J Korean Med Sci.
2020 Nov;35(44):e361. 10.3346/jkms.2020.35.e361.
Cerebrospinal Fluid Biomarkers for the Diagnosis and Classification of Alzheimer's Disease Spectrum
- Affiliations
-
- 1Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 2Neuroscience Center, Samsung Medical Center, Seoul, Korea
- 3Samsung Alzheimer's Research Center, Samsung Medical Center, Seoul, Korea
- 4Department of Clinical Research Design & Evaluation, SAIHST, Sungkyunkwan University, Seoul, Korea
- 5Stem Cell & Regenerative Medicine Institute, Samsung Medical Center, Seoul, Korea
- 6Department of Health Sciences and Technology, SAIHST, Sungkyunkwan University, Seoul, Korea
- KMID: 2508763
- DOI: http://doi.org/10.3346/jkms.2020.35.e361
Abstract
- Background
Cerebrospinal fluid (CSF) biomarkers are increasingly used in clinical practice for the diagnosis of Alzheimer's disease (AD). We aimed to 1) determine cutoff values of CSF biomarkers for AD, 2) investigate their clinical utility by estimating a concordance with amyloid positron emission tomography (PET), and 3) apply ATN (amyloid/tau/neurodegeneration) classification based on CSF results.
Methods
We performed CSF analysis in 51 normal controls (NC), 23 mild cognitive impairment (MCI) and 65 AD dementia (ADD) patients at the Samsung Medical Center in Korea. We attempted to develop cutoff of CSF biomarkers for differentiating ADD from NC using receiver operating characteristic analysis. We also investigated a concordance between CSF and amyloid PET results and applied ATN classification scheme based on CSF biomarker abnormalities to characterize our participants.
Results
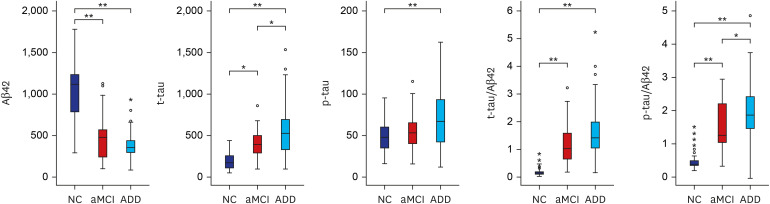
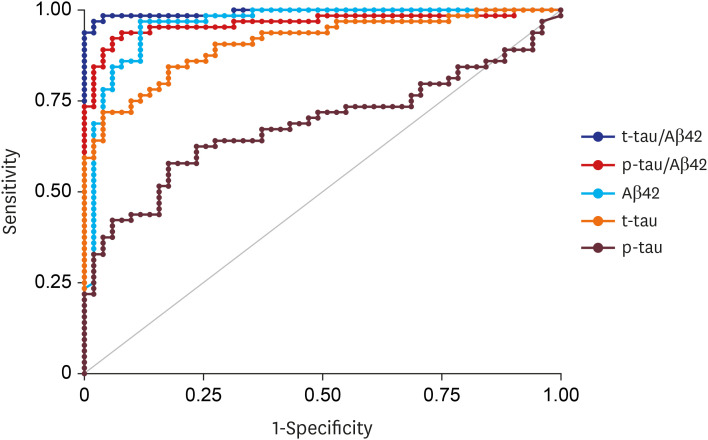
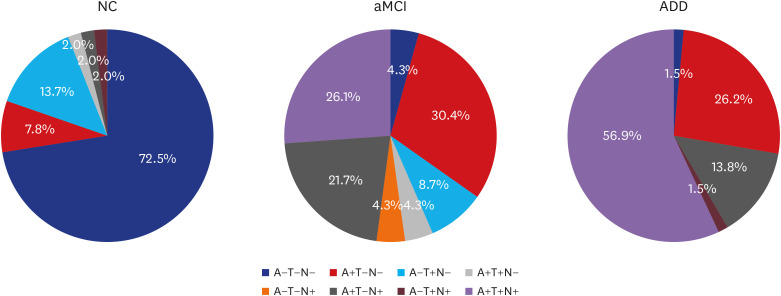
CSF Aβ42, total tau (t-tau) and phosphorylated tau (p-tau) significantly differed across the three groups. The area under curve for the differentiation between NC and ADD was highest in t-tau/Aβ42 (0.994) followed by p-tau/Aβ42 (0.963), Aβ42 (0.960), t-tau (0.918), and p-tau (0.684). The concordance rate between CSF Aβ42 and amyloid PET results was 92%. Finally, ATN classification based on CSF biomarker abnormalities led to a majority of NC categorized into A-T-N-(73%), MCI as A+T-N-(30%)/A+T+N+(26%), and ADD as A+T+N+(57%).
Conclusion
CSF biomarkers had high sensitivity and specificity in differentiating ADD from NC and were as accurate as amyloid PET. The ATN subtypes based on CSF biomarkers may further serve to predict the prognosis.
Keyword
Figure
Reference
-
1. Dubois B, Feldman HH, Jacova C, Hampel H, Molinuevo JL, Blennow K, et al. Advancing research diagnostic criteria for Alzheimer's disease: the IWG-2 criteria. Lancet Neurol. 2014; 13(6):614–629. PMID: 24849862.
Article2. McKhann G, Knopman D, Chertkow H, Hyman B, Jack C Jr, Kawas C. The diagnosis of dementia due to Alzheimer's disease: recommendations from the NIAA Association workgroups on diagnostic guidelines for AD. Alzheimers Dement. 2011; 7(3):263–269. PMID: 21514250.3. Albert MS, DeKosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, et al. The diagnosis of mild cognitive impairment due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7(3):270–279. PMID: 21514249.
Article4. McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7(3):263–269. PMID: 21514250.
Article5. Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, et al. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7(3):280–292. PMID: 21514248.
Article6. Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, et al. NIA-AA research framework: toward a biological definition of Alzheimer's disease. Alzheimers Dement. 2018; 14(4):535–562. PMID: 29653606.
Article7. Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Feldman HH, Frisoni GB, et al. A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology. 2016; 87(5):539–547. PMID: 27371494.
Article8. Müller EG, Edwin TH, Stokke C, Navelsaker SS, Babovic A, Bogdanovic N, et al. Amyloid-β PET-Correlation with cerebrospinal fluid biomarkers and prediction of Alzheimer´s disease diagnosis in a memory clinic. PLoS One. 2019; 14(8):e0221365. PMID: 31430334.
Article9. Jack CR Jr, Knopman DS, Jagust WJ, Petersen RC, Weiner MW, Aisen PS, et al. Tracking pathophysiological processes in Alzheimer's disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013; 12(2):207–216. PMID: 23332364.
Article10. Bateman RJ, Xiong C, Benzinger TL, Fagan AM, Goate A, Fox NC, et al. Clinical and biomarker changes in dominantly inherited Alzheimer's disease. N Engl J Med. 2012; 367(9):795–804. PMID: 22784036.
Article11. Apostolova LG, Hwang KS, Andrawis JP, Green AE, Babakchanian S, Morra JH, et al. 3D PIB and CSF biomarker associations with hippocampal atrophy in ADNI subjects. Neurobiol Aging. 2010; 31(8):1284–1303. PMID: 20538372.
Article12. Olsson B, Lautner R, Andreasson U, Öhrfelt A, Portelius E, Bjerke M, et al. CSF and blood biomarkers for the diagnosis of Alzheimer's disease: a systematic review and meta-analysis. Lancet Neurol. 2016; 15(7):673–684. PMID: 27068280.
Article13. Portelius E, Zetterberg H, Skillbäck T, Törnqvist U, Andreasson U, Trojanowski JQ, et al. Cerebrospinal fluid neurogranin: relation to cognition and neurodegeneration in Alzheimer's disease. Brain. 2015; 138(Pt 11):3373–3385. PMID: 26373605.
Article14. Mattsson N, Insel PS, Palmqvist S, Portelius E, Zetterberg H, Weiner M, et al. Cerebrospinal fluid tau, neurogranin, and neurofilament light in Alzheimer's disease. EMBO Mol Med. 2016; 8(10):1184–1196. PMID: 27534871.
Article15. Park SA, Chae WS, Kim HJ, Shin HS, Kim S, Im JY, et al. Cerebrospinal fluid biomarkers for the diagnosis of Alzheimer disease in South Korea. Alzheimer Dis Assoc Disord. 2017; 31(1):13–18. PMID: 28030437.
Article16. McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7(3):263–269. PMID: 21514250.
Article17. Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol. 1999; 56(3):303–308. PMID: 10190820.18. ClinincalTrials.gov. Updated 2020. Accessed July 21, 2020. https://clinicaltrials.gov/ct2/show/NCT02054208.19. Park SA, Kang JH, Kang ES, Ki CS, Roh JH, Youn YC, et al. A consensus in Korea regarding a protocol to reduce preanalytical sources of variability in the measurement of the cerebrospinal fluid biomarkers of Alzheimer's disease. J Clin Neurol. 2015; 11(2):132–141. PMID: 25851891.
Article20. Lee JH, Kim SH, Kim GH, Seo SW, Park HK, Oh SJ, et al. Identification of pure subcortical vascular dementia using 11C-Pittsburgh compound B. Neurology. 2011; 77(1):18–25. PMID: 21593437.
Article21. Barthel H, Luthardt J, Becker G, Patt M, Hammerstein E, Hartwig K, et al. Individualized quantification of brain β-amyloid burden: results of a proof of mechanism phase 0 florbetaben PET trial in patients with Alzheimer’s disease and healthy controls. Eur J Nucl Med Mol Imaging. 2011; 38(9):1702–1714. PMID: 21547601.
Article22. Kim SE, Woo S, Kim SW, Chin J, Kim HJ, Lee BI, et al. A nomogram for predicting amyloid PET positivity in amnestic mild cognitive impairment. J Alzheimers Dis. 2018; 66(2):681–691. PMID: 30320571.
Article23. Kim HJ, Lim TS, Lee SM, Kim TS, Kim Y, An YS, et al. Cerebrospinal fluid levels of β-Amyloid 40 and β-amyloid 42 are proportionately decreased in amyloid positron-emission tomography negative idiopathic normal-pressure hydrocephalus patients. J Clin Neurol. 2019; 15(3):353–359. PMID: 31286708.
Article24. Jeppsson A, Höltta M, Zetterberg H, Blennow K, Wikkelsø C, Tullberg M. Amyloid mis-metabolism in idiopathic normal pressure hydrocephalus. Fluids Barriers CNS. 2016; 13(1):13. PMID: 27472944.
Article25. Graff-Radford NR. Alzheimer CSF biomarkers may be misleading in normal-pressure hydrocephalus. Neurology. 2014; 83(17):1573–1575. PMID: 25332445.
Article26. Blennow K, Hampel H, Weiner M, Zetterberg H. Cerebrospinal fluid and plasma biomarkers in Alzheimer disease. Nat Rev Neurol. 2010; 6(3):131–144. PMID: 20157306.
Article27. Buerger K, Ewers M, Pirttilä T, Zinkowski R, Alafuzoff I, Teipel SJ, et al. CSF phosphorylated tau protein correlates with neocortical neurofibrillary pathology in Alzheimer's disease. Brain. 2006; 129(Pt 11):3035–3041. PMID: 17012293.
Article28. Clark CM, Xie S, Chittams J, Ewbank D, Peskind E, Galasko D, et al. Cerebrospinal fluid tau and beta-amyloid: how well do these biomarkers reflect autopsy-confirmed dementia diagnoses? Arch Neurol. 2003; 60(12):1696–1702. PMID: 14676043.29. Wahlund LO, Blennow K. Cerebrospinal fluid biomarkers for disease stage and intensity in cognitively impaired patients. Neurosci Lett. 2003; 339(2):99–102. PMID: 12614904.
Article30. Hansson O, Lehmann S, Otto M, Zetterberg H, Lewczuk P. Advantages and disadvantages of the use of the CSF Amyloid β (Aβ) 42/40 ratio in the diagnosis of Alzheimer's Disease. Alzheimers Res Ther. 2019; 11(1):34. PMID: 31010420.
Article31. Kim HJ, Lim TS, Lee SM, Kim TS, Kim Y, An YS, et al. Cerebrospinal fluid levels of β-amyloid 40 and β-amyloid 42 are proportionately decreased in amyloid positron-emission tomography negative idiopathic normal-pressure hydrocephalus patients. J Clin Neurol. 2019; 15(3):353–359. PMID: 31286708.
Article32. Altomare D, de Wilde A, Ossenkoppele R, Pelkmans W, Bouwman F, Groot C, et al. Applying the ATN scheme in a memory clinic population: the ABIDE project. Neurology. 2019; 93(17):e1635–46. PMID: 31597710.33. Hwang J, Jeong JH, Yoon SJ, Park KW, Kim EJ, Yoon B, et al. Clinical and biomarker characteristics according to clinical spectrum of Alzheimer's disease (AD) in the validation cohort of Korean brain aging study for the early diagnosis and prediction of AD. J Clin Med. 2019; 8(3):E341. PMID: 30862124.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Logopenic Progressive Aphasia Revealing Positive Cerebrospinal Fluid Biomarkers for Alzheimer's Disease
- Biological Predictors of Alzheimer's Disease Treatment
- Cerebrospinal Fluid Aspartate Aminotransferase in Alzheimer Disease and Vascular Dementia
- Cerebrospinal Biomarker Cut-off Methods Defined Only by Alzheimer's Disease Predict More Precisely Conversions of Mild Cognitive Impairment
- Harnessing Cerebrospinal Fluid Biomarkers in Clinical Trials for Treating Alzheimer's and Parkinson's Diseases: Potential and Challenges