World J Mens Health.
2020 Jan;38(1):103-114. 10.5534/wjmh.190034.
Proteomic Signatures in Spermatozoa Reveal the Role of Paternal Factors in Recurrent Pregnancy Loss
- Affiliations
-
- 1Redox Biology Laboratory, Department of Zoology, Center of Excellence in Environment and Public Health, Ravenshaw University, Cuttack, India. lsamanta@ravenshawuniversity.ac.in
- 2Department of Obstetrics and Gynaecology, Kar Clinic and Hospital Private Limited, Bhubaneswar, India.
- KMID: 2465415
- DOI: http://doi.org/10.5534/wjmh.190034
Abstract
- PURPOSE
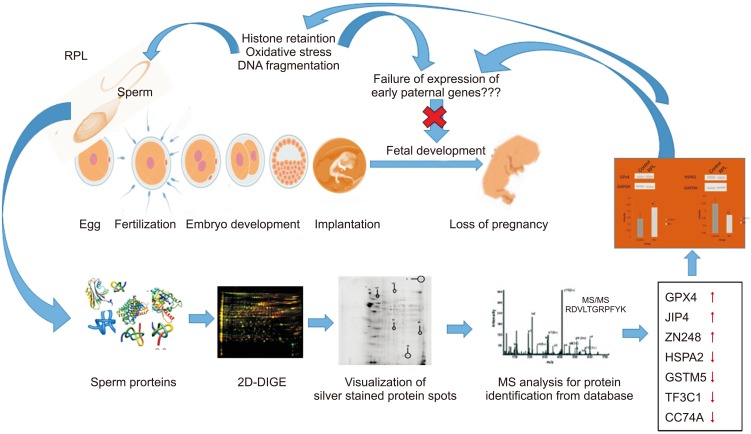
To identify the paternal factors responsible for aberrant embryo development leading to loss of foetus in recurrent pregnancy loss (RPL) through proteomic analysis of ejaculated spermatozoa.
MATERIALS AND METHODS
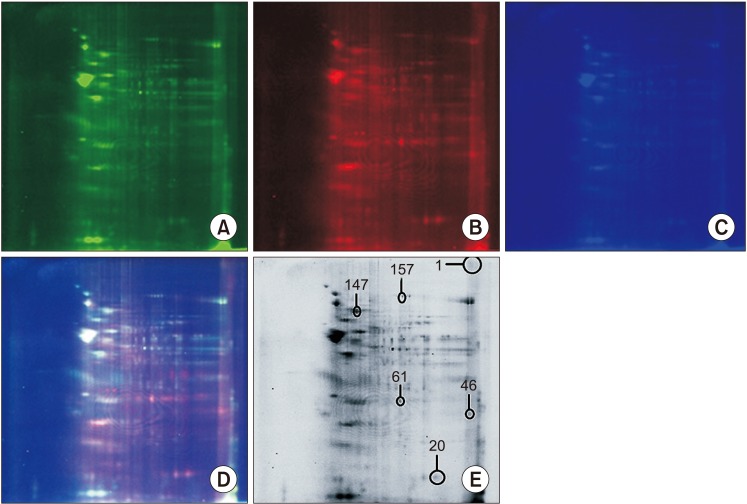
This prospective study consisted of male partners of RPL patients (n=16) experienced with two or more consecutive unexplained miscarriages and with no female factor abnormality as revealed by gynaecologic investigation including karyotyping and age matched fertile healthy volunteers (n=20). All samples were collected during 2013 to 2015 after getting institutional ethical approval and written consent from the participants. Seminal ejaculates were collected by masturbation after 2 to 3 days of sexual abstinence and analyzed according to World Health Organization 5th criteria 2010. Two-dimensional difference gel electrophoresis followed by mass spectrophotometric analysis was used to identify differentially expressed proteins (DEPs). Western blotting was used for validation of the key proteins.
RESULTS
The data identified 36 protein spots to be differentially expressed by more than 2-fold change with p<0.05 considered as significant. Matrix-assisted laser desorption/ionization time of flight/mass spectrometry identified GPx4, JIP4, ZN248 to be overexpressed while HSPA2, GSTM5, TF3C1, CC74A was underexpressed in RPL group. Western blot analysis confirmed the differential expression of key redox associated proteins GPx4 and HSPA2 in the RPL group. Functional analysis revealed the involvement of key biological processes that includes spermatogenesis, response to oxidative stress, protein folding and metabolic process.
CONCLUSIONS
The present study provides a snapshot of the altered protein expression levels consistent with the potential involvement of the sperm chromatin landscape in early embryonic development.
Keyword
MeSH Terms
-
Abortion, Spontaneous
Biological Processes
Blotting, Western
Chromatin
Embryo Loss
Embryonic Development
Female
Healthy Volunteers
Humans
Karyotyping
Male
Masturbation
Metabolism
Oxidation-Reduction
Oxidative Stress
Pregnancy*
Prospective Studies
Protein Folding
Proteomics
Sexual Abstinence
Spectrum Analysis
Spermatogenesis
Spermatozoa*
Two-Dimensional Difference Gel Electrophoresis
World Health Organization
Chromatin
Figure
Reference
-
1. Practice Committee of American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril. 2013; 99:63. PMID: 23095139.2. Bareh GM, Jacoby E, Binkley P, Chang TC, Schenken RS, Robinson RD. Sperm deoxyribonucleic acid fragmentation assessment in normozoospermic male partners of couples with unexplained recurrent pregnancy loss: a prospective study. Fertil Steril. 2016; 105:329–336.e1. PMID: 26607021.
Article3. Rogenhofer N, Ott J, Pilatz A, Wolf J, Thaler CJ, Windischbauer L, et al. Unexplained recurrent miscarriages are associated with an aberrant sperm protamine mRNA content. Hum Reprod. 2017; 32:1574–1582. PMID: 28854581.
Article4. Dewan S, Puscheck EE, Coulam CB, Wilcox AJ, Jeyendran RS. Y-chromosome microdeletions and recurrent pregnancy loss. Fertil Steril. 2006; 85:441–445. PMID: 16595224.
Article5. Gil-Villa AM, Cardona-Maya W, Agarwal A, Sharma R, Cadavid A. Assessment of sperm factors possibly involved in early recurrent pregnancy loss. Fertil Steril. 2010; 94:1465–1472. PMID: 19540481.
Article6. Coughlan C, Clarke H, Cutting R, Saxton J, Waite S, Ledger W, et al. Sperm DNA fragmentation, recurrent implantation failure and recurrent miscarriage. Asian J Androl. 2015; 17:681–685. PMID: 25814156.
Article7. Gil-Villa AM, Cardona-Maya W, Agarwal A, Sharma R, Cadavid A. Role of male factor in early recurrent embryo loss: do antioxidants have any effect. Fertil Steril. 2009; 92:565–571. PMID: 18829003.
Article8. Ramasamy R, Scovell JM, Kovac JR, Cook PJ, Lamb DJ, Lipshultz LI. Fluorescence in situ hybridization detects increased sperm aneuploidy in men with recurrent pregnancy loss. Fertil Steril. 2015; 103:906–909.e1. PMID: 25707335.
Article9. Zidi-Jrah I, Hajlaoui A, Mougou-Zerelli S, Kammoun M, Meniaoui I, Sallem A, et al. Relationship between sperm aneuploidy, sperm DNA integrity, chromatin packaging, traditional semen parameters, and recurrent pregnancy loss. Fertil Steril. 2016; 105:58–64. PMID: 26493117.
Article10. Meyer RG, Ketchum CC, Meyer-Ficca ML. Heritable sperm chromatin epigenetics: a break to remember. Biol Reprod. 2017; 97:784–797. PMID: 29099948.
Article11. Mohanty G, Swain N, Goswami C, Kar S, Samanta L. Histone retention, protein carbonylation, and lipid peroxidation in spermatozoa: possible role in recurrent pregnancy loss. Syst Biol Reprod Med. 2016; 62:201–212. PMID: 26980262.
Article12. Simon L, Carrell DT. Sperm DNA damage measured by comet assay. Methods Mol Biol. 2013; 927:137–146. PMID: 22992910.
Article13. World Health Organization. WHO laboratory manual for the examination and processing of human semen. 5th ed. Geneva: World Health Organization;2010.14. Baker MA, Witherdin R, Hetherington L, Cunningham-Smith K, Aitken RJ. Identification of post-translational modifications that occur during sperm maturation using difference in two-dimensional gel electrophoresis. Proteomics. 2005; 5:1003–1012. PMID: 15712234.
Article15. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951; 193:265–275. PMID: 14907713.
Article16. Hamada A, Sharma R, du Plessis SS, Willard B, Yadav SP, Sabanegh E, et al. Two-dimensional differential in-gel electrophoresis-based proteomics of male gametes in relation to oxidative stress. Fertil Steril. 2013; 99:1216–1226. PMID: 23312230.17. Frapsauce C, Pionneau C, Bouley J, Delarouziere V, Berthaut I, Ravel C, et al. Proteomic identification of target proteins in normal but nonfertilizing sperm. Fertil Steril. 2014; 102:372–380. PMID: 24882558.
Article18. Samanta L, Agarwal A, Swain N, Sharma R, Gopalan B, Esteves SC, et al. Proteomic signatures of sperm mitochondria in varicocele: clinical use as biomarkers of varicocele associated infertility. J Urol. 2018; 200:414–422. PMID: 29530785.
Article19. Aoshima K, Inoue E, Sawa H, Okada Y. Paternal H3K4 methylation is required for minor zygotic gene activation and early mouse embryonic development. EMBO Rep. 2015; 16:803–812. PMID: 25925669.
Article20. Govin J, Caron C, Escoffier E, Ferro M, Kuhn L, Rousseaux S, et al. Post-meiotic shifts in HSPA2/HSP70.2 chaperone activity during mouse spermatogenesis. J Biol Chem. 2006; 281:37888–37892. PMID: 17035236.
Article21. Redgrove KA, Anderson AL, McLaughlin EA, O'Bryan MK, Aitken RJ, Nixon B. Investigation of the mechanisms by which the molecular chaperone HSPA2 regulates the expression of sperm surface receptors involved in human sperm-oocyte recognition. Mol Hum Reprod. 2013; 19:120–135. PMID: 23247813.
Article22. Bromfield EG, Aitken RJ, Anderson AL, McLaughlin EA, Nixon B. The impact of oxidative stress on chaperone-mediated human sperm-egg interaction. Hum Reprod. 2015; 30:2597–2613. PMID: 26345691.
Article23. Bironaite D, Pivoriunas A, Venalis A. Upregulation of iHsp70 by mild heat shock protects rabbit myogenic stem cells: involvement of JNK signalling and c-Jun. Cell Biol Int. 2012; 36:1089–1096. PMID: 22946646.
Article24. Jagadish N, Rana R, Selvi R, Mishra D, Garg M, Yadav S, et al. Characterization of a novel human sperm-associated antigen 9 (SPAG9) having structural homology with c-Jun N-terminal kinase-interacting protein. Biochem J. 2005; 389:73–82. PMID: 15693750.
Article25. Barroso G, Valdespin C, Vega E, Kershenovich R, Avila R, Avendaño C, et al. Developmental sperm contributions: fertilization and beyond. Fertil Steril. 2009; 92:835–848. PMID: 19631936.
Article26. Ishizuka M, Ohtsuka E, Inoue A, Odaka M, Ohshima H, Tamura N, et al. Abnormal spermatogenesis and male infertility in testicular zinc finger protein Zfp318-knockout mice. Dev Growth Differ. 2016; 58:600–608. PMID: 27385512.27. Fan S, Zhao Y, Pan Z, Gao Z, Liang Z, Pan Z, et al. ZNF185-derived peptide induces fertility suppression in mice. J Pept Sci. 2018; 24:e3121. PMID: 30270484.
Article28. Meseguer M, de los Santos MJ, Simón C, Pellicer A, Remohí J, Garrido N. Effect of sperm glutathione peroxidases 1 and 4 on embryo asymmetry and blastocyst quality in oocyte donation cycles. Fertil Steril. 2006; 86:1376–1385. PMID: 16979635.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Efficacy on the Immunotherapy with Paternal Lymphocytes in Unexplained Infertility
- Recurrent pregnancy loss: can factor V Leiden mutations be a cause
- The degree of paternal attachment to neonate
- Twin Pregnancy and Delivery After Intracytoplasmic Sperm Injection Followed by Calcium Ionophore with Spermatozoa from a Globozoospermic Man: A Case Report
- Childhood Experiences, Paternal Attachment and Paternal Role of Primiparous Spouses