Korean J Physiol Pharmacol.
2021 May;25(3):217-225. 10.4196/kjpp.2021.25.3.217.
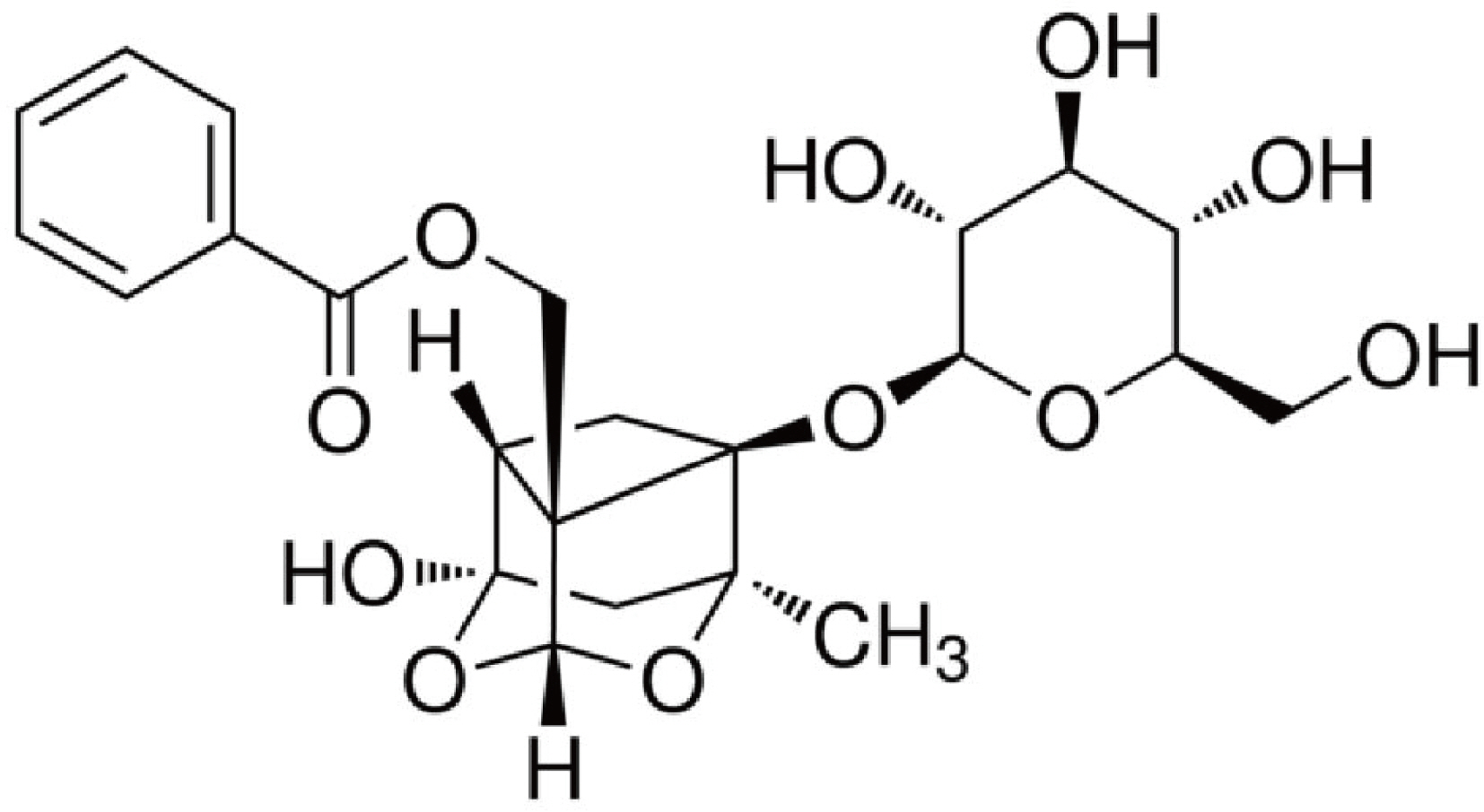
Paeoniflorin ameliorates neuropathic pain-induced depression-like behaviors in mice by inhibiting hippocampal neuroinflammation activated via TLR4/NF-kB pathway
- Affiliations
-
- 1Center of Morphological Experiment, Medical College of Yanbian University, China
- 2Department of Neurology, Affiliated Hospital of Yanbian University, Yanji, Jilin 133000, China
- KMID: 2514947
- DOI: http://doi.org/10.4196/kjpp.2021.25.3.217
Abstract
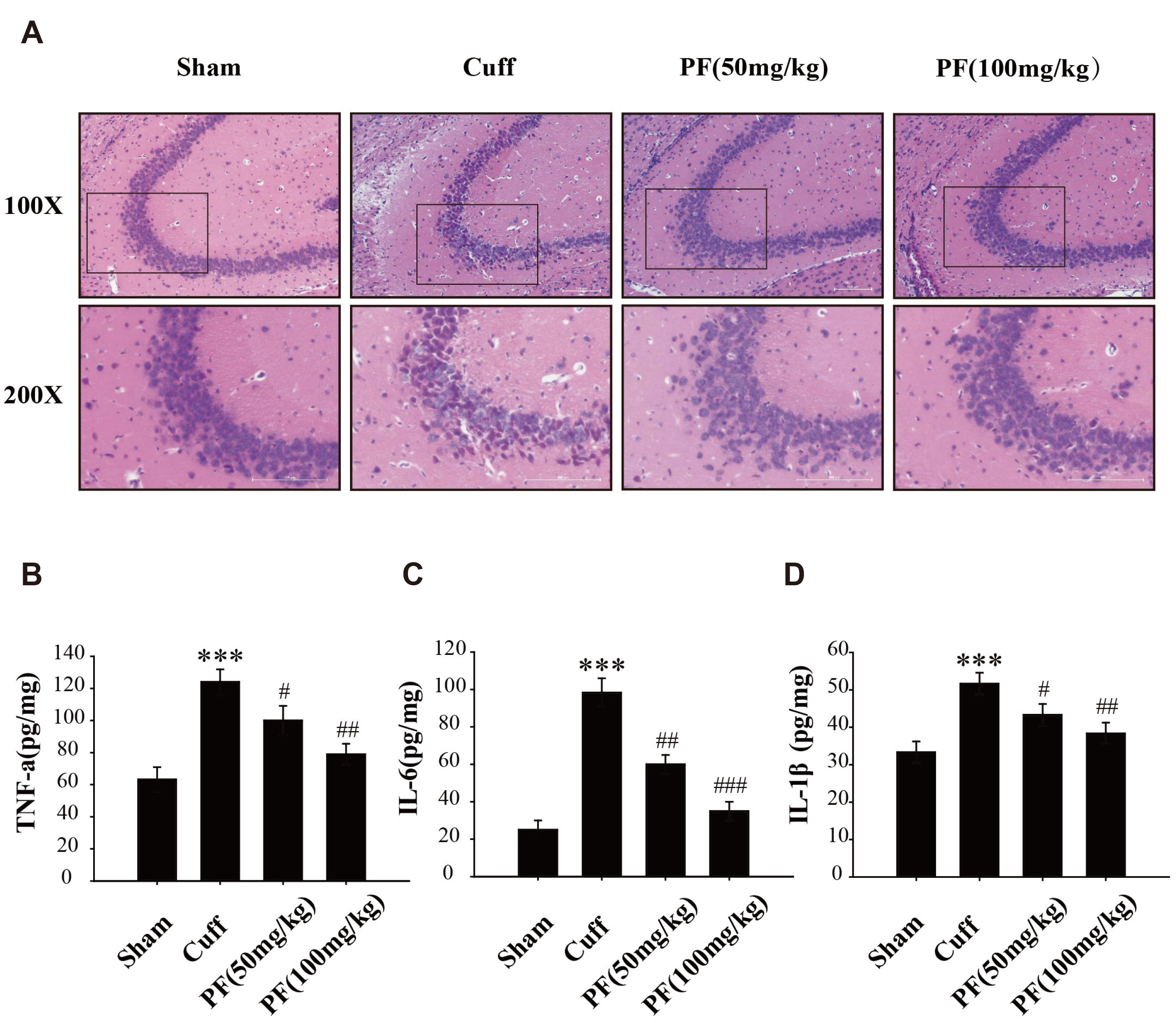
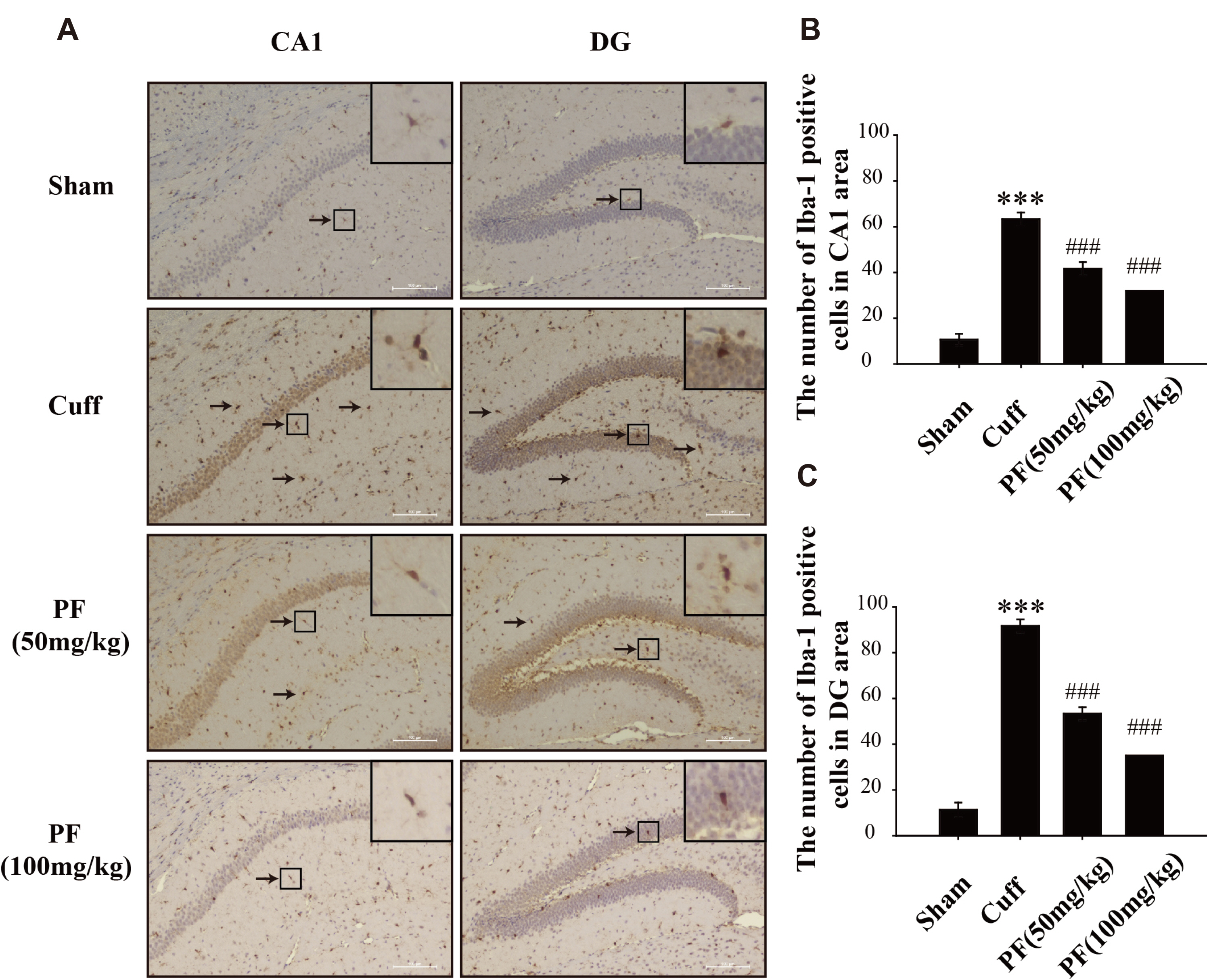
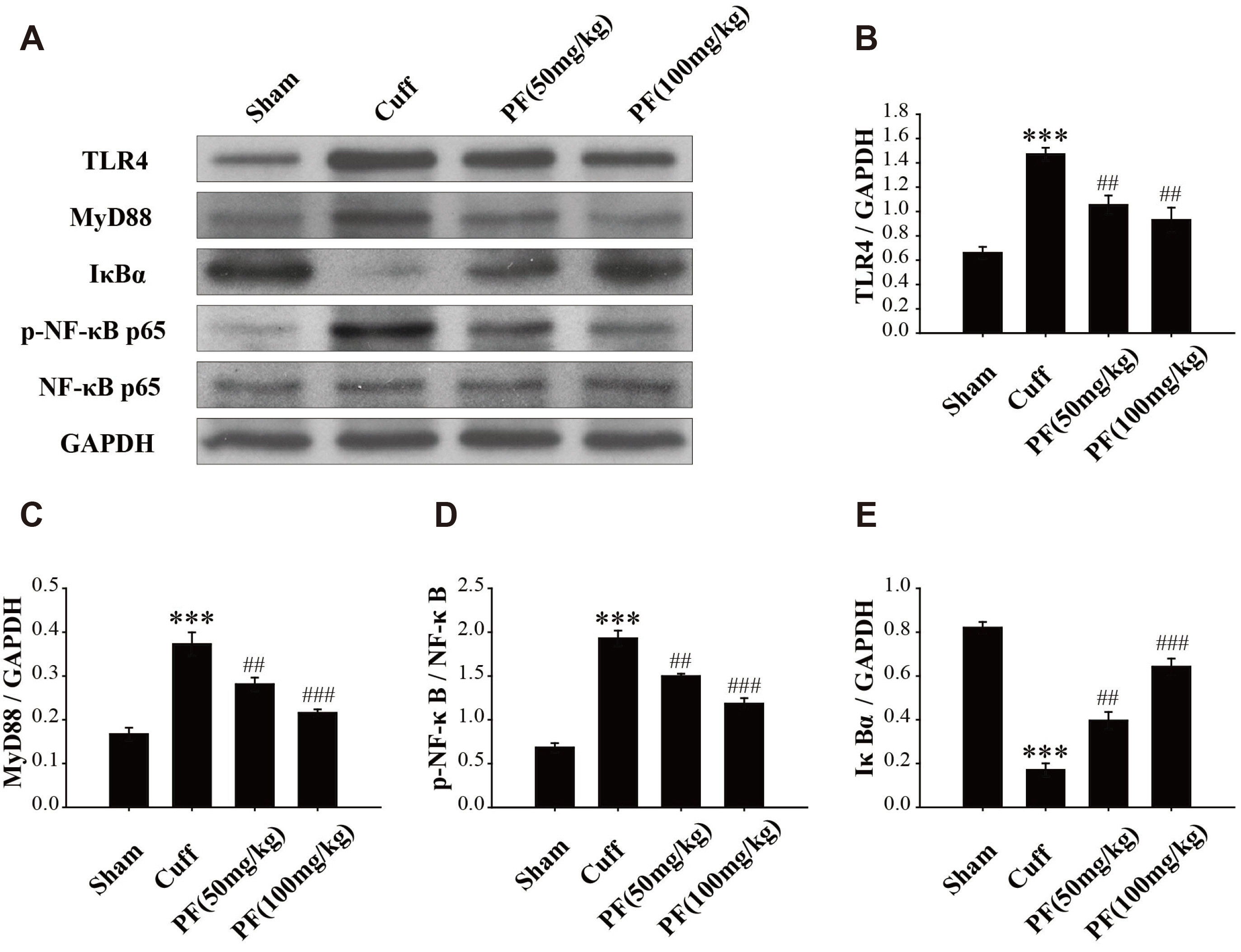
- Neuropathic pain (NP) that contributes to the comorbidity between pain and depression is a clinical dilemma. Neuroinflammatory responses are known to have potentially important roles in the initiation of NP and depressive mood. In this study, we aimed to investigate the effects of paeoniflorin (PF) on NP-induced depression-like behaviors by targeting the hippocampal neuroinflammation through the toll-like receptor 4 (TLR4)/nuclear factor-kappa B (NF-kB) signaling pathway. We used a murine model of NP caused by unilateral sciatic nerve cuffing (Cuff ). PF was injected intraperitoneally once a day for a total of 14 days. Pain and depression-like behavior changes were evaluated via behavioral tests. Pathological changes in the hippocampus of mice were observed by H&E staining. The levels of proinflammatory cytokines in the hippocampus were detected using ELISA. Activated microglia were measured by immunohistochemical staining. The TLR4/NF-kB signaling pathwayassociated protein expression in the hippocampus was detected by western blotting. We found that the PF could significantly alleviate Cuff-induced hyperalgesia and depressive behaviors, lessen the pathological damage to the hippocampal cell, reduce proinflammatory cytokines levels, and inhibit microglial over-activation. Furthermore, PF downregulated the expression levels of TLR4/NF-kB signaling pathwayrelated proteins in the hippocampus. These results indicate that PF is an effective drug for improving the comorbidity between NP and depression.
Keyword
Figure
Reference
-
1. Jensen TS, Baron R, Haanpää M, Kalso E, Loeser JD, Rice ASC, Treede RD. 2011; A new definition of neuropathic pain. Pain. 152:2204–2205. DOI: 10.1016/j.pain.2011.06.017. PMID: 21764514.
Article2. Conrad R, Wegener I, Geiser F, Kleiman A. 2013; Temperament, character, and personality disorders in chronic pain. Curr Pain Headache Rep. 17:318. DOI: 10.1007/s11916-012-0318-3. PMID: 23338770.
Article3. Radat F, Margot-Duclot A, Attal N. 2013; Psychiatric co-morbidities in patients with chronic peripheral neuropathic pain: a multicentre cohort study. Eur J Pain. 17:1547–1557. DOI: 10.1002/j.1532-2149.2013.00334.x. PMID: 23720357.
Article4. Mechawar N, Savitz J. 2016; Neuropathology of mood disorders: do we see the stigmata of inflammation? Transl Psychiatry. 6:e946. DOI: 10.1038/tp.2016.212. PMID: 27824355. PMCID: PMC5314124.
Article5. Mahajan GJ, Vallender EJ, Garrett MR, Challagundla L, Overholser JC, Jurjus G, Dieter L, Syed M, Romero DG, Benghuzzi H, Stockmeier CA. 2018; Altered neuro-inflammatory gene expression in hippocampus in major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 82:177–186. DOI: 10.1016/j.pnpbp.2017.11.017. PMID: 29175309. PMCID: PMC5801125.
Article6. Wang YL, Han QQ, Gong WQ, Pan DH, Wang LZ, Hu W, Yang M, Li B, Yu J, Liu Q. 2018; Microglial activation mediates chronic mild stress-induced depressive- and anxiety-like behavior in adult rats. J Neuroinflammation. 15:21. DOI: 10.1186/s12974-018-1054-3. PMID: 29343269. PMCID: PMC5773028.
Article7. Lei Y, Chen CJ, Yan XX, Li Z, Deng XH. 2017; Early-life lipopolysaccharide exposure potentiates forebrain expression of NLRP3 inflammasome proteins and anxiety-like behavior in adolescent rats. Brain Res. 1671:43–54. DOI: 10.1016/j.brainres.2017.06.014. PMID: 28655515.
Article8. Liu S, Liu YP, Song WB, Song XJ. 2013; EphrinB-EphB receptor signaling contributes to bone cancer pain via Toll-like receptor and proinflammatory cytokines in rat spinal cord. Pain. 154:2823–2835. DOI: 10.1016/j.pain.2013.08.017. PMID: 23973554.
Article9. Cheng Y, Pardo M, Armini RS, Martinez A, Mouhsine H, Zagury JF, Jope RS, Beurel E. 2016; Stress-induced neuroinflammation is mediated by GSK3-dependent TLR4 signaling that promotes susceptibility to depression-like behavior. Brain Behav Immun. 53:207–222. DOI: 10.1016/j.bbi.2015.12.012. PMID: 26772151. PMCID: PMC4783243.
Article10. Zhou YX, Gong XH, Zhang H, Peng C. 2020; A review on the pharmacokinetics of paeoniflorin and its anti-inflammatory and immunomodulatory effects. Biomed Pharmacother. 130:110505. DOI: 10.1016/j.biopha.2020.110505. PMID: 32682112.
Article11. Wang D, Liu L, Li S, Wang C. 2018; Effects of paeoniflorin on neurobehavior, oxidative stress, brain insulin signaling, and synaptic alterations in intracerebroventricular streptozotocin-induced cognitive impairment in mice. Physiol Behav. 191:12–20. DOI: 10.1016/j.physbeh.2018.03.016. PMID: 29572012.
Article12. Zheng M, Liu C, Fan Y, Yan P, Shi D, Zhang Y. 2017; Neuroprotection by Paeoniflorin in the MPTP mouse model of Parkinson's disease. Neuropharmacology. 116:412–420. DOI: 10.1016/j.neuropharm.2017.01.009. PMID: 28093210.
Article13. Gu X, Cai Z, Cai M, Liu K, Liu D, Zhang Q, Tan J, Ma Q. 2016; Protective effect of paeoniflorin on inflammation and apoptosis in the cerebral cortex of a transgenic mouse model of Alzheimer's disease. Mol Med Rep. 13:2247–2252. DOI: 10.3892/mmr.2016.4805. PMID: 26796245.
Article14. Zhou J, Wang L, Wang J, Wang C, Yang Z, Wang C, Zhu Y, Zhang J. 2016; Paeoniflorin and albiflorin attenuate neuropathic pain via MAPK pathway in chronic constriction injury rats. Evid Based Complement Alternat Med. 2016:8082753. DOI: 10.1155/2016/8082753. PMID: 27429639. PMCID: PMC4939188.
Article15. Yin D, Liu YY, Wang TX, Hu ZZ, Qu WM, Chen JF, Cheng NN, Huang ZL. 2016; Paeoniflorin exerts analgesic and hypnotic effects via adenosine A1 receptors in a mouse neuropathic pain model. Psychopharmacology (Berl). 233:281–293. DOI: 10.1007/s00213-015-4108-6. PMID: 26514553.
Article16. Hu B, Xu G, Zhang X, Xu L, Zhou H, Ma Z, Shen X, Zhu J, Shen R. 2018; Paeoniflorin attenuates inflammatory pain by inhibiting microglial activation and Akt-NF-κB signaling in the central nervous system. Cell Physiol Biochem. 47:842–850. DOI: 10.1159/000490076. PMID: 29807368.
Article17. Benbouzid M, Pallage V, Rajalu M, Waltisperger E, Doridot S, Poisbeau P, Freund-Mercier MJ, Barrot M. 2008; Sciatic nerve cuffing in mice: a model of sustained neuropathic pain. Eur J Pain. 12:591–599. DOI: 10.1016/j.ejpain.2007.10.002. PMID: 18006342.
Article18. Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. 1994; Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 53:55–63. DOI: 10.1016/0165-0270(94)90144-9. PMID: 7990513.
Article19. Wang Z, Huang H, Yang S, Huang S, Guo J, Tang Q, Qi F. 2016; Long-term effect of ropivacaine nanoparticles for sciatic nerve block on postoperative pain in rats. Int J Nanomedicine. 11:2081–2090. DOI: 10.2147/IJN.S101563. PMID: 27274236. PMCID: PMC4876796.20. Schneiderhan J, Orizondo C. 2017; Chronic pain: how to approach these 3 common conditions. J Fam Pract. 66:145–157. PMID: 28249053.21. Gustorff B, Dorner T, Likar R, Grisold W, Lawrence K, Schwarz F, Rieder A. 2008; Prevalence of self-reported neuropathic pain and impact on quality of life: a prospective representative survey. Acta Anaesthesiol Scand. 52:132–136. DOI: 10.1111/j.1399-6576.2007.01486.x. PMID: 17976220.
Article22. Kuthati Y, Lin SH, Chen IJ, Wong CS. 2019; Melatonin and their analogs as a potential use in the management of Neuropathic pain. J Formos Med Assoc. 118:1177–1186. DOI: 10.1016/j.jfma.2018.09.017. PMID: 30316678.
Article23. Qiu F, Zhong X, Mao Q, Huang Z. 2013; The antidepressant-like effects of paeoniflorin in mouse models. Exp Ther Med. 5:1113–1116. DOI: 10.3892/etm.2013.925. PMID: 23599734. PMCID: PMC3628593.
Article24. Qiu FM, Zhong XM, Mao QQ, Huang Z. 2013; Antidepressant-like effects of paeoniflorin on the behavioural, biochemical, and neurochemical patterns of rats exposed to chronic unpredictable stress. Neurosci Lett. 541:209–213. DOI: 10.1016/j.neulet.2013.02.029. PMID: 23481217.
Article25. Nam KN, Yae CG, Hong JW, Cho DH, Lee JH, Lee EH. 2013; Paeoniflorin, a monoterpene glycoside, attenuates lipopolysaccharide-induced neuronal injury and brain microglial inflammatory response. Biotechnol Lett. 35:1183–1189. DOI: 10.1007/s10529-013-1192-8. PMID: 23559368.
Article26. Wu YM, Jin R, Yang L, Zhang J, Yang Q, Guo YY, Li XB, Liu SB, Luo XX, Zhao MG. 2013; Phosphatidylinositol 3 kinase/protein kinase B is responsible for the protection of paeoniflorin upon H₂O₂-induced neural progenitor cell injury. Neuroscience. 240:54–62. DOI: 10.1016/j.neuroscience.2013.02.037. PMID: 23485815.27. Nguyen L, Kakeda S, Katsuki A, Sugimoto K, Otsuka Y, Ueda I, Igata R, Watanabe K, Kishi T, Iwata N, Korogi Y, Yoshimura R. 2019; Relationship between VEGF-related gene polymorphisms and brain morphology in treatment-naïve patients with first-episode major depressive disorder. Eur Arch Psychiatry Clin Neurosci. 269:785–794. DOI: 10.1007/s00406-018-0953-8. PMID: 30406404.
Article28. Taylor WD, Deng Y, Boyd BD, Donahue MJ, Albert K, McHugo M, Gandelman JA, Landman BA. 2020; Medial temporal lobe volumes in late-life depression: effects of age and vascular risk factors. Brain Imaging Behav. 14:19–29. DOI: 10.1007/s11682-018-9969-y. PMID: 30251182. PMCID: PMC6431585.
Article29. Mutso AA, Radzicki D, Baliki MN, Huang L, Banisadr G, Centeno MV, Radulovic J, Martina M, Miller RJ, Apkarian AV. 2012; Abnormalities in hippocampal functioning with persistent pain. J Neurosci. 32:5747–5756. DOI: 10.1523/JNEUROSCI.0587-12.2012. PMID: 22539837. PMCID: PMC3365570.
Article30. Jha MK, Jeon S, Suk K. 2012; Glia as a Link between neuroinflammation and neuropathic pain. Immune Netw. 12:41–47. DOI: 10.4110/in.2012.12.2.41. PMID: 22740789. PMCID: PMC3382663.
Article31. Burston JJ, Valdes AM, Woodhams SG, Mapp PI, Stocks J, Watson DJG, Gowler PRW, Xu L, Sagar DR, Fernandes G, Frowd N, Marshall L, Zhang W, Doherty M, Walsh DA, Chapman V. 2019; The impact of anxiety on chronic musculoskeletal pain and the role of astrocyte activation. Pain. 160:658–669. DOI: 10.1097/j.pain.0000000000001445. PMID: 30779717. PMCID: PMC6407811.
Article32. Ünal Ö, Akyol Y, Tander B, Ulus Y, Terzi Y, Kuru Ö. 2019; The relationship of illness perceptions with demographic features, pain severity, functional capacity, disability, depression, and quality of life in patients with chronic low back pain. Turk J Phys Med Rehabil. 65:301–308. DOI: 10.5606/tftrd.2019.3248. PMID: 31893266. PMCID: PMC6935732.
Article33. Kaster MP, Gadotti VM, Calixto JB, Santos AR, Rodrigues AL. 2012; Depressive-like behavior induced by tumor necrosis factor-α in mice. Neuropharmacology. 62:419–426. DOI: 10.1016/j.neuropharm.2011.08.018. PMID: 21867719.
Article34. Dellarole A, Morton P, Brambilla R, Walters W, Summers S, Bernardes D, Grilli M, Bethea JR. 2014; Neuropathic pain-induced depressive-like behavior and hippocampal neurogenesis and plasticity are dependent on TNFR1 signaling. Brain Behav Immun. 41:65–81. DOI: 10.1016/j.bbi.2014.04.003. PMID: 24938671. PMCID: PMC4167189.
Article35. Bachtell RK, Jones JD, Heinzerling KG, Beardsley PM, Comer SD. 2017; Glial and neuroinflammatory targets for treating substance use disorders. Drug Alcohol Depend. 180:156–170. DOI: 10.1016/j.drugalcdep.2017.08.003. PMID: 28892721. PMCID: PMC5790191.
Article36. Mika J, Zychowska M, Popiolek-Barczyk K, Rojewska E, Przewlocka B. 2013; Importance of glial activation in neuropathic pain. Eur J Pharmacol. 716:106–119. DOI: 10.1016/j.ejphar.2013.01.072. PMID: 23500198.
Article37. Krabbe KS, Reichenberg A, Yirmiya R, Smed A, Pedersen BK, Bruunsgaard H. 2005; Low-dose endotoxemia and human neuropsychological functions. Brain Behav Immun. 19:453–460. DOI: 10.1016/j.bbi.2005.04.010. PMID: 15963684.
Article38. Chen J, Wang Z, Zheng Z, Chen Y, Khor S, Shi K, He Z, Wang Q, Zhao Y, Zhang H, Li X, Li J, Yin J, Wang X, Xiao J. 2017; Neuron and microglia/macrophage-derived FGF10 activate neuronal FGFR2/PI3K/Akt signaling and inhibit microglia/macrophages TLR4/NF-κB-dependent neuroinflammation to improve functional recovery after spinal cord injury. Cell Death Dis. 8:e3090. DOI: 10.1038/cddis.2017.490. PMID: 28981091. PMCID: PMC5682656.
Article39. Zhou Z, Lin J, Huo R, Huang W, Zhang J, Wang L, Sun Y, Shen B, Li N. 2012; Total glucosides of paeony attenuated functional maturation of dendritic cells via blocking TLR4/5 signaling in vivo. Int Immunopharmacol. 14:275–282. DOI: 10.1016/j.intimp.2012.07.012. PMID: 22846756.
Article40. Wium-Andersen MK, Ørsted DD, Nielsen SF, Nordestgaard BG. 2013; Elevated C-reactive protein levels, psychological distress, and depression in 73, 131 individuals. JAMA Psychiatry. 70:176–184. DOI: 10.1001/2013.jamapsychiatry.102. PMID: 23266538.41. Dahl J, Ormstad H, Aass HC, Malt UF, Bendz LT, Sandvik L, Brundin L, Andreassen OA. 2014; The plasma levels of various cytokines are increased during ongoing depression and are reduced to normal levels after recovery. Psychoneuroendocrinology. 45:77–86. DOI: 10.1016/j.psyneuen.2014.03.019. PMID: 24845179.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Paeoniflorin ameliorates Aβ-stimulated neuroinflammation via regulation of NF-κB signaling pathway and Aβ degradation in C6 glial cells
- Paeoniflorin treatment regulates TLR4/NF-κB signaling, reduces cerebral oxidative stress and improves white matter integrity in neonatal hypoxic brain injury
- Epigallocatechin-3-gallate rescues LPS-impaired adult hippocampal neurogenesis through suppressing the TLR4-NF-kappaB signaling pathway in mice
- Grape seed proanthocyanidin extract ameliorates murine autoimmune arthritis through regulation of TLR4/MyD88/NF-κB signaling pathway
- MiR-590 Inhibits Endothelial Cell Apoptosis by Inactivating the TLR4/NF-κB Pathway in Atherosclerosis