Infect Chemother.
2019 Dec;51(4):355-364. 10.3947/ic.2019.51.4.355.
Screening and Treatment of Latent Tuberculosis Infection among Healthcare Workers at a Referral Hospital in Korea
- Affiliations
-
- 1Division of Infectious Diseases, Soonchunhyang University Seoul Hospital, Soonchunhyang University College of Medicine, Seoul, Korea.
- 2Respiratory and Allergy Medicine, Department of Internal Medicine, Soonchunhyang University Seoul Hospital, Soonchunhyang University College of Medicine, Seoul, Korea. kyklung@schmc.ac.kr
- KMID: 2466468
- DOI: http://doi.org/10.3947/ic.2019.51.4.355
Abstract
- BACKGROUND
Healthcare workers (HCWs) have a high risk of tuberculosis (TB) infection. Since August 2017, Korea has mandated the testing of latent TB infection (LTBI) and recommended treatment from HCWs at medical institutions. However, the acceptance/completion rate and adverse events of LTBI treatment have not been analyzed.
MATERIALS AND METHODS
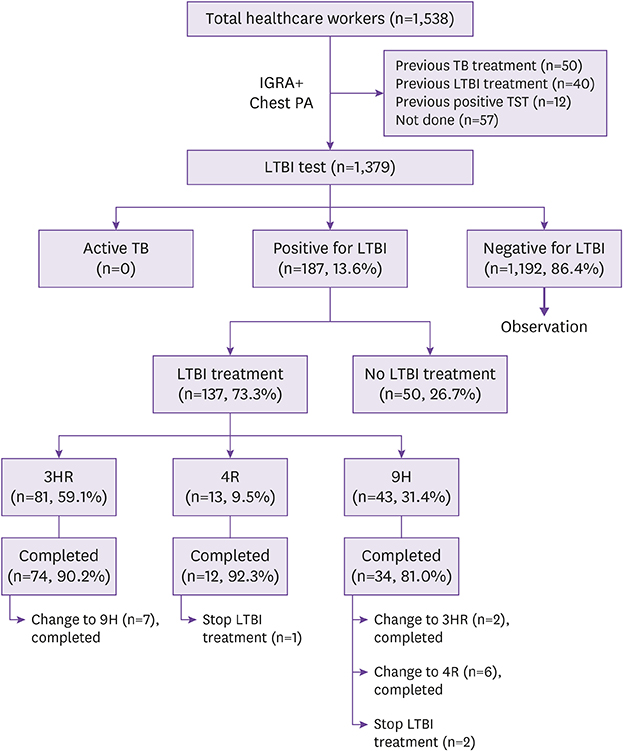
From February to August 2017, we conducted a retrospective study at a referral university hospital in Korea, to screen the interferon-gamma release assay (IGRA) tests conducted for all HCWs for detecting and treating LTBI. HCWs diagnosed with LTBI were offered a 9-month isoniazid (9H), 3-month isoniazid/rifampin (3HR), or 4-month rifampin regimen. We investigated the acceptance/completion rate, adverse events, and causes of discontinuation or change in LTBI medication. A major adverse event was one wherein a patient had any adverse event ≥grade 3 causing LTBI treatment interruption.
RESULTS
Of the 1,538 HCWs, 1,379 underwent IGRA testing for LTBI. Among them, 13.6% (187/1,379) tested positive and 73.3% (137/187) received treatment. The overall completion rate was 97.8% (134/137). HCWs were significantly more likely to complete first-line therapy with 3HR than with 9H (91.4% vs. 76.7%, P = 0.02). The most common major adverse event was hepatotoxicity (n = 7), followed by thrombocytopenia (n = 1) and anaphylactic shock (n = 1). Hepatotoxicity and hepatotoxicity (≥ grade 2) were more frequent in 9H than in 3HR (39.5% vs. 17.2%, P = 0.006 and 18.6% vs. 3.7%, P = 0.005, respectively). The median time to hepatotoxicity was 96 days (interquartile range, 20 - 103 days).
CONCLUSION
Completion of first-line therapy for LTBI is more likely with 3HR than with 9H. This might be related to the development of hepatotoxicity after around 3 months of treatment. Anaphylactic shock and platelet count should be carefully monitored in those receiving rifampin-containing regimens.
MeSH Terms
Figure
Cited by 1 articles
-
Institutional Tuberculosis Control and Elimination Program
Shi Nae Yu, Tae Hyong Kim, Su Ha Han, Yang-Ki Kim
Korean J Healthc Assoc Infect Control Prev. 2023;28(1):22-28. doi: 10.14192/kjicp.2023.28.1.22.
Reference
-
1. Lee JE, Kim YK, Kim TH, Kim KH, Lee EJ, Uh ST, Choi TY. What strategy can be applied to the patients with culture positive tuberculosis to reduce treatment delay in a private tertiary healthcare center? Infect Chemother. 2011; 43:42–47.
Article2. Park HY, Jeon K, Suh GY, Kwon OJ, Chung DR, Yoonchang SW, Kang ES, Koh WJ. Interferon-γ release assay for tuberculosis screening of healthcare workers at a Korean tertiary hospital. Scand J Infect Dis. 2010; 42:943–945.
Article3. Lee K, Han MK, Choi HR, Choi CM, Oh YM, Lee SD, Kim WS, Kim DS, Woo JH, Shim TS. Annual incidence of latent tuberculosis infection among newly employed nurses at a tertiary care university hospital. Infect Control Hosp Epidemiol. 2009; 30:1218–1222.
Article4. Park SY, Lee EJ, Kim YK, Lee SY, Kim GE, Jeong YS, Kim JH, Kim TH. Aggressive contact investigation of in-hospital exposure to active pulmonary tuberculosis. J Korean Med Sci. 2019; 34:e58.
Article5. Jensen PA, Lambert LA, Iademarco MF, Ridzon R; CDC. Guidelines for preventing the transmission of Mycobacterium tuberculosis in health-care settings, 2005. MMWR Recomm Rep. 2005; 54:1–141.6. National Institute for Health and Care Excellence (NICE). Tuberculosis: clinical diagnosis and management of tuberculosis, and measures for its prevention and control. Accessed 27 June 2019. Available at: http://www.nice.org.uk/guidance/cg117/chapter/1-Guideline.7. Jo KW, Hong Y, Park JS, Bae IG, Eom JS, Lee SR, Cho OH, Choo EJ, Heo JY, Woo JH, Shim TS. Prevalence of latent tuberculosis infection among health care workers in South Korea: a multicenter study. Tuberc Respir Dis (Seoul). 2013; 75:18–24.
Article8. Yeon JH, Seong H, Hur H, Park Y, Kim YA, Park YS, Han CH, Lee SM, Seo JH, Kang JG. Prevalence and risk factors of latent tuberculosis among Korean healthcare workers using whole-blood interferon-γ release assay. Sci Rep. 2018; 8:10113.
Article9. Cho KS. Tuberculosis control in the Republic of Korea. Epidemiol Health. 2018; 40:e2018036.
Article10. Centers for Disease Control and Prevention (CDC). Recommendations for use of an isoniazid-rifapentine regimen with direct observation to treat latent Mycobacterium tuberculosis infection. MMRW Morb Mortal Wkly Rep. 2011; 60:1650–1653.11. Arquello Perez E, Seo SK, Schneider WJ, Eisenstein C, Brown AE. Management of latent tuberculosis infection among healthcare workers: 10-year experience at a single center. Clin Infect Dis. 2017; 65:2105–2111.
Article12. Lee H, Koo GW, Min JH, Park TS, Park DW, Moon JY, Kim SH, Kim TH, Yoon HJ, Sohn JW. Factors associated with non-initiation of latent tuberculosis treatment among healthcare workers with a positive interferon-gamma releasing assay. Sci Rep. 2019; 9:61.
Article13. Khanna P, Nikolayevskyy V, Warburton F, Dobson E, Drobniewski F. Rate of latent tuberculosis infection detected by occupational health screening of nurses new to a London teaching hospital. Infect Control Hosp Epidemiol. 2009; 30:581–584.
Article14. NCI Common Terminology Criteria for Adverse Events. (CTCAE). CTCAE files. Accessed 27 June 2019. Available at: http://evs.nci.nih.gov/ftp1/CTCAE/About.html.15. Saukkonen JJ, Cohn DL, Jasmer RM, Schenker S, Jereb JA, Nolan CM, Peloquin CA, Gordin FM, Nunes D, Strader DB, Bernardo J, Venkataramanan R, Sterling TR. ATS (American Thoracic Society) Hepatotoxicity of Antituberculosis Therapy Subcommittee. An official ATS statement: hepatotoxicity of antituberculosis therapy. Am J Respir Crit Care Med. 2006; 174:935–952.
Article16. Schein YL, Madebo T, Andersen HE, Arnesen TM, Dyrhol-Riise AM, Tveiten H, White RA, Winje BA. Treatment completion for latent tuberculosis infection in Norway: a prospective cohort study. BMC Infect Dis. 2018; 18:587.
Article17. Sterling TR, Villarino ME, Borisov AS, Shang N, Gordin F, Bliven-Sizemore E, Hackman J, Hamilton CD, Menzies D, Kerrigan A, Weis SE, Weiner M, Wing D, Conde MB, Bozeman L, Horsburgh CR Jr, Chaisson RE. TB Trials Consortium PREVENT TB Study Team. Three months of rifapentine and isoniazid for latent tuberculosis infection. N Engl J Med. 2011; 365:2155–2166.
Article18. Belknap R, Holland D, Fenq PJ, Milet JP, Caylà JA, Martinson NA, Wright A, Chen MP, Moro RN, Scott NA, Arevalo B, Miró JM, Villarino ME, Weiner M, Borisov AS. TB Trials Consortium iAdhere Study Team. Self-administered versus directly observed once-weekly isoniazid and rifapentine treatment of latent tuberculosis infection: a randomized trial. Ann Intern Med. 2017; 167:689–697.
Article19. Getahun H, Matteelli A, Chaisson RE, Raviglione M. Latent Mycobacterium tuberculosis infection. N Engl J Med. 2015; 372:2127–2135.20. Fountain FF, Tolley E, Chrisman CR, Self TH. Isoniazid hepatotoxicity associated with treatment of latent tuberculosis infection: a 7-year evaluation from a public health tuberculosis clinic. Chest. 2005; 128:116–123.
Article21. Park SJ, Jo KW, Yoo B, Lee CK, Kim YG, Yang SK, Byeon JS, Kim KJ, Ye BD, Park SH, Shim TS. Comparison of LTBI treatment regimens for patients receiving anti-tumor necrosis factor therapy. Int J Tuberc Lung Dis. 2015; 19:342–348.
Article22. Njie GJ, Morris SB, Woodruff RY, Moro RN, Vernon AA, Borisov AS. Isoniazid-rifapentine for latent tuberculosis infection: a systematic review and meta-analysis. Am J Prev Med. 2018; 55:244–252.
Article23. Menzies D, Adjobimey M, Ruslami R, Trajman A, Sow O, Kim H, Obeng Baah J, Marks GB, Long R, Hoeppner V, Elwood K, Al-Jahdali H, Gninafon M, Apriani L, Koesoemadinata RC, Kritski A, Rolla V, Bah B, Camara A, Boakye I, Cook VJ, Goldberg H, Valiquette C, Hornby K, Dion MJ, Li PZ, Hill PC, Schwartzman K, Benedetti A. Four months of rifampin or nine months of isoniazid for latent tuberculosis in adults. N Engl J Med. 2018; 379:440–453.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnosis and Treatment of Latent Tuberculosis Infection in Healthcare Workers
- Diagnosis and Treatment of Latent Tuberculosis Infection for Healthcare Workers
- Prevalence and Risk Factors of Latent Tuberculosis Infection among Healthcare Workers Using Tuberculin Skin Test and Interferon-γ Release Assay at a Tertiary Hospital in South Korea
- Analysis of Prevalence and Risk Factors for Latent Tuberculosis Infection among Healthcare Workers
- A Retrospective Descriptive Study on the Status of Active Tuberculosis among Healthcare Workers in a Tertiary Hospital