J Clin Neurol.
2019 Oct;15(4):527-536. 10.3988/jcn.2019.15.4.527.
Relationships between [¹â¸F]-THK5351 Retention and Language Functions in Primary Progressive Aphasia
- Affiliations
-
- 1Neuroscience Research Institute, Gachon University, Incheon, Korea.
- 2Department of Neurology, Inha University School of Medicine, Incheon, Korea.
- 3Department of Neuroscience, College of Medicine, Gachon University, Incheon, Korea.
- 4Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 5Department of Neurology, Gil Medical Center, Gachon University College of Medicine, Incheon, Korea. ynoh@gachon.ac.kr
- 6Department of Neurology, Seoul Medical Center, Seoul, Korea.
- 7Department of Neurology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea.
- 8Department of Health Science and Technology, GAIHST, Gachon University, Incheon, Korea.
- KMID: 2467763
- DOI: http://doi.org/10.3988/jcn.2019.15.4.527
Abstract
- BACKGROUND AND PURPOSE
There are three distinct subtypes of primary progressive aphasia (PPA): the nonfluent/agrammatic variant (nfvPPA), the semantic variant (svPPA), and the logopenic variant (lvPPA). We sought to characterize the pattern of [¹â¸F]-THK5351 retention across all three subtypes and determine the topography of [¹â¸F]-THK5351 retention correlated with each neurolinguistic score.
METHODS
We enrolled 50 participants, comprising 13 PPA patients (3 nfvPPA, 5 svPPA, and 5 lvPPA) and 37 subjects with normal cognition (NC) who underwent 3.0-tesla magnetic resonance imaging, [¹â¸F]-THK5351 positron-emission tomography scans, and detailed neuropsychological tests. The PPA patients additionally participated in extensive neurolinguistic tests. Voxel-wise and region-of-interest-based analyses were performed to analyze [¹â¸F]-THK5351 retention.
RESULTS
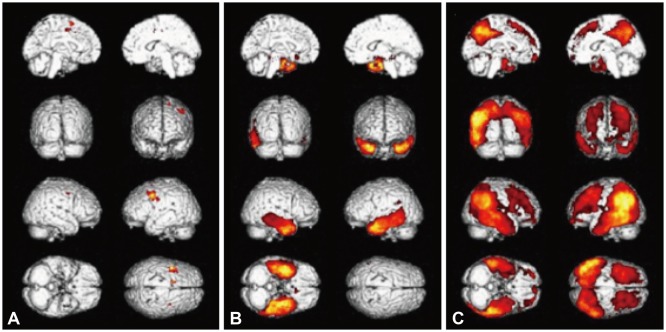
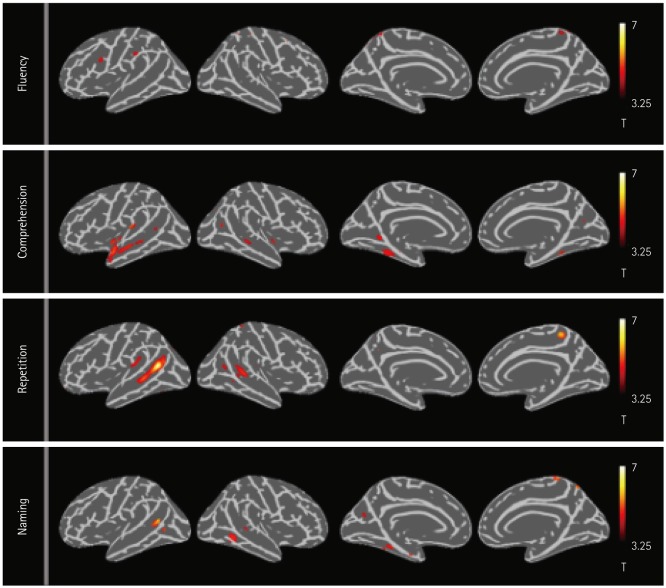
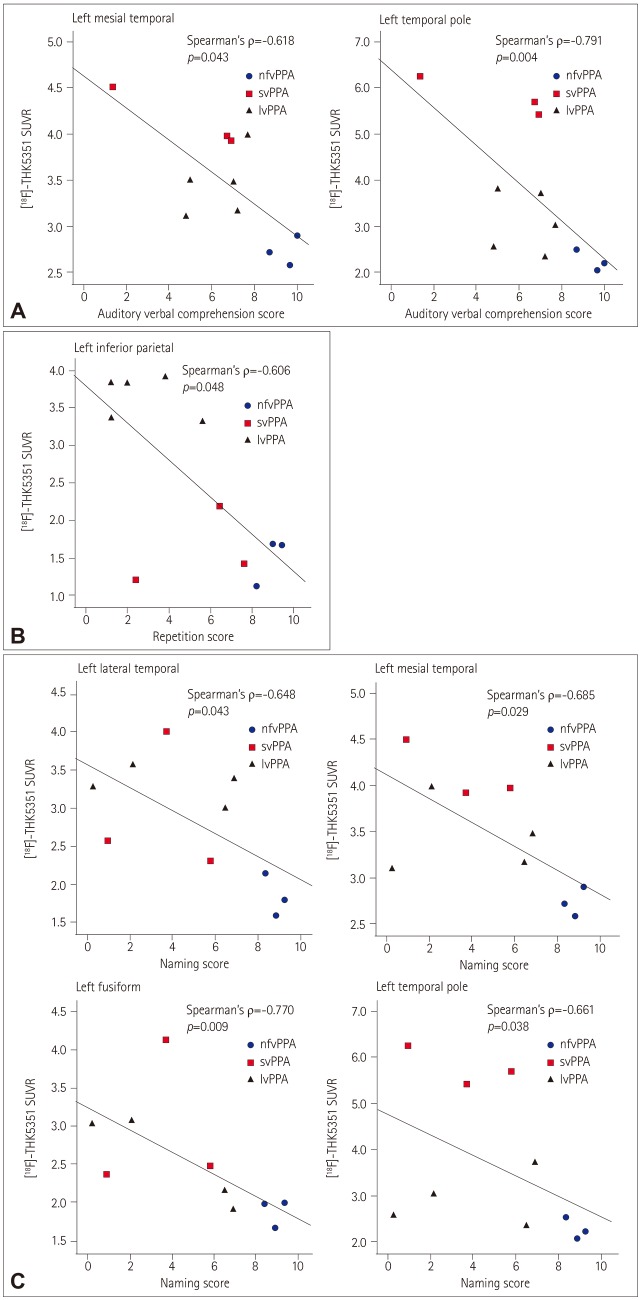
The nfvPPA patients exhibited higher [¹â¸F]-THK5351 retention in the the left inferior frontal and precentral gyri. In svPPA patients, [¹â¸F]-THK5351 retention was elevated in the anteroinferior and lateral temporal cortices compared to the NC group (left>right). The lvPPA patients exhibited predominant [¹â¸F]-THK5351 retention in the inferior parietal, lateral temporal, and dorsolateral prefrontal cortices, and the precuneus (left>right). [¹â¸F]-THK5351 retention in the left inferior frontal area was associated with lower fluency scores. Comprehension was correlated with [¹â¸F]-THK5351 retention in the left temporal cortices. Repetition was associated with [¹â¸F]-THK5351 retention in the left inferior parietal and posterior temporal areas, while naming difficulty was correlated with retention in the left fusiform and temporal cortices.
CONCLUSIONS
The pattern of [¹â¸F]-THK5351 retention was well matched with clinical and radiological findings for each PPA subtype, in agreement with the anatomical and functional location of each language domain.
Keyword
MeSH Terms
Figure
Reference
-
2. Gorno-Tempini ML, Hillis AE, Weintraub S, Kertesz A, Mendez M, Cappa SF, et al. Classification of primary progressive aphasia and its variants. Neurology. 2011; 76:1006–1014. PMID: 21325651.
Article3. Goedert M, Spillantini MG, Jakes R, Rutherford D, Crowther RA. Multiple isoforms of human microtubule-associated protein tau: sequences and localization in neurofibrillary tangles of Alzheimer's disease. Neuron. 1989; 3:519–526. PMID: 2484340.
Article4. Josephs KA, Hodges JR, Snowden JS, Mackenzie IR, Neumann M, Mann DM, et al. Neuropathological background of phenotypical variability in frontotemporal dementia. Acta Neuropathol. 2011; 122:137–153. PMID: 21614463.
Article5. Spinelli EG, Mandelli ML, Miller ZA, Santos-Santos MA, Wilson SM, Agosta F, et al. Typical and atypical pathology in primary progressive aphasia variants. Ann Neurol. 2017; 81:430–443. PMID: 28133816.
Article6. Grossman M. Primary progressive aphasia: clinicopathological correlations. Nat Rev Neurol. 2010; 6:88–97. PMID: 20139998.
Article7. Harada R, Okamura N, Furumoto S, Furukawa K, Ishiki A, Tomita N, et al. 18F-THK5351: a novel PET radiotracer for imaging neurofibrillary pathology in Alzheimer disease. J Nucl Med. 2016; 57:208–214. PMID: 26541774.8. Ng KP, Pascoal TA, Mathotaarachchi S, Therriault J, Kang MS, Shin M, et al. Monoamine oxidase B inhibitor, selegiline, reduces 18F-THK5351 uptake in the human brain. Alzheimers Res Ther. 2017; 9:25. PMID: 28359327.
Article9. Yoon CW, Jeong HJ, Seo S, Lee SY, Suh MK, Heo JH, et al. 18F-THK5351 PET imaging in nonfluent-agrammatic variant primary progressive aphasia. Dement Neurocogn Disord. 2018; 17:110–119. PMID: 30906400.10. Lee H, Seo S, Lee SY, Jeong HJ, Woo SH, Lee KM, et al. [18F]-THK5351 PET imaging in patients with semantic variant primary progressive aphasia. Alzheimer Dis Assoc Disord. 2018; 32:62–69. PMID: 29028649.11. Kim H, Na DL. Normative data on the Korean version of the Western Aphasia Battery. J Clin Exp Neuropsychol. 2004; 26:1011–1020. PMID: 15590457.
Article12. Kang Y, Jahng S, Na DL. Seoul Neuropsychological Screening Battery (SNSB-II). 2nd ed. Seoul: Human Brain Research & Consulting Co.;2012.13. Kang JM, Lee SY, Seo S, Jeong HJ, Woo SH, Lee H, et al. Tau positron emission tomography using [18F]THK5351 and cerebral glucose hypometabolism in Alzheimer's disease. Neurobiol Aging. 2017; 59:210–219. PMID: 28890300.14. Okamura N, Furumoto S, Fodero-Tavoletti MT, Mulligan RS, Harada R, Yates P, et al. Non-invasive assessment of Alzheimer's disease neurofibrillary pathology using 18F-THK5105 PET. Brain. 2014; 137:1762–1771. PMID: 24681664.15. Lockhart SN, Baker SL, Okamura N, Furukawa K, Ishiki A, Furumoto S, et al. Dynamic PET measures of tau accumulation in cognitively normal older adults and Alzheimer's disease patients measured using [18F] THK-5351. PLoS One. 2016; 11:e0158460. PMID: 27355840.
Article16. Thurfjell L, Lilja J, Lundqvist R, Buckley C, Smith A, Vandenberghe R. Automated quantification of 18F-flutemetamol PET activity for categorizing scans as negative or positive for brain amyloid:concordance with visual image reads. J Nucl Med. 2014; 55:1623–1628. PMID: 25146124.17. Greve DN, Salat DH, Bowen SL, Izquierdo-Garcia D, Schultz AP, Catana C, et al. Different partial volume correction methods lead to different conclusions: an 18F-FDG-PET study of aging. Neuroimage. 2016; 132:334–343. PMID: 26915497.18. Greve DN, Svarer C, Fisher PM, Feng L, Hansen AE, Baare W, et al. Cortical surface-based analysis reduces bias and variance in kinetic modeling of brain PET data. Neuroimage. 2014; 92:225–236. PMID: 24361666.
Article19. Cho H, Kim HJ, Choi JY, Ryu YH, Lee MS, Na DL, et al. 18F-flortaucipir uptake patterns in clinical subtypes of primary progressive aphasia. Neurobiol Aging. 2019; 75:187–197. PMID: 30594046.20. Josephs KA, Martin PR, Botha H, Schwarz CG, Duffy JR, Clark HM, et al. [18F]AV-1451 tau-PET and primary progressive aphasia. Ann Neurol. 2018; 83:599–611. PMID: 29451323.21. Kobayashi R, Hayashi H, Kawakatsu S, Ishiki A, Okamura N, Arai H, et al. [18F]THK-5351 PET imaging in early-stage semantic variant primary progressive aphasia: a report of two cases and a literature review. BMC Neurol. 2018; 18:109. PMID: 30089453.
Article22. Itabashi R, Nishio Y, Kataoka Y, Yazawa Y, Furui E, Matsuda M, et al. Damage to the left precentral gyrus is associated with apraxia of speech in acute stroke. Stroke. 2016; 47:31–36. PMID: 26645260.
Article23. Rohrer JD, Rossor MN, Warren JD. Apraxia in progressive nonfluent aphasia. J Neurol. 2010; 257:569–574. PMID: 19908082.
Article24. Dronkers NF, Wilkins DP, Van Valin RD Jr, Redfern BB, Jaeger JJ. Lesion analysis of the brain areas involved in language comprehension. Cognition. 2004; 92:145–177. PMID: 15037129.
Article25. Holland R, Lambon Ralph MA. The anterior temporal lobe semantic hub is a part of the language neural network: selective disruption of irregular past tense verbs by rTMS. Cereb Cortex. 2010; 20:2771–2775. PMID: 20223808.
Article26. Fridriksson J, Kjartansson O, Morgan PS, Hjaltason H, Magnusdottir S, Bonilha L, et al. Impaired speech repetition and left parietal lobe damage. J Neurosci. 2010; 30:11057–11061. PMID: 20720112.
Article27. Hickok G, Poeppel D. The cortical organization of speech processing. Nat Rev Neurosci. 2007; 8:393–402. PMID: 17431404.
Article28. Migliaccio R, Boutet C, Valabregue R, Ferrieux S, Nogues M, Lehéricy S, et al. The brain network of naming: a lesson from primary progressive aphasia. PLoS One. 2016; 11:e0148707. PMID: 26901052.
Article29. Harada R, Okamura N, Furumoto S, Furukawa K, Ishiki A, Tomita N, et al. Validation of the binding specificity of tau PET tracer [18F] THK-5351 on postmortem human brain samples. Alzheimers Dement. 2015; 11:P271.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Two Cases of Primary Progressive Aphasia
- ¹â¸F-THK5351 PET Imaging in Nonfluent-Agrammatic Variant Primary Progressive Aphasia
- Evaluation of Aphasia
- A Case of the Fluent Form of Primary Progressive Aphasia Presenting as Conduction Aphasia
- Corticobasal Degeneration Presenting as Non-Fluent/Agrammatic Primary Progressive Aphasia: A Case Report