Int J Stem Cells.
2021 Aug;14(3):286-297. 10.15283/ijsc20188.
The Role of miR-34c-5p in Osteogenic Differentiation of Bone Marrow Mesenchymal Stem Cells
- Affiliations
-
- 1Department of Spine Surgery, The Sixth Affiliated Hospital of Wenzhou Medical University, Lishui People’s Hospital, Lishui, China
- 2Pharmacy College, Wenzhou Medical University, Wenzhou, China
- 3Department of Pharmacy, The Sixth Affiliated Hospital of Wenzhou Medical University, Lishui People’s Hospital, Lishui, China
- KMID: 2519371
- DOI: http://doi.org/10.15283/ijsc20188
Abstract
- Background and Objectives
Osteogenic differentiation of bone marrow mesenchymal stem cells (BMSCs) plays a critical role in the success of lumbar spinal fusion with autogenous bone graft. This study aims to explore the role and specific mechanism of miR-34c-5p in osteogenic differentiation of BMSCs.
Methods and Results
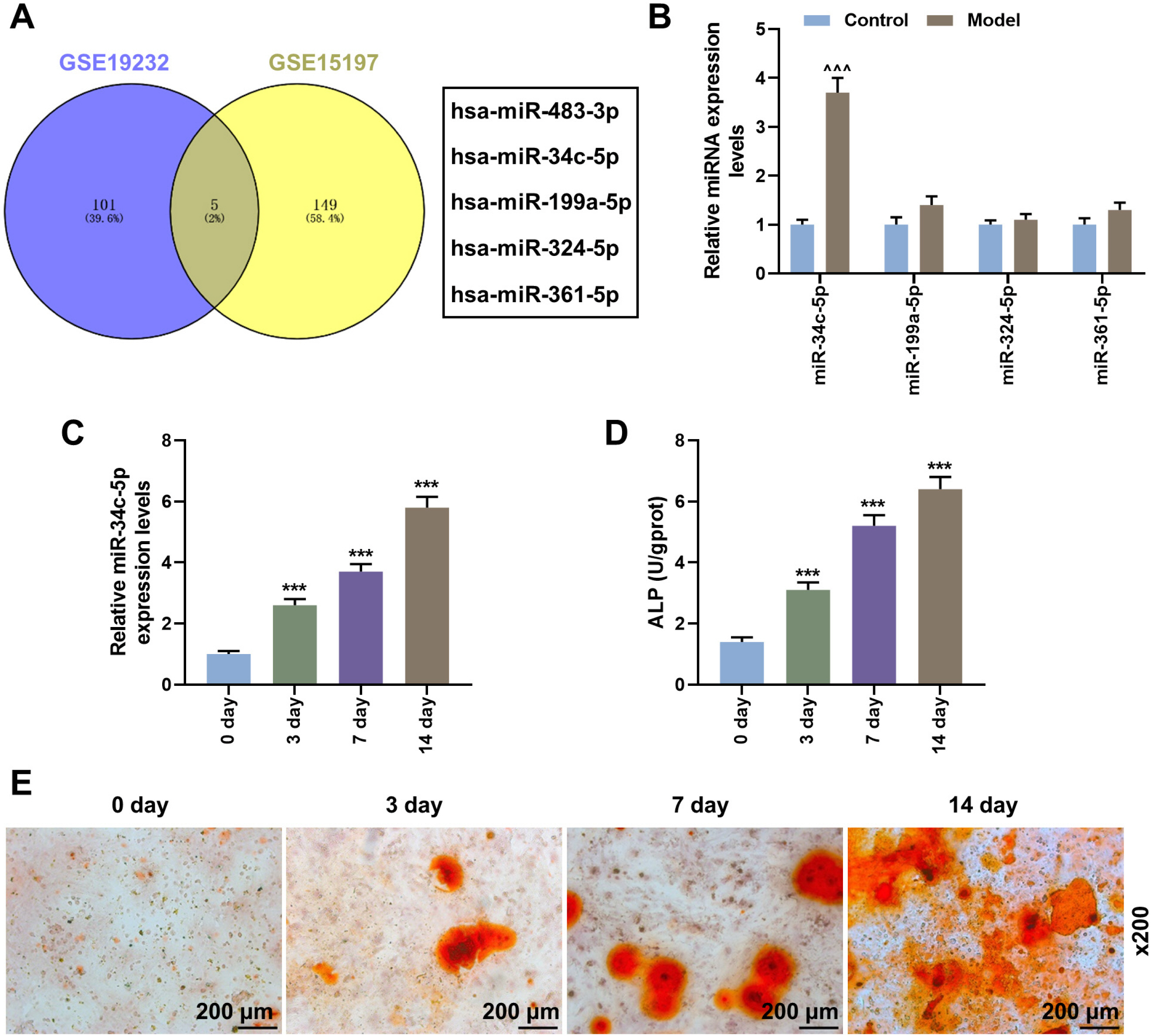
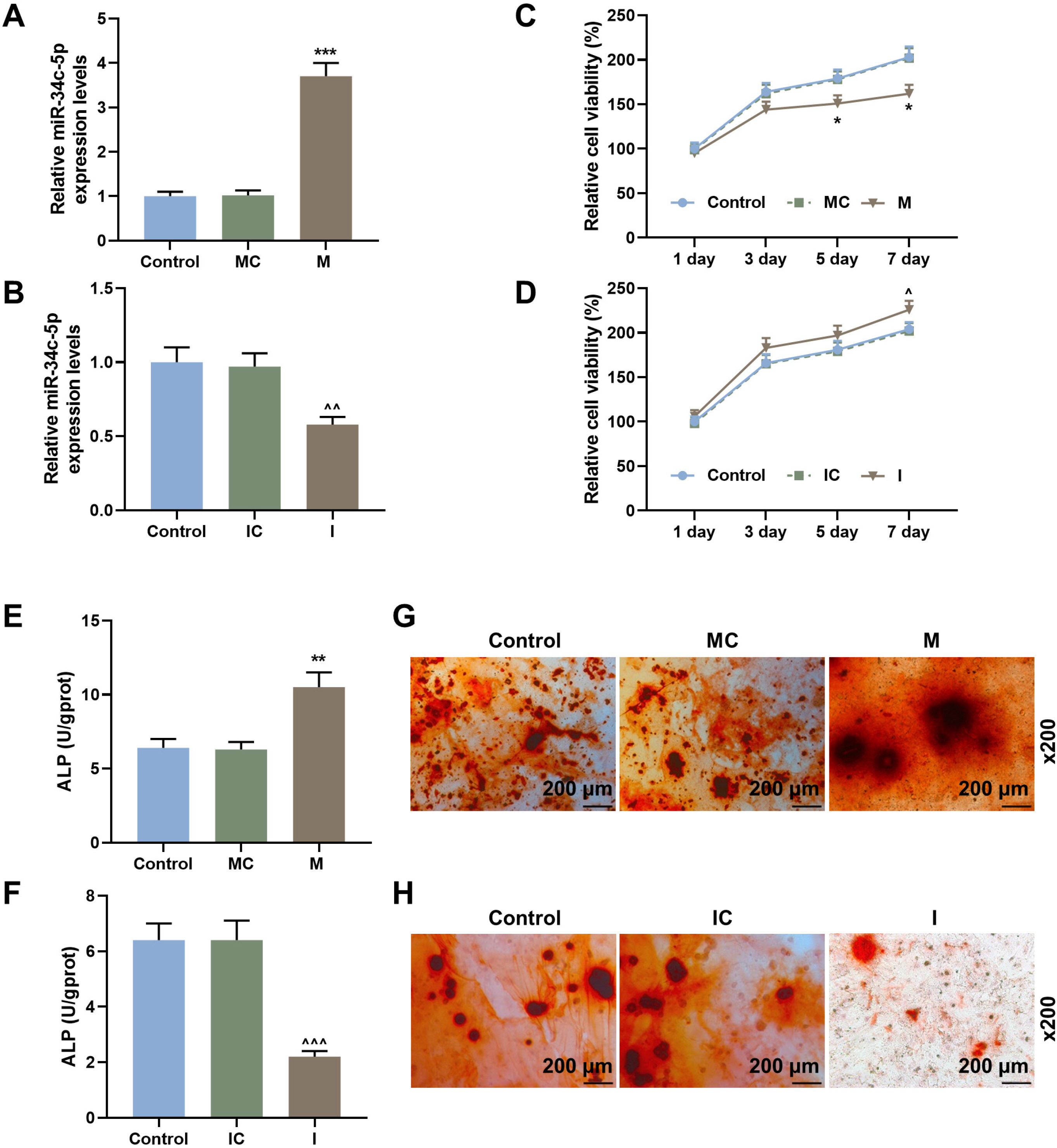
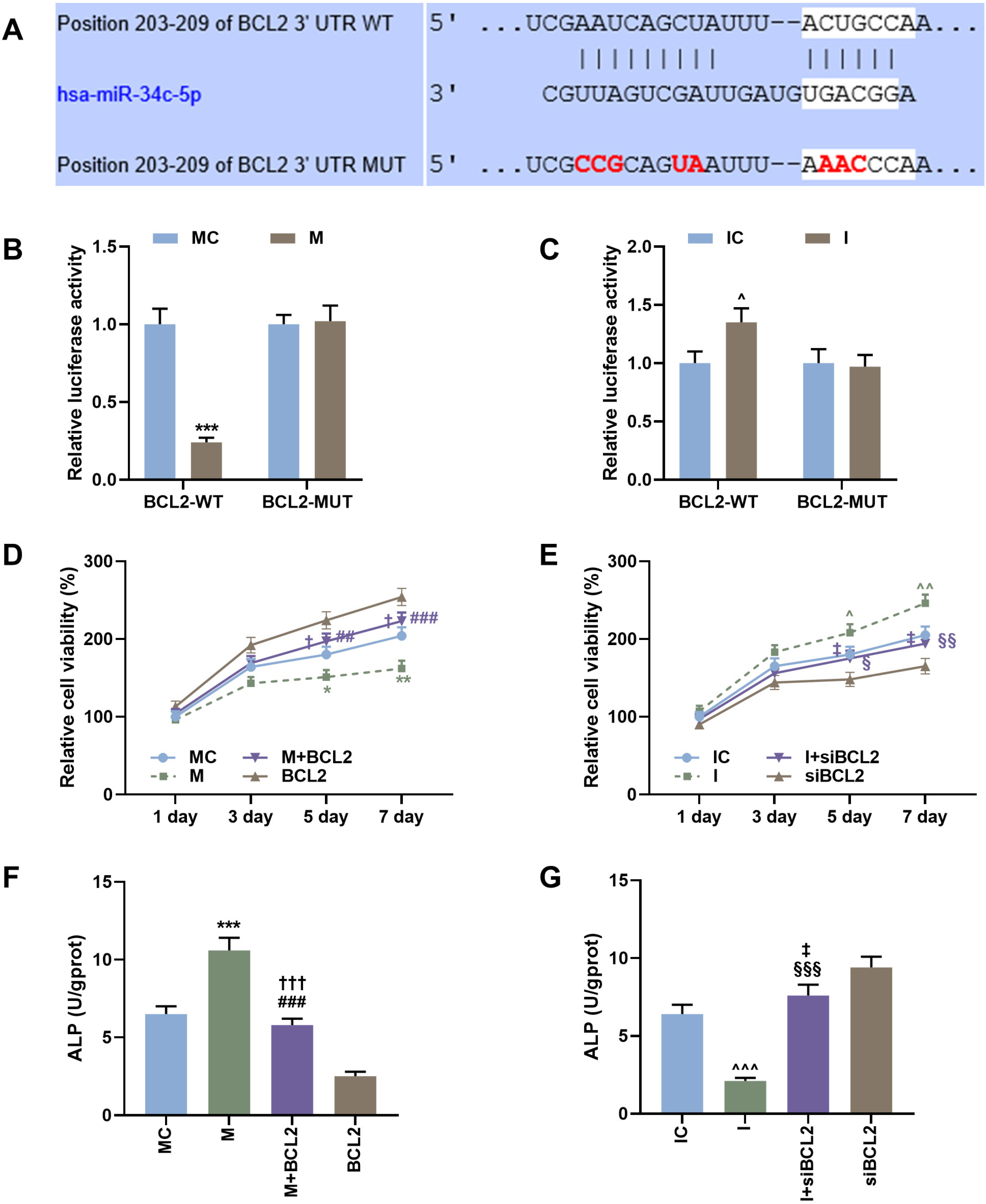
Rabbit model of lumbar fusion was established by surgery. The osteogenic differentiation dataset of mesenchymal stem cells was obtained from the Gene Expression Omnibus (GEO) database, and differentially expressed miRNAs were analyzed using R language (limma package). The expressions of miR-34c-5p, miR-199a-5p, miR-324-5p, miR-361-5p, RUNX2, OCN and Bcl-2 were determined by qRT-PCR and Western blot. ELISA, Alizarin red staining and CCK-8 were used to detect the ALP content, calcium deposition and proliferation of BMSCs. The targeted binding sites between miR-34c-5p and Bcl-2 were predicted by the Target database and verified using dual-luciferase reporter assay. MiR-34c-5p expression was higher in rabbit lumbar fusion model and differentiated BMSCs than normal rabbit or BMSCs. The content of ALP and the deposition of calcium increased with the osteogenic differentiation of BMSCs. Upregulation of miR-34c-5p reduced cell proliferation and promoted ALP content, calcium deposition, RUNX2 and OCN expression compared with the control group. The effects of miR-34c-5p inhibitor were the opposite. In addition, miR-34c-5p negatively correlated with Bcl-2. Upregulation of Bcl-2 reversed the effects of miR-34c-5p on ALP content, calcium deposition, and the expressions of RUNX2 and OCN.
Conclusions
miR-34c-5p could promote osteogenic differentiation and suppress proliferation of BMSCs by inhibiting Bcl-2.
Keyword
Figure
Reference
-
References
1. Schnake KJ, Rappert D, Storzer B, Schreyer S, Hilber F, Mehren C. 2019; Lumbar fusion-Indications and techniques. Orthopade. 48:50–58. German. DOI: 10.1007/s00132-018-03670-w. PMID: 30552449.2. Chavda S, Levin L. 2018; Human studies of vertical and horizontal alveolar ridge augmentation comparing different types of bone graft materials: a systematic review. J Oral Implantol. 44:74–84. DOI: 10.1563/aaid-joi-D-17-00053. PMID: 29135351.
Article3. Jordana F, Le Visage C, Weiss P. 2017; Bone substitutes. Med Sci (Paris). 33:60–65. French. DOI: 10.1051/medsci/20173301010. PMID: 28120757.4. Manolagas SC. 2000; Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr Rev. 21:115–137. DOI: 10.1210/edrv.21.2.0395. PMID: 10782361.
Article5. Park D, Spencer JA, Koh BI, Kobayashi T, Fujisaki J, Clemens TL, Lin CP, Kronenberg HM, Scadden DT. 2012; Endogenous bone marrow MSCs are dynamic, fate-restricted participants in bone maintenance and regeneration. Cell Stem Cell. 10:259–272. DOI: 10.1016/j.stem.2012.02.003. PMID: 22385654. PMCID: PMC3652251.
Article6. Gómez-Puerto MC, Verhagen LP, Braat AK, Lam EW, Coffer PJ, Lorenowicz MJ. 2016; Activation of autophagy by FOXO3 regulates redox homeostasis during osteogenic differentiation. Autophagy. 12:1804–1816. DOI: 10.1080/15548627.2016.1203484. PMID: 27532863. PMCID: PMC5079670.
Article7. Wang C, Meng H, Wang X, Zhao C, Peng J, Wang Y. 2016; Differentiation of bone marrow mesenchymal stem cells in osteoblasts and adipocytes and its role in treatment of osteoporosis. Med Sci Monit. 22:226–233. DOI: 10.12659/MSM.897044. PMID: 26795027. PMCID: PMC4727494.
Article8. Tomé M, López-Romero P, Albo C, Sepúlveda JC, Fernández-Gutiérrez B, Dopazo A, Bernad A, González MA. 2011; miR-335 orchestrates cell proliferation, migration and differentiation in human mesenchymal stem cells. Cell Death Differ. 18:985–995. DOI: 10.1038/cdd.2010.167. PMID: 21164520. PMCID: PMC3131940.
Article9. Krol J, Loedige I, Filipowicz W. 2010; The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet. 11:597–610. DOI: 10.1038/nrg2843. PMID: 20661255.
Article10. Oskowitz AZ, Lu J, Penfornis P, Ylostalo J, McBride J, Flemington EK, Prockop DJ, Pochampally R. 2008; Human multipotent stromal cells from bone marrow and microRNA: regulation of differentiation and leukemia inhibitory factor expression. Proc Natl Acad Sci U S A. 105:18372–18377. DOI: 10.1073/pnas.0809807105. PMID: 19011087. PMCID: PMC2587615.
Article11. Wang B, Yu P, Li T, Bian Y, Weng X. 2015; MicroRNA expression in bone marrow mesenchymal stem cells from mice with steroid-induced osteonecrosis of the femoral head. Mol Med Rep. 12:7447–7454. DOI: 10.3892/mmr.2015.4386. PMID: 26459755.
Article12. Tamura M, Uyama M, Sugiyama Y, Sato M. 2013; Canonical Wnt signaling activates miR-34 expression during osteoblastic differentiation. Mol Med Rep. 8:1807–1811. DOI: 10.3892/mmr.2013.1713. PMID: 24100761.
Article13. Liu Y, Xu F, Pei HX, Zhu X, Lin X, Song CY, Liang QH, Liao EY, Yuan LQ. 2016; Vaspin regulates the osteogenic differentiation of MC3T3-E1 through the PI3K-Akt/miR-34c loop. Sci Rep. 6:25578. DOI: 10.1038/srep25578. PMID: 27156573. PMCID: PMC4860647.
Article14. Yang X, Yang J, Lei P, Wen T. 2019; LncRNA MALAT1 shuttled by bone marrow-derived mesenchymal stem cells-secreted exosomes alleviates osteoporosis through mediating microRNA-34c/SATB2 axis. Aging (Albany NY). 11:8777–8791. DOI: 10.18632/aging.102264. PMID: 31659145. PMCID: PMC6834402.
Article15. Hagman Z, Larne O, Edsjö A, Bjartell A, Ehrnström RA, Ulmert D, Lilja H, Ceder Y. 2010; miR-34c is downregulated in prostate cancer and exerts tumor suppressive functions. Int J Cancer. 127:2768–2776. DOI: 10.1002/ijc.25269. PMID: 21351256.
Article16. Ebrahim AS, Sabbagh H, Liddane A, Raufi A, Kandouz M, Al-Katib A. 2016; Hematologic malignancies: newer strategies to counter the BCL-2 protein. J Cancer Res Clin Oncol. 142:2013–2022. DOI: 10.1007/s00432-016-2144-1. PMID: 27043233.
Article17. Heng BC, Ye X, Liu Y, Dissanayaka WL, Cheung GS, Zhang C. 2016; Effects of recombinant overexpression of Bcl2 on the proliferation, apoptosis, and osteogenic/odontogenic differentiation potential of dental pulp stem cells. J Endod. 42:575–583. DOI: 10.1016/j.joen.2016.01.013. PMID: 26898562.
Article18. Yuan B, Yu WY, Dai LS, Gao Y, Ding Y, Yu XF, Chen J, Zhang JB. 2015; Expression of microRNA-26b and identification of its target gene EphA2 in pituitary tissues in Yanbian cattle. Mol Med Rep. 12:5753–5761. DOI: 10.3892/mmr.2015.4192. PMID: 26252447. PMCID: PMC4581756.
Article19. Liu S, Xie X, Lei H, Zou B, Xie L. 2019; Identification of Key circRNAs/lncRNAs/miRNAs/mRNAs and pathways in preeclampsia using bioinformatics analysis. Med Sci Monit. 25:1679–1693. DOI: 10.12659/MSM.912801. PMID: 30833538. PMCID: PMC6413561.
Article20. Berezikov E, Guryev V, van de Belt J, Wienholds E, Plasterk RH, Cuppen E. 2005; Phylogenetic shadowing and computational identification of human microRNA genes. Cell. 120:21–24. DOI: 10.1016/j.cell.2004.12.031. PMID: 15652478.
Article21. Lu TX, Rothenberg ME. 2018; MicroRNA. J Allergy Clin Immunol. 141:1202–1207. DOI: 10.1016/j.jaci.2017.08.034. PMID: 29074454. PMCID: PMC5889965.
Article22. Fu YC, Zhao SR, Zhu BH, Guo SS, Wang XX. 2019; MiRNA-27a-3p promotes osteogenic differentiation of human mesenchymal stem cells through targeting ATF3. Eur Rev Med Pharmacol Sci. 23(3 Suppl):73–80. DOI: 10.26355/eurrev_201908_18632. PMID: 31389577.23. Cheng F, Yang MM, Yang RH. 2019; MiRNA-365a-3p promotes the progression of osteoporosis by inhibiting osteogenic differentiation via targeting RUNX2. Eur Rev Med Pharmacol Sci. 23:7766–7774. DOI: 10.26355/eurrev_201909_18986. PMID: 31599402.24. Chen R, Qiu H, Tong Y, Liao F, Hu X, Qiu Y, Liao Y. 2019; MiRNA-19a-3p alleviates the progression of osteoporosis by targeting HDAC4 to promote the osteogenic differentiation of hMSCs. Biochem Biophys Res Commun. 516:666–672. DOI: 10.1016/j.bbrc.2019.06.083. PMID: 31248594.
Article25. Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP. 1997; Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem. 64:295–312. DOI: 10.1002/(SICI)1097-4644(199702)64:2<295::AID-JCB12>3.0.CO;2-I. PMID: 9027589.
Article26. Alborzi A, Mac K, Glackin CA, Murray SS, Zernik JH. 1996; Endochondral and intramembranous fetal bone development: osteoblastic cell proliferation, and expression of alkaline phosphatase, m-twist, and histone H4. J Craniofac Genet Dev Biol. 16:94–106. PMID: 8773900.27. Liu TM, Lee EH. 2013; Transcriptional regulatory cascades in Runx2-dependent bone development. Tissue Eng Part B Rev. 19:254–263. DOI: 10.1089/ten.teb.2012.0527. PMID: 23150948. PMCID: PMC3627420.
Article28. Jeong JH, Choi JY. 2011; Interrelationship of Runx2 and estrogen pathway in skeletal tissues. BMB Rep. 44:613–618. DOI: 10.5483/BMBRep.2011.44.10.613. PMID: 22026994.
Article29. Zhang Y, Xie RL, Croce CM, Stein JL, Lian JB, van Wijnen AJ, Stein GS. 2011; A program of microRNAs controls osteogenic lineage progression by targeting transcription factor Runx2. Proc Natl Acad Sci U S A. 108:9863–9868. DOI: 10.1073/pnas.1018493108. PMID: 21628588. PMCID: PMC3116419.
Article30. Zhang Y, Xie RL, Gordon J, LeBlanc K, Stein JL, Lian JB, van Wijnen AJ, Stein GS. 2012; Control of mesenchymal lineage progression by microRNAs targeting skeletal gene regulators Trps1 and Runx2. J Biol Chem. 287:21926–21935. DOI: 10.1074/jbc.M112.340398. PMID: 22544738. PMCID: PMC3381153.
Article31. Hou Z, Wang Z, Tao Y, Bai J, Yu B, Shen J, Sun H, Xiao L, Xu Y, Zhou J, Wang Z, Geng D. 2019; KLF2 regulates osteoblast differentiation by targeting of Runx2. Lab Invest. 99:271–280. DOI: 10.1038/s41374-018-0149-x. PMID: 30429507.
Article32. Ma J, Wang Z, Zhao J, Miao W, Ye T, Chen A. 2018; Resveratrol attenuates lipopolysaccharides (LPS)-induced inhibition of osteoblast differentiation in MC3T3-E1 cells. Med Sci Monit. 24:2045–2052. DOI: 10.12659/MSM.905703. PMID: 29624568. PMCID: PMC5903312.
Article33. Hu Y, Yang Q, Wang L, Wang S, Sun F, Xu D, Jiang J. 2018; Knockdown of the oncogene lncRNA NEAT1 restores the availability of miR-34c and improves the sensitivity to cisplatin in osteosarcoma. Biosci Rep. 38:BSR20180375. DOI: 10.1042/BSR20180375. PMID: 29654165. PMCID: PMC6435545.
Article34. Liu WM, Pang RT, Chiu PC, Wong BP, Lao K, Lee KF, Yeung WS. 2012; Sperm-borne microRNA-34c is required for the first cleavage division in mouse. Proc Natl Acad Sci U S A. 109:490–494. DOI: 10.1073/pnas.1110368109. PMID: 22203953. PMCID: PMC3258645.
Article35. Dietrich JB. 1997; Apoptosis and anti-apoptosis genes in the Bcl-2 family. Arch Physiol Biochem. 105:125–135. French. DOI: 10.1076/apab.105.2.125.12927. PMID: 9296841.36. Piro LD. 2004; Apoptosis, Bcl-2 antisense, and cancer therapy. Oncology (Williston Park). 18(13 Suppl 10):5–10. PMID: 15651171.37. Bruckheimer EM, Cho SH, Sarkiss M, Herrmann J, McDonnell TJ. 1998; The Bcl-2 gene family and apoptosis. Adv Biochem Eng Biotechnol. 62:75–105. DOI: 10.1007/BFb0102306. PMID: 9755641.
Article38. Chen G, Li P, Liu Z, Zeng R, Ma X, Chen Y, Xu H, Li Z, Lin H. 2019; Enrichment of miR-126 enhances the effects of endothelial progenitor cell-derived microvesicles on modulating MC3T3-E1 cell function via Erk1/2-Bcl-2 signalling pathway. Prion. 13:106–115. DOI: 10.1080/19336896.2019.1607464. PMID: 31050590. PMCID: PMC7000145.
Article39. Mocetti P, Silvestrini G, Ballanti P, Patacchioli FR, Di Grezia R, Angelucci L, Bonucci E. 2001; Bcl-2 and Bax expression in cartilage and bone cells after high-dose corticosterone treatment in rats. Tissue Cell. 33:1–7. DOI: 10.1054/tice.2000.0144. PMID: 11292165.
Article40. Boot-Handford RP, Michaelidis TM, Hillarby MC, Zambelli A, Denton J, Hoyland JA, Freemont AJ, Grant ME, Wallis GA. 1998; The bcl-2 knockout mouse exhibits marked changes in osteoblast phenotype and collagen deposition in bone as well as a mild growth plate phenotype. Int J Exp Pathol. 79:329–335. DOI: 10.1046/j.1365-2613.1998.790411.x. PMID: 10193316. PMCID: PMC3220196.
Article41. Zhang W, Pantschenko AG, McCarthy MB, Gronowicz G. 2007; Bone-targeted overexpression of Bcl-2 increases osteoblast adhesion and differentiation and inhibits mineralization in vitro. Calcif Tissue Int. 80:111–122. DOI: 10.1007/s00223-006-0168-2. PMID: 17308993.
Article42. Kang J, Sun Y, Deng Y, Liu Q, Li D, Liu Y, Guan X, Tao Z, Wang X. 2020; Autophagy-endoplasmic reticulum stress inhibition mechanism of superoxide dismutase in the formation of calcium oxalate kidney stones. Biomed Pharmacother. 121:109649. DOI: 10.1016/j.biopha.2019.109649. PMID: 31733571.
Article43. Nagase Y, Iwasawa M, Akiyama T, Kadono Y, Nakamura M, Oshima Y, Yasui T, Matsumoto T, Hirose J, Nakamura H, Miyamoto T, Bouillet P, Nakamura K, Tanaka S. 2009; Anti-apoptotic molecule Bcl-2 regulates the differentiation, activation, and survival of both osteoblasts and osteoclasts. J Biol Chem. 284:36659–36669. DOI: 10.1074/jbc.M109.016915. PMID: 19846553. PMCID: PMC2794780.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Exploring upregulated genes during osteogenic differentiation of hMSCs
- MiR-182-5p Mediated by Exosomes Derived From Bone Marrow Mesenchymal Stem Cell Attenuates Inflammatory Responses by Targeting TLR4 in a Mouse Model of Myocardial Infraction
- MicroRNA-181a-5p Curbs Osteogenic Differentiation and Bone Formation Partially Through Impairing Runx1-Dependent Inhibition of AIF-1 Transcription
- Kartogenin Promotes the BMSCs Chondrogenic Differentiation in Osteoarthritis by Down-Regulation of miR-145-5p Targeting Smad4 Pathway
- MiR-29a-3p Inhibits Proliferation and Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells via Targeting FOXO3 and Repressing Wnt/β-Catenin Signaling in Steroid-Associated Osteonecrosis