Transl Clin Pharmacol.
2019 Jun;27(2):80-85. 10.12793/tcp.2019.27.2.80.
Comparison of pharmacokinetic characteristics of two Tegoprazan (CJ-12420) formulations in healthy male subjects
- Affiliations
-
- 1Department of Clinical Pharmacology and Therapeutics, Seoul National University College of Medicine and Hospital, Seoul 03080, Republic of Korea.
- 2Clinical Development Division, CJ HealthCare Corp., Seoul 04551, Republic of Korea.
- 3Department of Pharmacology, School of Medicine, Chonbuk National University, Jeonju 54907, Republic of Korea. mgkim@jbnu.ac.kr
- KMID: 2457576
- DOI: http://doi.org/10.12793/tcp.2019.27.2.80
Abstract
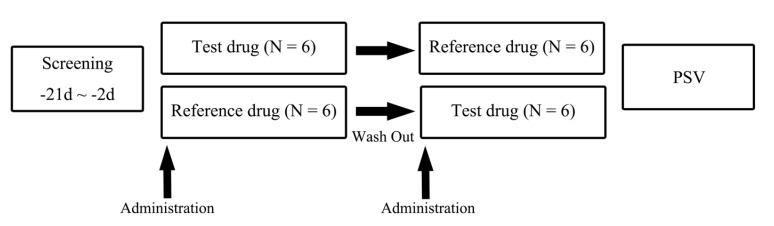
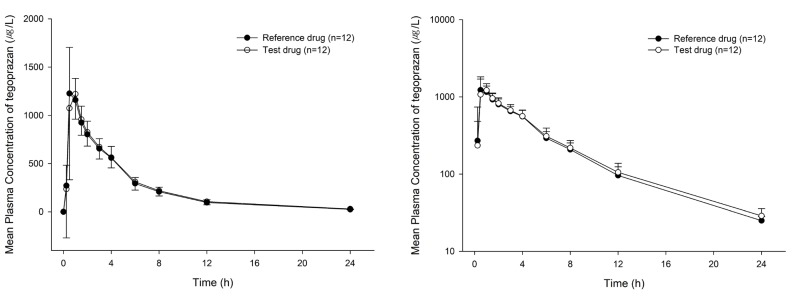
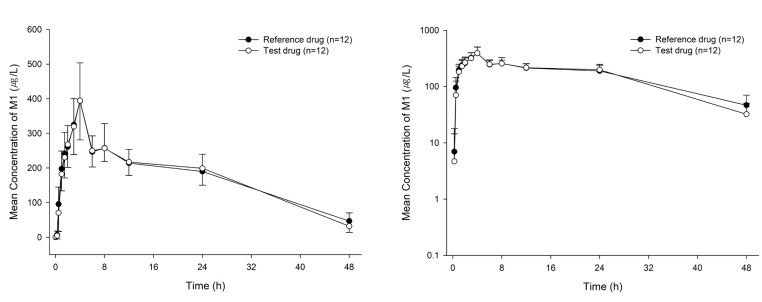
- Proton-pump inhibitors (PPIs) are effectively used to treat acid-related diseases, including gastroesophageal reflux disease (GERD); however, many unmet medical needs still exist. As a new treatment option, potassium-competitive acid blockers (P-CABs), such as tegoprazan, have been developed. This study was performed to compare the pharmacokinetics (PKs) between two formulations (test and reference drugs) of tegoprazan 100 mg tablets. A randomized, single oral dose, two-treatment, two-period, two-sequence study was conducted with 12 healthy subjects. Each subject received the test drug or reference drug in the first period and the alternative treatment in the second period. For PK evaluation, blood samples were collected up to 48 hours post-dose in each period. The plasma concentrations of tegoprazan and its active metabolite (M1) were measured by liquid chromatography-tandem mass spectrometry. PK parameters, including maximum plasma concentration (C(max)) and area under the concentration-time curve from zero to the last measurable time (AUC(last)), were estimated using a non-compartmental method. The plasma concentration-time profiles of the two formulations were comparable. The geometric mean ratios [90% confidence intervals (CIs)] of the test drug to the reference drug for C(max) and AUC(last) were 0.98 (0.85-1.12) and 1.03 (0.93-1.13), respectively. The corresponding values of M1 were 0.99 (0.89-1.11) and 1.01 (0.93-1.09), respectively. The two formulations of tegoprazan exhibited comparable PK profiles, fulfilling the regulatory criteria for bioequivalence.
Keyword
MeSH Terms
Figure
Reference
-
1. Maradey-Romero C, Fass R. New and future drug development for gastroesophageal reflux disease. J Neurogastroenterol Motil. 2014; 20:6–16. DOI: 10.5056/jnm.2014.20.1.6. PMID: 24466441.2. Schubert ML, Peura DA. Control of gastric acid secretion in health and disease. Gastroenterology. 2008; 134:1842–1860. DOI: 10.1053/j.gastro.2008.05.021. PMID: 18474247.
Article3. Jang HJ, Choi MH, Shin WG, Kim KH, Chung YW, Kim KO, et al. Has peptic ulcer disease changed during the past ten years in Korea? A prospective multi-center study. Dig Dis Sci. 2008; 53:1527–1531. PMID: 17932759.
Article4. Arikawa Y, Nishida H, Kurasawa O, Hasuoka A, Hirase K, Inatomi N, et al. Discovery of a novel pyrrole derivative 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1 H-pyrrol-3-yl]-N-methylmethanamine fumarate (TAK-438) as a potassium-competitive acid blocker (P-CAB). J Med Chem. 2012; 55:4446–4456. DOI: 10.1021/jm300318t. PMID: 22512618.5. Jeon HK, Kim GH. Can Nocturnal Acid-breakthrough Be Reduced by Long-acting Proton Pump Inhibitors? J Neurogastroenterol Motil. 2017; 23:145–148. DOI: 10.5056/jnm17037. PMID: 28372039.
Article6. Tytgat GN. Shortcomings of the first-generation proton pump inhibitors. Eur J Gastroenterol Hepatol. 2001; 13:S29–S33. PMID: 11430506.7. Hori Y, Imanishi A, Matsukawa J, Tsukimi Y, Nishida H, Arikawa Y, et al. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther. 2010; 335:231–238. DOI: 10.1124/jpet.110.170274. PMID: 20624992.
Article8. Hatlebakk JG, Katz PO, Camacho-Lobato L, Castell DO. Proton pump inhibitors: better acid suppression when taken before a meal than without a meal. Aliment Pharmacol Ther. 2000; 14:1267–1272. PMID: 11012470.
Article9. Teixeira MZ. Rebound acid hypersecretion after withdrawal of gastric acid suppressing drugs: new evidence of similitude. Homeopathy. 2011; 100:148–156. DOI: 10.1016/j.homp.2011.05.003. PMID: 21784332.
Article10. Hunt RH, Scarpignato C. Potassium-competitive acid blockers (P-CABs): are they finally ready for prime time in acid-related disease? Clin Transl Gastroenterol. 2015; 6:e119. DOI: 10.1038/ctg.2015.39. PMID: 26513137.
Article11. Han Sung pil. Safety, tolerablity, pharmacokinetics, and pharmacodynamics of escalating single and multiple oral doses of tegoprazan, a Novel potassium-competive acid blocker (P_CAB) in healthy male subjects. KDDW 2017, P-UG-075.12. Kagami T, Sahara S, Ichikawa H, Uotani T, Yamade M, Sugimoto M, et al. Potent acid inhibition by vonoprazan in comparison with esomeprazole, with reference to CYP2C19 genotype. Aliment Pharmacol Ther. 2016; 43:1048–1059. DOI: 10.1111/apt.13588. PMID: 26991399.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of pharmacokinetic drugdrug interaction between tegoprazan and clarithromycin in healthy subjects
- Comparison of Pharmacodynamics between Tegoprazan and Dexlansoprazole Regarding Nocturnal Acid Breakthrough: A Randomized Crossover Study
- Pharmacokinetics and Safety of Two Voriconazole Formulations after Intravenous Infusion in Healthy Korean Volunteers
- Pharmacokinetic properties and bioequivalence of gefitinib 250 mg in healthy Korean male subjects
- Pharmacokinetics comparison of solifenacin tartrate and solifenacin succinate: a randomized, open-label, single-dose, 2-way crossover study in healthy male volunteers