Lab Anim Res.
2018 Dec;34(4):295-301. 10.5625/lar.2018.34.4.295.
Nucleotide-binding oligomerization domain 1 is dispensable for host immune responses against pulmonary infection of Acinetobacter baumannii in mice
- Affiliations
-
- 1Laboratory Animal Medicine, College of Veterinary Medicine, Chonnam National University, Gwangju, Korea. jonpark@jnu.ac.kr
- KMID: 2459308
- DOI: http://doi.org/10.5625/lar.2018.34.4.295
Abstract
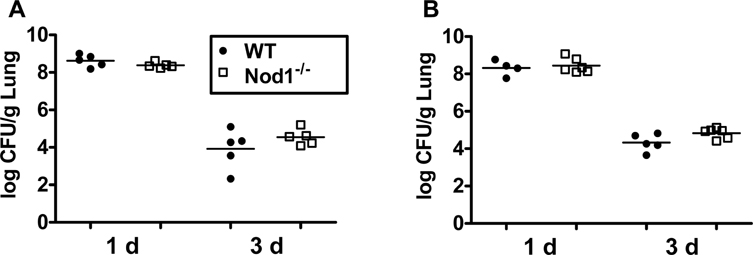
- Nucleotide-binding domain 1 (Nod1) is a cytosolic receptor that is responsible for the recognition of a bacterial peptidoglycan motif containing meso-diaminophimelic acid. In this study, we sought to identify the role of Nod1 in host defense in vivo against pulmonary infection by multidrug resistant Acinetobacter baumannii. Wildtype (WT) and Nod1-deficient mice were intranasally infected with 3×107 CFU of A. baumannii and sacrificed at 1 and 3 days post-infection (dpi). Bacterial CFUs, cytokines production, histopathology, and mouse β-defensins (mBD) in the lungs of infected mice were evaluated. The production of cytokines in response to A. baumannii was also measured in WT and Nod1-deficient macrophages. The bacterial clearance in the lungs was not affected by Nod1 deficiency. Levels of IL-6, TNF-α, and IL-1β in the lung homogenates were comparable at days 1 and 3 between WT and Nod1-deficient mice, except the TNF-α level at day 3, which was higher in Nod1-deficient mice. There was no significant difference in lung pathology and expression of mBDs (mBD1, 2, 3, and 4) between WT and Nod1-deficient mice infected with A. baumannii. The production of IL-6, TNF-α, and NO by macrophages in response to A. baumannii was also comparable in WT and Nod1-deficient mice. Our results indicated that Nod1 does not play an important role in host immune responses against A. baumannii infection.
MeSH Terms
Figure
Reference
-
1. Doughari HJ, Ndakidemi PA, Human IS, Benade S. The ecology, biology and pathogenesis of Acinetobacter spp.: an overview. Microbes Environ. 2011; 26(2):101–112.
Article2. Antunes LC, Visca P, Towner KJ. Acinetobacter baumannii: evolution of a global pathogen. Pathog Dis. 2014; 71(3):292–301.
Article3. McConnell MJ, Actis L, Pachón J. Acinetobacter baumannii: human infections, factors contributing to pathogenesis and animal models. FEMS Microbiol Rev. 2013; 37(2):130–155.
Article4. Scott P, Deye G, Srinivasan A, Murray C, Moran K, Hulten E, Fishbain J, Craft D, Riddell S, Lindler L, Mancuso J, Milstrey E, Bautista CT, Patel J, Ewell A, Hamilton T, Gaddy C, Tenney M, Christopher G, Petersen K, Endy T, Petruccelli B. An outbreak of multidrug-resistant Acinetobacter baumannii-calcoaceticus complex infection in the US military health care system associated with military operations in Iraq. Clin Infect Dis. 2007; 44(12):1577–1584.
Article5. Moradi J, Hashemi FB, Bahador A. Antibiotic Resistance of Acinetobacter baumannii in Iran: A Systemic Review of the Published Literature. Osong Public Health Res Perspect. 2015; 6(2):79–86.
Article6. Kale SD, Dikshit N, Kumar P, Balamuralidhar V, Khameneh HJ, Bin Abdul, Koh TH, Tan GGY, Tan TT, Mortellaro A, Sukumaran B. Nod2 is required for the early innate immune clearance of Acinetobacter baumannii from the lungs. Sci Rep. 2017; 7(1):17429.
Article7. Franchi L, Warner N, Viani K, Nuñez G. Function of Nod-like receptors in microbial recognition and host defense. Immunol Rev. 2009; 227(1):106–128.
Article8. Mathews RJ, Sprakes MB, McDermott MF. NOD-like receptors and inflammation. Arthritis Res Ther. 2008; 10(6):228.
Article9. Caruso R, Warner N, Inohara N, Núñez G. NOD1 and NOD2: signaling, host defense, and inflammatory disease. Immunity. 2014; 41(6):898–908.
Article10. Moreira LO, Zamboni DS. NOD1 and NOD2 Signaling in Infection and Inflammation. Front Immunol. 2012; 3:328.
Article11. Shimada K, Chen S, Dempsey PW, Sorrentino R, Alsabeh R, Slepenkin AV, Peterson E, Doherty TM, Underhill D, Crother TR, Arditi M. The NOD/RIP2 pathway is essential for host defenses against Chlamydophila pneumoniae lung infection. PLoS Pathog. 2009; 5(4):e1000379.
Article12. Lipinski S, Till A, Sina C, Arlt A, Grasberger H, Schreiber S, Rosenstiel P. DUOX2-derived reactive oxygen species are effectors of NOD2-mediated antibacterial responses. J Cell Sci. 2009; 122(pt19):3522–3530.
Article13. Kapetanovic R, Jouvion G, Fitting C, Parlato M, Blanchet C, Huerre M, Cavaillon JM, Adib-Conquy M. Contribution of NOD2 to lung inflammation during Staphylococcus aureus-induced pneumonia. Microbes Infect. 2010; 12(10):759–767.
Article14. Pandey AK, Yang Y, Jiang Z, Fortune SM, Coulombe F, Behr MA, Fitzgerald KA, Sassetti CM, Kelliher MA. NOD2, RIP2 and IRF5 play a critical role in the type I interferon response to Mycobacterium tuberculosis. PLoS Pathog. 2009; 5(7):e1000500.
Article15. Berrington WR, Iyer R, Wells RD, Smith KD, Skerrett SJ, Hawn TR. NOD1 and NOD2 regulation of pulmonary innate immunity to Legionella pneumophila. Eur J Immunol. 2010; 40(12):3519–3527.
Article16. Bist P, Dikshit N, Koh TH, Mortellaro A, Tan TT, Sukumaran B. The Nod1, Nod2, and Rip2 axis contributes to host immune defense against intracellular Acinetobacter baumannii infection. Infect Immun. 2014; 82(3):1112–1122.
Article17. Kale SD, Dikshit N, Kumar P, Balamuralidhar V, Khameneh HJ, Bin Abdul Malik N, Koh TH, Tan GGY, Tan TT, Mortellaro A, Sukumaran B. Nod2 is required for the early innate immune clearance of Acinetobacter baumannii from the lungs. Sci Rep. 2017; 7(1):17429.
Article18. Kim YG, Park JH, Daignault S, Fukase K, Núñez G. Cross-tolerization between Nod1 and Nod2 signaling results in reduced refractoriness to bacterial infection in Nod2-deficient macrophages. J Immunol. 2008; 181(6):4340–4346.
Article19. Kang MJ, Jo SG, Kim DJ, Park JH. NLRP3 inflammasome mediates interleukin-1β production in immune cells in response to Acinetobacter baumannii and contributes to pulmonary inflammation in mice. Immunology. 2017; 150(4):495–505.20. Celada A, Gray PW, Rinderknecht E, Schreiber RD. Evidence for a gamma-interferon receptor that regulates macrophage tumoricidal activity. J Exp Med. 1984; 160(1):55–74.
Article21. March C, Regueiro V, Llobet E, Moranta D, Morey P, Garmendia J, Bengoechea JA. Dissection of host cell signal transduction during Acinetobacter baumannii-triggered inflammatory response. PLoS One. 2010; 5(4):e10033.22. Qiu H, KuoLee R, Harris G, Van Rooijen N, Patel GB, Chen W. Role of macrophages in early host resistance to respiratory Acinetobacter baumannii infection. PLoS One. 2012; 7(6):e40019.
Article23. Chamaillard M, Hashimoto M, Horie Y, Masumoto J, Qiu S, Saab L, Ogura Y, Kawasaki A, Fukase K, Kusumoto S, Valvano MA, Foster SJ, Mak TW, Nuñez G, Inohara N. An essential role for NOD1 in host recognition of bacterial peptidoglycan containing diaminopimelic acid. Nat Immunol. 2003; 4(7):702–707.
Article24. Davis KM, Nakamura S, Weiser JN. Nod2 sensing of lysozyme-digested peptidoglycan promotes macrophage recruitment and clearance of S. pneumoniae colonization in mice. The J Clin Invest. 2011; 121(9):3666–9676.
Article25. Hommes TJ, van Lieshout MH, van 't Veer C, Florquin S, Bootsma HJ, Hermans PW, de Vos AF, van der Poll T. Role of Nucleotide-Binding Oligomerization Domain-Containing (NOD) 2 in Host Defense during Pneumococcal Pneumonia. PLoS One. 2015; 10(12):e0145138.
Article26. Divangahi M, Mostowy S, Coulombe F, Kozak R, Guillot L, Veyrier F, Kobayashi KS, Flavell RA, Gros P, Behr MA. NOD2-deficient mice have impaired resistance to Mycobacterium tuberculosis infection through defective innate and adaptive immunity. J Immunol. 2008; 181(10):7157–7165.
Article27. Lee JY, Lee MS, Kim DJ, Yang SJ, Lee SJ, Noh EJ, Shin SJ, Park JH. Nucleotide-Binding Oligomerization Domain 2 Contributes to Limiting Growth of Mycobacterium abscessus in the Lung of Mice by Regulating Cytokines and Nitric Oxide Production. Front Immunol. 2017; 8:1477.
Article28. Sabbah A, Chang TH, Harnack R, Frohlich V, Tominaga K, Dube PH, Xiang Y, Bose S. Activation of innate immune antiviral responses by Nod2. Nat Immunol. 2009; 10(10):1073–1080.
Article29. Frutuoso MS, Hori JI, Pereira MS, Junior DS, Sônego F, Kobayashi KS, Flavell RA, Cunha FQ, Zamboni DS. The pattern recognition receptors Nod1 and Nod2 account for neutrophil recruitment to the lungs of mice infected with Legionella pneumophila. Microbes Infect. 2010; 12(11):819–827.
Article30. Sônego F, Castanheira FVS, Horta CV, Kanashiro A, Czaikoski PG, Zamboni DS, Alves-Filho JC, Cunha FQ. The host control of a clinical isolate strain of P. aeruginosa infection is independent of Nod-1 but depends on MyD88. Inflamm Res. 2018; 67(5):435–443.
Article31. Baricelli J, Rocafull MA, Vázquez D, Bastidas B, Báez-Ramirez E, Thomas LE. β-defensin-2 in breast milk displays a broad antimicrobial activity against pathogenic bacteria. J Pediatr (Rio J). 2015; 91(1):36–43.
Article32. Kim YG, Park JH, Shaw MH, Franchi L, Inohara N, Núñez G. The cytosolic sensors Nod1 and Nod2 are critical for bacterial recognition and host defense after exposure to Toll-like receptor ligands. Immunity. 2008; 28(2):246–257.
Article33. Park JH, Kim YG, Núñez G. RICK promotes inflammation and lethality after gram-negative bacterial infection in mice stimulated with lipopolysaccharide. Infect Immun. 2009; 77(4):1569–1578.
Article34. Jeong YJ, Kim CH, Kim JC, Oh SM, Lee KB, Park JH, Kim DJ. RIP2/RICK-dependent cytokine production upon Yersinia enterocolitica infection in macrophages with TLR4 deficiency. Scand J Immunol. 2013; 78(5):401–407.35. Kim CH, Jeong YJ, Lee J, Jeon SJ, Park SR, Kang MJ, Park JH, Park JH. Essential role of toll-like receptor 4 in Acinetobacter baumannii-induced immune responses in immune cells. Microb Pathog. 2013; 54:20–25.
Article36. Clarke TB, Davis KM, Lysenko ES, Zhou AY, Yu Y, Weiser JN. Recognition of peptidoglycan from the microbiota by Nod1 enhances systemic innate immunity. Nat Med. 2010; 16(2):228–231.
Article37. Jeong YJ, Kang MJ, Lee SJ, Kim CH, Kim JC, Kim TH, Kim DJ, Kim D, Núñez G, Park JH. Nod2 and Rip2 contribute to innate immune responses in mouse neutrophils. Immunology. 2014; 143(2):269–276.
Article38. Ekman AK, Cardell LO. The expression and function of Nod-like receptors in neutrophils. Immunology. 2010; 130(1):55–63.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cathelicidin-related Antimicrobial Peptide Contributes to Host Immune Responses Against Pulmonary Infection with Acinetobacter baumannii in Mice
- Expression of Nucleotide-oligomerization Domain (NOD) and Related Genes in Mouse Tissues Infected with Mycobacterium leprae
- Successful Treatment of Acinetobacter Baumannii Meningitis with Colistin
- Clinical Features of Acinetobacter Baumannii Keratitis
- NLRP6 as a Negative Regulator of Innate Immunity