Anat Cell Biol.
2019 Dec;52(4):369-377. 10.5115/acb.19.119.
Benefits of hesperidin in central nervous system disorders: a review
- Affiliations
-
- 1Department of Veterinary Anatomy, College of Veterinary Medicine and Veterinary Medical Research Institute, Jeju National University, Jeju, Korea. shint@jejunu.ac.kr
- 2Department of Veterinary Toxicology, College of Veterinary Medicine and Institute of Veterinary Science, Kangwon National University, Chuncheon, Korea.
- 3Laboratory of Comparative Animal Medicine, Division of Animal Life Science, Institute of Agriculture, Tokyo University of Agriculture and Technology, Tokyo, Japan.
- 4Laboratory of Veterinary Molecular Pathology and Therapeutics, Division of Animal Life Science, Graduate School, Institute of Agriculture, Tokyo University of Agriculture and Technology, Tokyo, Japan.
- KMID: 2466688
- DOI: http://doi.org/10.5115/acb.19.119
Abstract
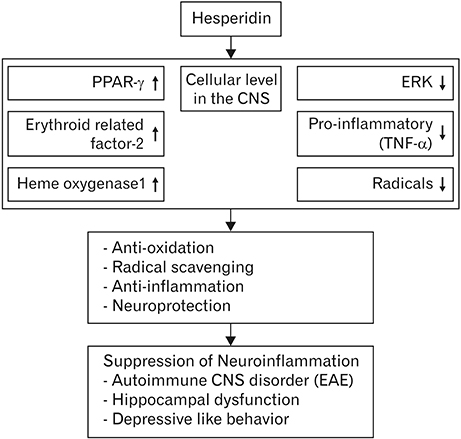
- Citrus species contain significant amounts of flavonoids that possess antioxidant activities; furthermore, dietary citrus is not associated with adverse effects or cytotoxicity in healthy individuals. Hesperidin, which is an abundant flavanone glycoside in the peel of citrus fruits, possesses a variety of biological capabilities that include antioxidant and anti-inflammatory actions. Over the last few decades, many studies have been investigated the biological actions of hesperidin and its aglycone, hesperetin, as well as their underlying mechanisms. Due to the antioxidant effects of hesperidin and its derivatives, the cardioprotective and anti-cancer effects of these compounds have been widely reviewed. Although the biological activities of hesperidin in neurodegenerative diseases have been evaluated, its potential involvement in a variety of central nervous system (CNS) disorders, including autoimmune demyelinating disease, requires further investigation in terms of the underlying mechanisms. Thus, the present review will focus on the potential role of hesperidin in diverse models of CNS neuroinflammation, including experimental autoimmune encephalomyelitis, with special consideration given to its antioxidant and anti-inflammatory effects in neurodegenerative disease models. Additionally, current evidence provides information regarding the nutraceutical use of hesperidin to prevent various CNS disorders.
Keyword
MeSH Terms
Figure
Reference
-
1. Garg A, Garg S, Zaneveld LJ, Singla AK. Chemistry and pharmacology of the Citrus bioflavonoid hesperidin. Phytother Res. 2001; 15:655–669.2. Umeno A, Horie M, Murotomi K, Nakajima Y, Yoshida Y. Antioxidative and antidiabetic effects of natural polyphenols and isoflavones. Molecules. 2016; 21:E708.3. Roohbakhsh A, Parhiz H, Soltani F, Rezaee R, Iranshahi M. Neuropharmacological properties and pharmacokinetics of the citrus flavonoids hesperidin and hesperetin: a mini-review. Life Sci. 2014; 113:1–6.4. Camps-Bossacoma M, Franch À, Pérez-Cano FJ, Castell M. Influence of hesperidin on the systemic and intestinal rat immune response. Nutrients. 2017; 9:E580.5. Estruel-Amades S, Massot-Cladera M, Pérez-Cano FJ, Franch À, Castell M, Camps-Bossacoma M. Hesperidin effects on gut microbiota and gut-associated lymphoid tissue in healthy rats. Nutrients. 2019; 11:E324.6. Testai L, Calderone V. Nutraceutical value of citrus flavanones and their implications in cardiovascular disease. Nutrients. 2017; 9:E502.7. Parhiz H, Roohbakhsh A, Soltani F, Rezaee R, Iranshahi M. Antioxidant and anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: an updated review of their molecular mechanisms and experimental models. Phytother Res. 2015; 29:323–331.8. Roohbakhsh A, Parhiz H, Soltani F, Rezaee R, Iranshahi M. Molecular mechanisms behind the biological effects of hesperidin and hesperetin for the prevention of cancer and cardiovascular diseases. Life Sci. 2015; 124:64–74.9. Homayouni F, Haidari F, Hedayati M, Zakerkish M, Ahmadi K. Blood pressure lowering and anti-inflammatory effects of hesperidin in type 2 diabetes: a randomized double-blind controlled clinical trial. Phytother Res. 2018; 32:1073–1079.10. Sawikr Y, Yarla NS, Peluso I, Kamal MA, Aliev G, Bishayee A. Neuroinflammation in Alzheimer's disease: the preventive and therapeutic potential of polyphenolic nutraceuticals. Adv Protein Chem Struct Biol. 2017; 108:33–57.11. Jung UJ, Kim SR. Beneficial effects of flavonoids against Parkinson's disease. J Med Food. 2018; 21:421–432.12. Antunes MS, Goes AT, Boeira SP, Prigol M, Jesse CR. Protective effect of hesperidin in a model of Parkinson's disease induced by 6-hydroxydopamine in aged mice. Nutrition. 2014; 30:1415–1422.13. Kumar P, Kumar A. Protective effect of hesperidin and naringin against 3-nitropropionic acid induced Huntington's like symptoms in rats: possible role of nitric oxide. Behav Brain Res. 2010; 206:38–46.14. Menze ET, Tadros MG, Abdel-Tawab AM, Khalifa AE. Potential neuroprotective effects of hesperidin on 3-nitropropionic acidinduced neurotoxicity in rats. Neurotoxicology. 2012; 33:1265–1275.15. Antunes MS, Jesse CR, Ruff JR, de Oliveira Espinosa D, Gomes NS, Altvater EE, Donato F, Giacomeli R, Boeira SP. Hesperidin reverses cognitive and depressive disturbances induced by olfactory bulbectomy in mice by modulating hippocampal neurotrophins and cytokine levels and acetylcholinesterase activity. Eur J Pharmacol. 2016; 789:411–420.16. El-Marasy SA, Abdallah HM, El-Shenawy SM, El-Khatib AS, El-Shabrawy OA, Kenawy SA. Anti-depressant effect of hesperidin in diabetic rats. Can J Physiol Pharmacol. 2014; 92:945–952.17. Ciftci O, Ozcan C, Kamisli O, Cetin A, Basak N, Aytac B. Hesperidin, a citrus flavonoid, has the ameliorative effects against Experimental Autoimmune Encephalomyelitis (EAE) in a C57BL/J6 Mouse Model. Neurochem Res. 2015; 40:1111–1120.18. Haghmorad D, Mahmoudi MB, Salehipour Z, Jalayer Z, Momtazi Brojeni AA, Rastin M, Kokhaei P, Mahmoudi M. Hesperidin ameliorates immunological outcome and reduces neuroinflammation in the mouse model of multiple sclerosis. J Neuroimmunol. 2017; 302:23–33.19. Gaur V, Kumar A. Hesperidin pre-treatment attenuates NOmediated cerebral ischemic reperfusion injury and memory dysfunction. Pharmacol Rep. 2010; 62:635–648.20. Kosari-Nasab M, Shokouhi G, Ghorbanihaghjo A, Abbasi MM, Salari AA. Hesperidin attenuates depression-related symptoms in mice with mild traumatic brain injury. Life Sci. 2018; 213:198–205.21. Hajialyani M, Hosein Farzaei M, Echeverría J, Nabavi SM, Uriarte E, Sobarzo-Sánchez E. Hesperidin as a neuroprotective agent: a review of animal and clinical evidence. Molecules. 2019; 24:E648.22. Hwang SL, Shih PH, Yen GC. Neuroprotective effects of citrus flavonoids. J Agric Food Chem. 2012; 60:877–885.23. Cirmi S, Ferlazzo N, Lombardo GE, Ventura-Spagnolo E, Gangemi S, Calapai G, Navarra M. Neurodegenerative diseases: might citrus flavonoids play a protective role? Molecules. 2016; 21:E1312.24. Raza SS, Khan MM, Ahmad A, Ashafaq M, Khuwaja G, Tabassum R, Javed H, Siddiqui MS, Safhi MM, Islam F. Hesperidin ameliorates functional and histological outcome and reduces neuroinflammation in experimental stroke. Brain Res. 2011; 1420:93–105.25. Said UZ, Saada HN, Abd-Alla MS, Elsayed ME, Amin AM. Hesperidin attenuates brain biochemical changes of irradiated rats. Int J Radiat Biol. 2012; 88:613–618.26. Rotimi SO, Bankole GE, Adelani IB, Rotimi OA. Hesperidin prevents lipopolysaccharide-induced endotoxicity in rats. Immunopharmacol Immunotoxicol. 2016; 38:364–371.27. Chen B, Lu Y, Chen Y, Cheng J. The role of Nrf2 in oxidative stress-induced endothelial injuries. J Endocrinol. 2015; 225:R83–R99.28. Zhu C, Dong Y, Liu H, Ren H, Cui Z. Hesperetin protects against H(2)O(2)-triggered oxidative damage via upregulation of the Keap1-Nrf2/HO-1 signal pathway in ARPE-19 cells. Biomed Pharmacother. 2017; 88:124–133.29. Elshazly SM, Abd El Motteleb DM, Ibrahim IA. Hesperidin protects against stress induced gastric ulcer through regulation of peroxisome proliferator activator receptor gamma in diabetic rats. Chem Biol Interact. 2018; 291:153–161.30. Agrawal YO, Sharma PK, Shrivastava B, Ojha S, Upadhya HM, Arya DS, Goyal SN. Hesperidin produces cardioprotective activity via PPAR-γ pathway in ischemic heart disease model in diabetic rats. PLoS One. 2014; 9:e111212.31. Mahmoud AM. Hesperidin protects against cyclophosphamideinduced hepatotoxicity by upregulation of PPARγ and abrogation of oxidative stress and inflammation. Can J Physiol Pharmacol. 2014; 92:717–724.32. Justin-Thenmozhi A, Dhivya Bharathi M, Kiruthika R, Manivasagam T, Borah A, Essa MM. Attenuation of aluminum chlorideinduced neuroinflammation and caspase activation through the AKT/GSK-3β pathway by hesperidin in Wistar rats. Neurotox Res. 2018; 34:463–476.33. Maekawa S, Sato K, Fujita K, Daigaku R, Tawarayama H, Murayama N, Moritoh S, Yabana T, Shiga Y, Omodaka K, Maruyama K, Nishiguchi KM, Nakazawa T. The neuroprotective effect of hesperidin in NMDA-induced retinal injury acts by suppressing oxidative stress and excessive calpain activation. Sci Rep. 2017; 7:6885.34. Martínez MC, Fernandez SP, Loscalzo LM, Wasowski C, Paladini AC, Marder M, Medina JH, Viola H. Hesperidin, a flavonoid glycoside with sedative effect, decreases brain pERK1/2 levels in mice. Pharmacol Biochem Behav. 2009; 92:291–296.35. Li M, Shao H, Zhang X, Qin B. Hesperidin alleviates lipopolysaccharide-induced neuroinflammation in mice by promoting the miRNA-132 pathway. Inflammation. 2016; 39:1681–1689.36. Nones J, Spohr TC, Gomes FC. Effects of the flavonoid hesperidin in cerebral cortical progenitors in vitro: indirect action through astrocytes. Int J Dev Neurosci. 2012; 30:303–313.37. Youdim KA, Dobbie MS, Kuhnle G, Proteggente AR, Abbott NJ, Rice-Evans C. Interaction between flavonoids and the blood-brain barrier: in vitro studies. J Neurochem. 2003; 85:180–192.38. Li C, Zug C, Qu H, Schluesener H, Zhang Z. Hesperidin ameliorates behavioral impairments and neuropathology of transgenic APP/PS1 mice. Behav Brain Res. 2015; 281:32–42.39. Schwartz M, Deczkowska A. Neurological disease as a failure of brain-immune crosstalk: the multiple faces of neuroinflammation. Trends Immunol. 2016; 37:668–679.40. Shin T, Kojima T, Tanuma N, Ishihara Y, Matsumoto Y. The subarachnoid space as a site for precursor T cell proliferation and effector T cell selection in experimental autoimmune encephalomyelitis. J Neuroimmunol. 1995; 56:171–178.41. Shin T, Ahn M, Matsumoto Y. Mechanism of experimental autoimmune encephalomyelitis in Lewis rats: recent insights from macrophages. Anat Cell Biol. 2012; 45:141–148.42. Ginwala R, McTish E, Raman C, Singh N, Nagarkatti M, Nagarkatti P, Sagar D, Jain P, Khan ZK. Apigenin, a natural flavonoid, attenuates EAE severity through the modulation of dendritic cell and other immune cell functions. J Neuroimmune Pharmacol. 2016; 11:36–47.43. Verbeek R, van Tol EA, van Noort JM. Oral flavonoids delay recovery from experimental autoimmune encephalomyelitis in SJL mice. Biochem Pharmacol. 2005; 70:220–228.44. Filho CB, Del Fabbro L, de Gomes MG, Goes AT, Souza LC, Boeira SP, Jesse CR. Kappa-opioid receptors mediate the antidepressant-like activity of hesperidin in the mouse forced swimming test. Eur J Pharmacol. 2013; 698:286–291.45. Donato F, de Gomes MG, Goes AT, Filho CB, Del Fabbro L, Antunes MS, Souza LC, Boeira SP, Jesse CR. Hesperidin exerts antidepressant-like effects in acute and chronic treatments in mice: possible role of l-arginine-NO-cGMP pathway and BDNF levels. Brain Res Bull. 2014; 104:19–26.46. Souza LC, de Gomes MG, Goes AT, Del Fabbro L, Filho CB, Boeira SP, Jesse CR. Evidence for the involvement of the serotonergic 5-HT(1A) receptors in the antidepressant-like effect caused by hesperidin in mice. Prog Neuropsychopharmacol Biol Psychiatry. 2013; 40:103–109.47. Justin Thenmozhi A, William Raja TR, Manivasagam T, Janakiraman U, Essa MM. Hesperidin ameliorates cognitive dysfunction, oxidative stress and apoptosis against aluminium chloride induced rat model of Alzheimer's disease. Nutr Neurosci. 2017; 20:360–368.48. Javed H, Vaibhav K, Ahmed ME, Khan A, Tabassum R, Islam F, Safhi MM, Islam F. Effect of hesperidin on neurobehavioral, neuroinflammation, oxidative stress and lipid alteration in intracerebroventricular streptozotocin induced cognitive impairment in mice. J Neurol Sci. 2015; 348:51–59.49. Chang CY, Lin TY, Lu CW, Huang SK, Wang YC, Chou SS, Wang SJ. Hesperidin inhibits glutamate release and exerts neuroprotection against excitotoxicity induced by kainic acid in the hippocampus of rats. Neurotoxicology. 2015; 50:157–169.50. Rong Z, Pan R, Xu Y, Zhang C, Cao Y, Liu D. Hesperidin pretreatment protects hypoxia-ischemic brain injury in neonatal rat. Neuroscience. 2013; 255:292–299.51. Verbeek R, Plomp AC, van Tol EA, van Noort JM. The flavones luteolin and apigenin inhibit in vitro antigen-specific proliferation and interferon-gamma production by murine and human autoimmune T cells. Biochem Pharmacol. 2004; 68:621–629.52. van der Veen RC. Nitric oxide and T helper cell immunity. Int Immunopharmacol. 2001; 1:1491–1500.53. Nath N, Morinaga O, Singh I. S-nitrosoglutathione a physiologic nitric oxide carrier attenuates experimental autoimmune encephalomyelitis. J Neuroimmune Pharmacol. 2010; 5:240–251.54. Willenborg DO, Staykova M, Fordham S, O'Brien N, Linares D. The contribution of nitric oxide and interferon gamma to the regulation of the neuro-inflammation in experimental autoimmune encephalomyelitis. J Neuroimmunol. 2007; 191:16–25.55. Osorio-Querejeta I, Sáenz-Cuesta M, Muñoz-Culla M, Otaegui D. Models for studying myelination, demyelination and remyelination. Neuromolecular Med. 2017; 19:181–192.56. Plemel JR, Michaels NJ, Weishaupt N, Caprariello AV, Keough MB, Rogers JA, Yukseloglu A, Lim J, Patel VV, Rawji KS, Jensen SK, Teo W, Heyne B, Whitehead SN, Stys PK, Yong VW. Mechanisms of lysophosphatidylcholine-induced demyelination: a primary lipid disrupting myelinopathy. Glia. 2018; 66:327–347.57. Baradaran S, Hajizadeh Moghaddam A, Ghasemi-Kasman M. Hesperetin reduces myelin damage and ameliorates glial activation in lysolecithin-induced focal demyelination model of rat optic chiasm. Life Sci. 2018; 207:471–479.58. Naeimi R, Baradaran S, Ashrafpour M, Moghadamnia AA, Ghasemi-Kasman M. Querectin improves myelin repair of optic chiasm in lyolecithin-induced focal demyelination model. Biomed Pharmacother. 2018; 101:485–493.59. Planche V, Panatier A, Hiba B, Ducourneau EG, Raffard G, Dubourdieu N, Maitre M, Lesté-Lasserre T, Brochet B, Dousset V, Desmedt A, Oliet SH, Tourdias T. Selective dentate gyrus disruption causes memory impairment at the early stage of experimental multiple sclerosis. Brain Behav Immun. 2017; 60:240–254.60. Mandolesi G, Grasselli G, Musumeci G, Centonze D. Cognitive deficits in experimental autoimmune encephalomyelitis: neuroinflammation and synaptic degeneration. Neurol Sci. 2010; 31:Suppl 2. S255–S259.61. Ziehn MO, Avedisian AA, Tiwari-Woodruff S, Voskuhl RR. Hippocampal CA1 atrophy and synaptic loss during experimental autoimmune encephalomyelitis, EAE. Lab Invest. 2010; 90:774–786.62. Kurkowska-Jastrzębska I, Swiątkiewicz M, Zaremba M, Cudna A, Piechal A, Pyrzanowska J, Widy-Tyszkiewicz E, Członkowska A. Neurodegeneration and inflammation in hippocampus in experimental autoimmune encephalomyelitis induced in rats by one: time administration of encephalitogenic T cells. Neuroscience. 2013; 248:690–698.63. Nones J, E Spohr TC, Gomes FC. Hesperidin, a flavone glycoside, as mediator of neuronal survival. Neurochem Res. 2011; 36:1776–1784.64. Levite M. Glutamate, T cells and multiple sclerosis. J Neural Transm (Vienna). 2017; 124:775–798.65. Jovanova-Nesic K, Shoenfeld Y, Spector NH. Aluminum excytotoxicity and neuroautotoimmunity: the role of the brain expression of CD32+ (FcγRIIa), ICAM-1+ and CD3ξ in aging. Curr Aging Sci. 2012; 5:209–217.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neurosteroids and neurological disorders
- The value of computerized axial tomography of the brain in children with central nervous system disorders
- Characteristics of Swallowing Disorder in Pediatric Patients with Central Nervous System Disorders: Comparing to Central Nervous System Intact Patients with Gastro-intestinal Disorders
- Central Nervous System-Peripheral Immune System Dialogue in Neurological Disorders: Possible Application of Neuroimmunology in Urology
- Two Cases of Post-traumatic Dyskinesia