Allergy Asthma Immunol Res.
2019 Nov;11(6):818-829. 10.4168/aair.2019.11.6.818.
Association Between Organic Dust Exposure and Adult-Asthma: A Systematic Review and Meta-Analysis of Case-Control Studies
- Affiliations
-
- 1Department of Emergency, Jinan Children's Hospital, Qilu Children's Hospital of Shandong University, Jinan 250022, China. zhangmingming67148@163.com
- KMID: 2459186
- DOI: http://doi.org/10.4168/aair.2019.11.6.818
Abstract
- BACKGROUND
Exposure to organic dust has been widely investigated as a potential risk factor for asthma with different results. To clarify a potential relationship, we performed the present meta-analysis to integrate the results of studies examining the association of organic dust exposure with asthma.
METHODS
A comprehensive literature search in the electronic databases including EMBASE, PubMed and Cochrane Library databases (up to August 2018) was conducted. The adjusted odds ratios (ORs) with corresponding 95% confidence interval (CI) for organic dust exposure and asthma were retrieved and pooled to generate summary effect estimates in Revman 5.2.
RESULTS
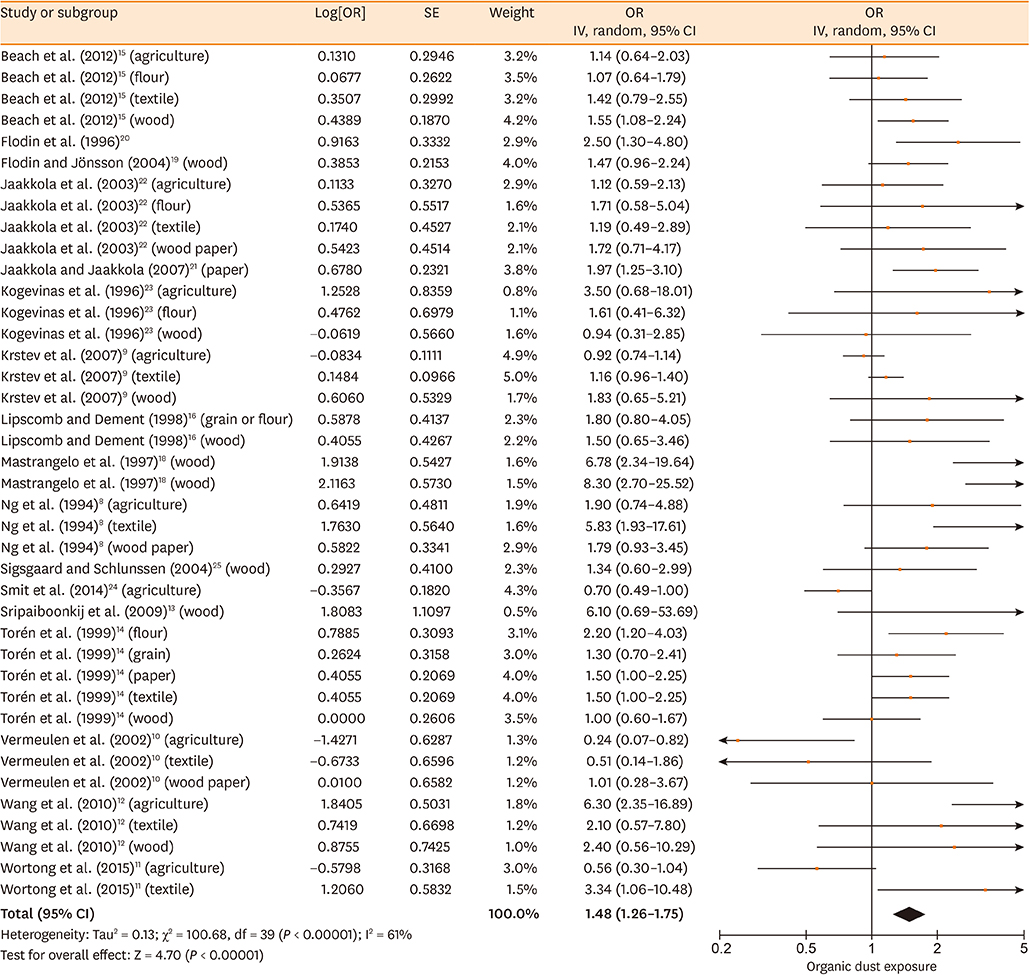
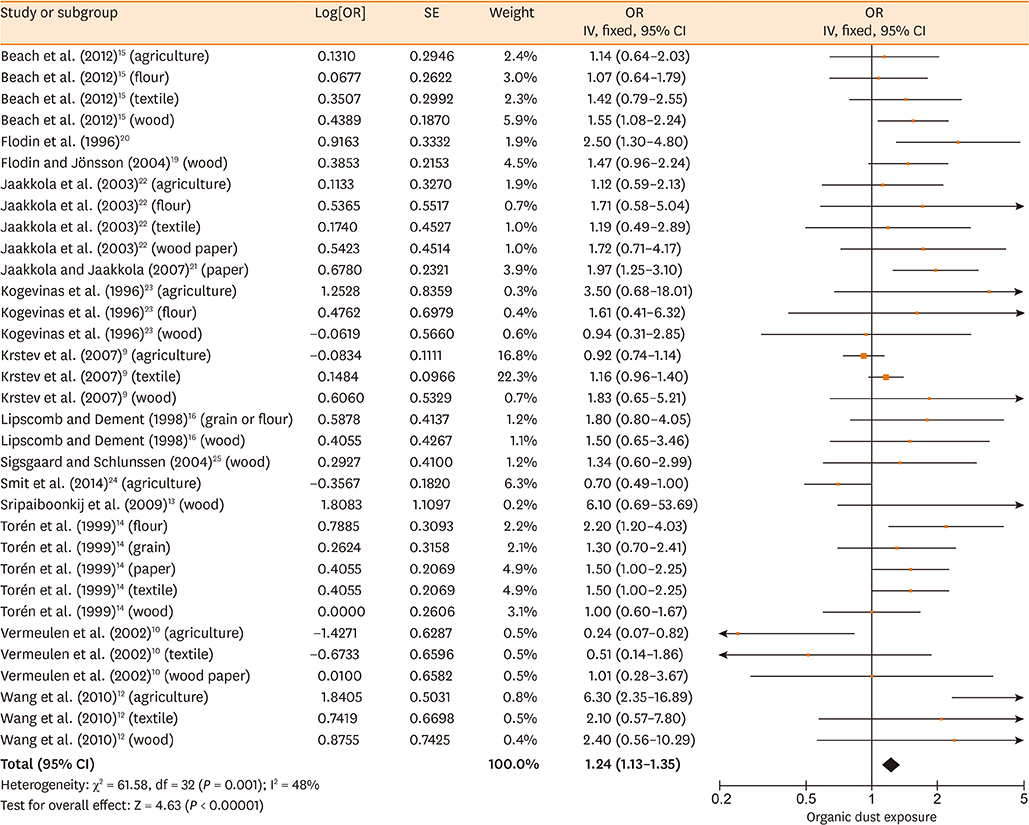
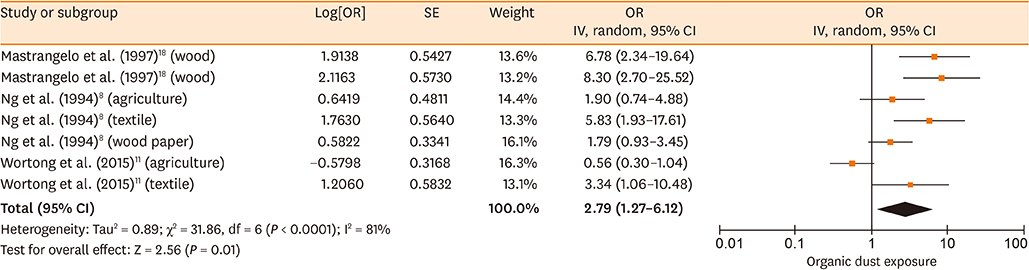
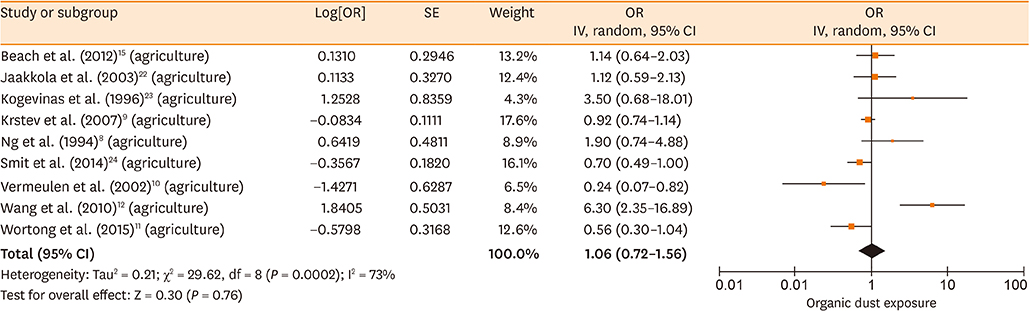
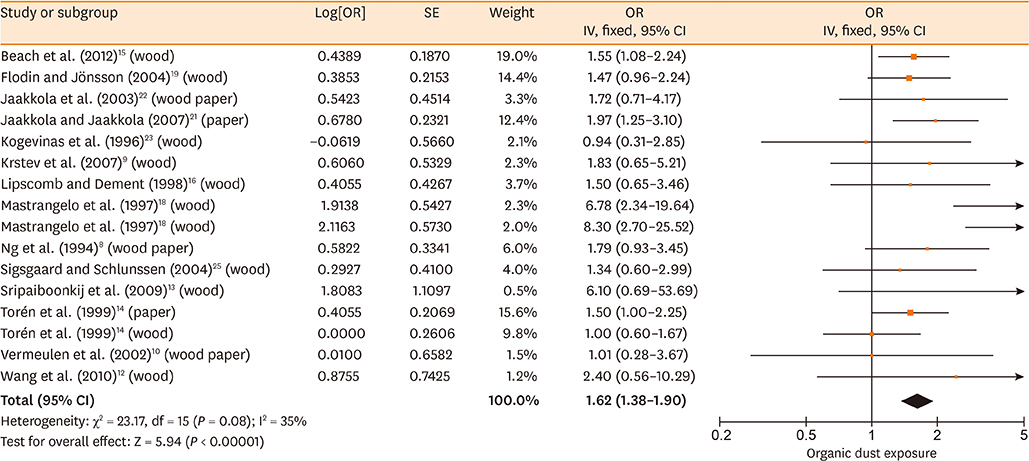
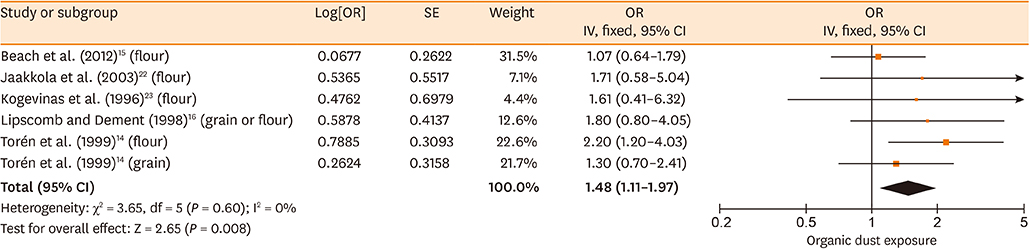
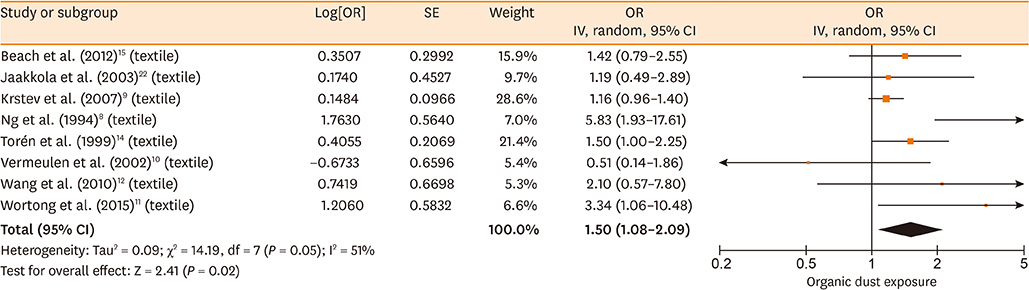
Database searches retrieved 1,016 records. A total of 17 studies containing 3,619 cases and 6,585 controls were finally included in our meta-analysis. The summary estimates suggested that organic dust exposure was positively associated with asthma (OR, 1.48; 95% CI, 1.26-1.75; P < 0.00001), whether among population-based case-control studies (OR, 1.24; 95% CI, 1.13-1.35; P < 0.00001) or hospital-based case-control studies (OR, 2.79; 95% CI, 1.27-6.12; P = 0.01). Subgroup analysis showed that paper/wood (OR, 1.62; 95% CI, 1.38-1.90; P < 0.00001), flour/grain (OR, 1.48; 95% CI, 1.11-1.97; P = 0.008), and textile dust (OR, 1.50; 95% CI, 1.08-2.09; P = 0.02) exposure were significantly associated with asthma.
CONCLUSIONS
Based on the studies evaluated, our meta-analysis results prompt that organic dust exposure is a risk factor inducing asthma, although precise analysis focus on specific organic dust materials is still warranted.
Keyword
Figure
Reference
-
1. Wouters IM, Spaan S, Douwes J, Doekes G, Heederik D. Overview of personal occupational exposure levels to inhalable dust, endotoxin, beta(1-->3)-glucan and fungal extracellular polysaccharides in the waste management chain. Ann Occup Hyg. 2006; 50:39–53.2. Langley RL. Consequences of respiratory exposures in the farm environment. N C Med J. 2011; 72:477–480.
Article3. Millner PD. Bioaerosols associated with animal production operations. Bioresour Technol. 2009; 100:5379–5385.
Article4. Bolund AC, Miller MR, Sigsgaard T, Schlünssen V. The effect of organic dust exposure on long-term change in lung function: a systematic review and meta-analysis. Occup Environ Med. 2017; 74:531–542.
Article5. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010; 25:603–605.
Article6. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–560.
Article7. Gumedze FN, Jackson D. A random effects variance shift model for detecting and accommodating outliers in meta-analysis. BMC Med Res Methodol. 2011; 11:19.
Article8. Ng TP, Hong CY, Goh LG, Wong ML, Koh KT, Ling SL. Risks of asthma associated with occupations in a community-based case-control study. Am J Ind Med. 1994; 25:709–718.
Article9. Krstev S, Ji BT, Shu XO, Blair A, Zheng W, Lubin J, et al. Occupation and adult-onset asthma among Chinese women in a population-based cohort. Am J Ind Med. 2007; 50:265–273.
Article10. Vermeulen R, Heederik D, Kromhout H, Smit HA. Respiratory symptoms and occupation: a cross-sectional study of the general population. Environ Health. 2002; 1:5.
Article11. Wortong D, Chaiear N, Boonsawat W. Risk of asthma in relation to occupation: a hospital-based case-control study. Asian Pac J Allergy Immunol. 2015; 33:152–160.
Article12. Wang TN, Lin MC, Wu CC, Leung SY, Huang MS, Chuang HY, et al. Risks of exposure to occupational asthmogens in atopic and nonatopic asthma: a case-control study in Taiwan. Am J Respir Crit Care Med. 2010; 182:1369–1376.13. Sripaiboonkij P, Phanprasit W, Jaakkola MS. Respiratory and skin effects of exposure to wood dust from the rubber tree Hevea brasiliensis. Occup Environ Med. 2009; 66:442–447.
Article14. Torén K, Järvholm B, Brisman J, Hagberg S, Hermansson BA, Lillienberg L. Adult-onset asthma and occupational exposures. Scand J Work Environ Health. 1999; 25:430–435.
Article15. Beach J, Burstyn I, Cherry N. Estimating the extent and distribution of new-onset adult asthma in British Columbia using frequentist and Bayesian approaches. Ann Occup Hyg. 2012; 56:719–727.16. Lipscomb HJ, Dement JM. Respiratory diseases among union carpenters: cohort and case-control analyses. Am J Ind Med. 1998; 33:131–150.
Article17. Schlünssen V, Schaumburg I, Heederik D, Taudorf E, Sigsgaard T. Indices of asthma among atopic and non-atopic woodworkers. Occup Environ Med. 2004; 61:504–511.18. Mastrangelo G, Bombana S, Priante E, Gallo A, Saia B. Repeated case-control studies as a method of surveillance for asthma in occupations. J Occup Environ Med. 1997; 39:51–57.
Article19. Flodin U, Jönsson P. Non-sensitising air pollution at workplaces and adult onset asthma. Int Arch Occup Environ Health. 2004; 77:17–22.
Article20. Flodin U, Ziegler J, Jönsson P, Axelson O. Bronchial asthma and air pollution at workplaces. Scand J Work Environ Health. 1996; 22:451–456.
Article21. Jaakkola MS, Jaakkola JJ. Office work exposures and adult-onset asthma. Environ Health Perspect. 2007; 115:1007–1011.
Article22. Jaakkola JJ, Piipari R, Jaakkola MS. Occupation and asthma: a population-based incident case-control study. Am J Epidemiol. 2003; 158:981–987.
Article23. Kogevinas M, Antó JM, Soriano JB, Tobias A, Burney P. The risk of asthma attributable to occupational exposures. A population-based study in Spain. Spanish Group of the European Asthma Study. Am J Respir Crit Care Med. 1996; 154:137–143.
Article24. Smit LA, Hooiveld M, van der Sman-de Beer F, Opstal-van Winden AW, Beekhuizen J, Wouters IM, et al. Air pollution from livestock farms, and asthma, allergic rhinitis and COPD among neighbouring residents. Occup Environ Med. 2014; 71:134–140.
Article25. Sigsgaard T, Schlunssen V. Occupational asthma diagnosis in workers exposed to organic dust. Ann Agric Environ Med. 2004; 11:1–7.26. Kim YS, Choi JP, Kim MH, Park HK, Yang S, Kim YS, et al. IgG sensitization to extracellular vesicles in indoor dust is closely associated with the prevalence of non-eosinophilic asthma, COPD, and lung cancer. Allergy Asthma Immunol Res. 2016; 8:198–205.
Article27. Wunschel J, Poole JA. Occupational agriculture organic dust exposure and its relationship to asthma and airway inflammation in adults. J Asthma. 2016; 53:471–477.
Article28. Kim JL, Henneberger PK, Lohman S, Olin AC, Dahlman-Höglund A, Andersson E, et al. Impact of occupational exposures on exacerbation of asthma: a population-based asthma cohort study. BMC Pulm Med. 2016; 16:148.
Article29. Søyseth V, Johnsen HL, Henneberger PK, Kongerud J. The incidence of work-related asthma-like symptoms and dust exposure in Norwegian smelters. Am J Respir Crit Care Med. 2012; 185:1280–1285.
Article30. Garshick E, Laden F, Hart JE, Caron A. Residence near a major road and respiratory symptoms in U.S. veterans. Epidemiology. 2003; 14:728–736.
Article31. Poole JA, Dooley GP, Saito R, Burrell AM, Bailey KL, Romberger DJ, et al. Muramic acid, endotoxin, 3-hydroxy fatty acids, and ergosterol content explain monocyte and epithelial cell inflammatory responses to agricultural dusts. J Toxicol Environ Health A. 2010; 73:684–700.
Article32. Poole JA, Wyatt TA, Von Essen SG, Hervert J, Parks C, Mathisen T, et al. Repeat organic dust exposure-induced monocyte inflammation is associated with protein kinase C activity. J Allergy Clin Immunol. 2007; 120:366–373.
Article33. Poole JA, Wyatt TA, Kielian T, Oldenburg P, Gleason AM, Bauer A, et al. Toll-like receptor 2 regulates organic dust-induced airway inflammation. Am J Respir Cell Mol Biol. 2011; 45:711–719.
Article34. Bauer C, Kielian T, Wyatt TA, Romberger DJ, West WW, Gleason AM, et al. Myeloid differentiation factor 88-dependent signaling is critical for acute organic dust-induced airway inflammation in mice. Am J Respir Cell Mol Biol. 2013; 48:781–789.
Article35. Robbe P, Spierenburg EA, Draijer C, Brandsma CA, Telenga E, van Oosterhout AJ, et al. Shifted T-cell polarisation after agricultural dust exposure in mice and men. Thorax. 2014; 69:630–637.
Article36. Poole JA, Gleason AM, Bauer C, West WW, Alexis N, van Rooijen N, et al. CD11c(+)/CD11b(+) cells are critical for organic dust-elicited murine lung inflammation. Am J Respir Cell Mol Biol. 2012; 47:652–659.37. Robbe P, Draijer C, Borg TR, Luinge M, Timens W, Wouters IM, et al. Distinct macrophage phenotypes in allergic and nonallergic lung inflammation. Am J Physiol Lung Cell Mol Physiol. 2015; 308:L358–67.
Article38. Poole JA, Gleason AM, Bauer C, West WW, Alexis N, Reynolds SJ, et al. αβ T cells and a mixed Th1/Th17 response are important in organic dust-induced airway disease. Ann Allergy Asthma Immunol. 2012; 109:266–273.e2.
Article39. Guillam MT, Martin S, Le Guelennec M, Puterflam J, Le Bouquin S, Huneau-Salaün A. Dust exposure and health of workers in duck hatcheries. Ann Agric Environ Med. 2017; 24:360–365.40. Hinson AV, Lokossou VK, Schlünssen V, Agodokpessi G, Sigsgaard T, Fayomi B. Cotton dust exposure and respiratory disorders among textile workers at a textile company in the southern part of Benin. Int J Environ Res Public Health. 2016; 13:E895.
Article41. Christiani DC, Ye TT, Zhang S, Wegman DH, Eisen EA, Ryan LA, et al. Cotton dust and endotoxin exposure and long-term decline in lung function: results of a longitudinal study. Am J Ind Med. 1999; 35:321–331.
Article42. Glindmeyer HW, Lefante JJ, Jones RN, Rando RJ, Abdel Kader HM, Weill H. Exposure-related declines in the lung function of cotton textile workers. Relationship to current workplace standards. Am Rev Respir Dis. 1991; 144:675–683.
Article43. Dimich-Ward H, Beking K, DyBuncio A, Chan-Yeung M, Du W, Karlen B, et al. Occupational exposure influences on gender differences in respiratory health. Lung. 2012; 190:147–154.
Article44. Viegas S, Faísca VM, Dias H, Clérigo A, Carolino E, Viegas C. Occupational exposure to poultry dust and effects on the respiratory system in workers. J Toxicol Environ Health A. 2013; 76:230–239.
Article45. Parsons MA, Beach J, Senthilselvan A. Association of living in a farming environment with asthma incidence in Canadian children. J Asthma. 2017; 54:239–249.
Article46. Kääriö H, Nieminen JK, Karvonen AM, Huttunen K, Schröder PC, Vaarala O, et al. Circulating dendritic cells, farm exposure and asthma at early age. Scand J Immunol. 2016; 83:18–25.
Article47. Viegas S, Caetano LA, Korkalainen M, Faria T, Pacífico C, Carolino E, et al. Cytotoxic and inflammatory potential of air samples from occupational settings with exposure to organic dust. Toxics. 2017; 5:E8.
Article48. Hoppe Parr KA, Hađina S, Kilburg-Basnyat B, Wang Y, Chavez D, Thorne PS, et al. Modification of sample processing for the Limulus amebocyte lysate assay enhances detection of inflammogenic endotoxin in intact bacteria and organic dust. Innate Immun. 2017; 23:307–318.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Organic solvent exposure for the chronic kidney disease: updated systematic review with meta-analysis
- Exposure to Dichlorodiphenyltrichloroethane and the Risk of Breast Cancer: A Systematic Review and Meta-analysis
- Clinical Year-in-Review of Occupational Lung Disease
- Systematic Review and Meta-analysis in Digestive Cancer Research
- An Introduction of the Systematic Review and Meta-Analysis