1016/0028-3908(94)00129-G. PMID: 7623957. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0029187946&origin=inward.
Article12. Nicoletti F, Meek JL, Iadarola MJ, Chuang DM, Roth BL, Costa E. 1986; Coupling of inositol phospholipid metabolism with excitatory amino acid recognition sites in rat hippocampus. J Neurochem. 46:40–46. DOI: 10.1111/j.1471-4159.1986.tb12922.x. PMID: 2866236. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0022639103&origin=inward.
Article13. Sugiyama H, Ito I, Hirono C. 1987; A new type of glutamate receptor linked to inositol phospholipid metabolism. Nature. 325:531–533. DOI: 10.1038/325531a0. PMID: 2880300. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0023092256&origin=inward.
Article14. El-Hassar L, Hagenston AM, D'Angelo LB, Yeckel MF. 2011; Metabotropic glutamate receptors regulate hippocampal CA1 pyramidal neuron excitability via Ca²+ wave-dependent activation of SK and TRPC channels. J Physiol. 589(Pt 13):3211–3229. DOI: 10.1113/jphysiol.2011.209783. PMID: 21576272. PMCID: PMC3145935. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=79959742217&origin=inward.
Article15. Lujan R, Nusser Z, Roberts JD, Shigemoto R, Somogyi P. 1996; Perisynaptic location of metabotropic glutamate receptors mGluR1 and mGluR5 on dendrites and dendritic spines in the rat hippocampus. Eur J Neurosci. 8:1488–1500. DOI: 10.1111/j.1460-9568.1996.tb01611.x. PMID: 8758956. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0030014918&origin=inward.
Article16. Forsythe ID, Barnes-Davies M. 1997; Synaptic transmission: well-placed modulators. Curr Biol. 7:R362–R365. DOI: 10.1016/S0960-9822(06)00175-8. PMID: 9197230. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0031172242&origin=inward.
Article17. Yang JS, Perveen S, Ha TJ, Kim SY, Yoon SH. 2015; Cyanidin-3-glucoside inhibits glutamate-induced Zn2+ signaling and neuronal cell death in cultured rat hippocampal neurons by inhibiting Ca2+-induced mitochondrial depolarization and formation of reactive oxygen species. Brain Res. 1606:9–20. DOI: 10.1016/j.brainres.2015.02.028. PMID: 25721794. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84926178949&origin=inward.
Article18. Shim EY, Kim HJ, Kim MJ, Rhie DJ, Jo YH, Kim MS, Hahn JS, Lee MY, Yoon SH. 2006; Desensitization of somatostatin-induced inhibition of low extracellular magnesium concentration-induced calcium spikes in cultured rat hippocampal neurons. Brain Res. 1111:61–71. DOI: 10.1016/j.brainres.2006.06.081. PMID: 16879804. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=33748335799&origin=inward.
Article19. Lévêque M, Penna A, Le Trionnaire S, Belleguic C, Desrues B, Brinchault G, Jouneau S, Lagadic-Gossmann D, Martin-Chouly C. 2018; Phagocytosis depends on TRPV2-mediated calcium influx and requires TRPV2 in lipids rafts: alteration in macrophages from patients with cystic fibrosis. Sci Rep. 8:4310. DOI: 10.1038/s41598-018-22558-5. PMID: 29523858. PMCID: PMC5844937. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85043993308&origin=inward.
Article20. Martin LJ, Blackstone CD, Huganir RL, Price DL. 1992; Cellular localization of a metabotropic glutamate receptor in rat brain. Neuron. 9:259–270. DOI: 10.1016/0896-6273(92)90165-A. PMID: 1323311. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0026794813&origin=inward.
Article21. Shigemoto R, Nomura S, Ohishi H, Sugihara H, Nakanishi S, Mizuno N. 1993; Immunohistochemical localization of a metabotropic glutamate receptor, mGluR5, in the rat brain. Neurosci Lett. 163:53–57. DOI: 10.1016/0304-3940(93)90227-C. PMID: 8295733. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0027378595&origin=inward.
Article22. Gasparini F, Lingenhöhl K, Stoehr N, Flor PJ, Heinrich M, Vranesic I, Biollaz M, Allgeier H, Heckendorn R, Urwyler S, Varney MA, Johnson EC, Hess SD, Rao SP, Sacaan AI, Santori EM, Veliçelebi G, Kuhn R. 1999; 2-Methyl-6-(phenylethynyl)-pyridine (MPEP), a potent, selective and systemically active mGlu5 receptor antagonist. Neuropharmacology. 38:1493–1503. DOI: 10.1016/S0028-3908(99)00082-9. PMID: 10530811. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0032853255&origin=inward.
Article23. Bruno V, Battaglia G, Kingston A, O'Neill MJ, Catania MV, Di Grezia R, Nicoletti F. 1999; Neuroprotective activity of the potent and selective mGlu1a metabotropic glutamate receptor antagonist, (+)-2-methyl-4 carboxyphenylglycine (LY367385): comparison with LY357366, a broader spectrum antagonist with equal affinity for mGlu1a and mGlu5 receptors. Neuropharmacology. 38:199–207. DOI: 10.1016/S0028-3908(98)00159-2. PMID: 10218860. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0033046392&origin=inward.
Article24. Schoepp DD, Goldsworthy J, Johnson BG, Salhoff CR, Baker SR. 1994; 3,5-dihydroxyphenylglycine is a highly selective agonist for phosphoinositide-linked metabotropic glutamate receptors in the rat hippocampus. J Neurochem. 63:769–772. DOI: 10.1046/j.1471-4159.1994.63020769.x. PMID: 8035201. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0027934857&origin=inward.
Article25. Gee CE, Benquet P, Gerber U. 2003; Group I metabotropic glutamate receptors activate a calcium-sensitive transient receptor potential-like conductance in rat hippocampus. J Physiol. 546(Pt 3):655–664. DOI: 10.1113/jphysiol.2002.032961. PMID: 12562994. PMCID: PMC2342598. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0037320710&origin=inward.
Article26. Phelan KD, Shwe UT, Abramowitz J, Wu H, Rhee SW, Howell MD, Gottschall PE, Freichel M, Flockerzi V, Birnbaumer L, Zheng F. 2013; Canonical transient receptor channel 5 (TRPC5) and TRPC1/4 contribute to seizure and excitotoxicity by distinct cellular mechanisms. Mol Pharmacol. 83:429–438. DOI: 10.1124/mol.112.082271. PMID: 23188715. PMCID: PMC3558807. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84872708983&origin=inward.
Article27. Narayanan KL, Subramaniam S, Bengston CP, Irmady K, Unsicker K, von Bohlen und Halbach O. 2014; Role of transient receptor potential channel 1 (TRPC1) in glutamate-induced cell death in the hippocampal cell line HT22. J Mol Neurosci. 52:425–433. DOI: 10.1007/s12031-013-0171-9. PMID: 24242951. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84894346169&origin=inward.
Article28. Lepannetier S, Gualdani R, Tempesta S, Schakman O, Seghers F, Kreis A, Yerna X, Slimi A, de Clippele M, Tajeddine N, Voets T, Bon RS, Beech DJ, Tissir F, Gailly P. 2018; Activation of TRPC1 channel by metabotropic glutamate receptor mGluR5 modulates synaptic plasticity and spatial working memory. Front Cell Neurosci. 12:318. DOI: 10.3389/fncel.2018.00318. PMID: 30271326. PMCID: PMC6149316. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85054827276&origin=inward.
Article29. Yerna X, Schakman O, Ratbi I, Kreis A, Lepannetier S, de Clippele M, Achouri Y, Tajeddine N, Tissir F, Gualdani R, Gailly P. 2020; Role of the TRPC1 channel in hippocampal long-term depression and in spatial memory extinction. Int J Mol Sci. 21:1712. DOI: 10.3390/ijms21051712. PMID: 32138218. PMCID: PMC7084652. PMID: 8e14f08f2515411aa3999468d10f5162. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85081003715&origin=inward.
Article30. Strübing C, Krapivinsky G, Krapivinsky L, Clapham DE. 2001; TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron. 29:645–655. DOI: 10.1016/S0896-6273(01)00240-9. PMID: 11301024. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0035051978&origin=inward.
Article31. Fowler MA, Sidiropoulou K, Ozkan ED, Phillips CW, Cooper DC. 2007; Corticolimbic expression of TRPC4 and TRPC5 channels in the rodent brain. PLoS One. 2:e573. DOI: 10.1371/journal.pone.0000573. PMID: 17593972. PMCID: PMC1892805. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=39349091413&origin=inward.
Article32. Chung YH, Ahn HS, Kim D, Shin DH, Kim SS, Kim KY, Lee WB, Cha CI. 2006; Immunohistochemical study on the distribution of TRPC channels in the rat hippocampus. Brain Res. 1085:132–137. DOI: 10.1016/j.brainres.2006.02.087. PMID: 16580647. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=33646581699&origin=inward.
Article33. Deshpande LS, Lou JK, Mian A, Blair RE, Sombati S, Attkisson E, DeLorenzo RJ. 2008; Time course and mechanism of hippocampal neuronal death in an in vitro model of status epilepticus: role of NMDA receptor activation and NMDA dependent calcium entry. Eur J Pharmacol. 583:73–83. DOI: 10.1016/j.ejphar.2008.01.025. PMID: 18289526. PMCID: PMC2323609. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=39649085329&origin=inward.
Article34. Abe T, Sugihara H, Nawa H, Shigemoto R, Mizuno N, Nakanishi S. 1992; Molecular characterization of a novel metabotropic glutamate receptor mGluR5 coupled to inositol phosphate/Ca2+ signal transduction. J Biol Chem. 267:13361–13368. DOI: 10.1016/S0021-9258(18)42219-3. PMID: 1320017.
Article35. Shigemoto R, Nakanishi S, Mizuno N. 1992; Distribution of the mRNA for a metabotropic glutamate receptor (mGluR1) in the central nervous system: an in situ hybridization study in adult and developing rat. J Comp Neurol. 322:121–135. DOI: 10.1002/cne.903220110. PMID: 1430307. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0026716350&origin=inward.
Article36. Reboreda A, Theissen FM, Valero-Aracama MJ, Arboit A, Corbu MA, Yoshida M. 2018; Do TRPC channels support working memory? Comparing modulations of TRPC channels and working memory through G-protein coupled receptors and neuromodulators. Behav Brain Res. 354:64–83. DOI: 10.1016/j.bbr.2018.02.042. PMID: 29501506. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85045021580&origin=inward.
Article37. Gualdani R, Gailly P. 2020; How TRPC channels modulate hippocampal function. Int J Mol Sci. 21:3915. DOI: 10.3390/ijms21113915. PMID: 32486187. PMCID: PMC7312571. PMID: c4f79047dd0f45f2afbcf20a8fe0dd69. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85085909373&origin=inward.38. Shen M, Thayer SA. 1998; Cannabinoid receptor agonists protect cultured rat hippocampal neurons from excitotoxicity. Mol Pharmacol. 54:459–462. DOI: 10.1124/mol.54.3.459. PMID: 9730904. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0031706164&origin=inward.
Article39. Narayanan KL, Irmady K, Subramaniam S, Unsicker K, von Bohlen und Halbach O. 2008; Evidence that TRPC1 is involved in hippocampal glutamate-induced cell death. Neurosci Lett. 446:117–122. DOI: 10.1016/j.neulet.2008.09.034. PMID: 18822346. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=53849092028&origin=inward.
Article40. Phelan KD, Mock MM, Kretz O, Shwe UT, Kozhemyakin M, Greenfield LJ, Dietrich A, Birnbaumer L, Freichel M, Flockerzi V, Zheng F. 2012; Heteromeric canonical transient receptor potential 1 and 4 channels play a critical role in epileptiform burst firing and seizure-induced neurodegeneration. Mol Pharmacol. 81:384–392. DOI: 10.1124/mol.111.075341. PMID: 22144671. PMCID: PMC3286301. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84857401210&origin=inward.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Apigenin on Glutamate-induced [Ca2+]i Increases in Cultured Rat Hippocampal Neurons
- Roles of metabotropic glutamate receptor 5 in low [Mg2+ ]o -induced interictal epileptiform activity in rat hippocampal slices
- Roles of Metabotropic Glutamate Receptors in Inhibitory Synaptic Transmission in Rat Medial Vestibular Nucleus Neurons
- Cyanidin-3-glucoside inhibits amyloid β₂₅₋₃₅-induced neuronal cell death in cultured rat hippocampal neurons
- Differential effect of homocysteic acid and cysteic acid on changes of inositol phosphates and (Ca2+)i in rat cerebellar granule cells